Abstract
A 38-year-old woman was admitted to hospital because of fever and headache. Increased cerebrospinal cell count and protein without evidence of infection led to a diagnosis of aseptic meningitis. Although she improved with acyclovir and glyceol, she experienced left forearm pain and sensory disturbance with drop fingers. Poor derivation of compound muscle action potentials in the left radial nerve was observed, leading to a diagnosis of mononeuritis multiplex with sensorimotor neuropathy. Because the patient had primary Sjögren’s syndrome with anti-Ro/SS-A antibody and salivary gland hypofunction, treatment with methylprednisolone, intravenous immunoglobulin, and intravenous cyclophosphamide was followed by oral glucocorticoid therapy. After these intensive therapies, her drop fingers gradually improved, although sensory disturbance remained. In conclusion, we report a case of aseptic meningitis and subsequent mononeuritis multiplex that was successfully treated with intensive immunotherapy in a patient with primary Sjögren’s syndrome.
Keywords
Introduction
Primary Sjögren’s syndrome (SS) has glandular and extraglandular manifestations with the appearance of autoantibodies, including anti-Ro/SS-A antibody.1,2 With regard to extraglandular manifestations, the European Alliance of Associations for Rheumatology (EULAR) SS Disease Activity Index 3 provides 12 evaluation categories. Among these categories, aseptic meningitis is involved in lymphocytic meningitis as a form of central nerve damage, and mononeuritis multiplex is classified as peripheral nerve damage. Given that the presence of aseptic meningitis and mononeuritis multiplex is classified as a highly active stage in the clinical symptoms of primary SS, the management of these manifestations is crucial. Brito-Zerón et al. 4 reported that 55 of 563 (9.7%) primary SS patients showed peripheral neuropathy; among these 55 patients, sensorimotor polyneuropathy (n = 24, 43.6%), pure sensory neuropathy (n = 15, 27.2%), and mononeuritis multiplex (n = 15, 27.2%) were observed. Although cases of aseptic meningitis in primary SS have been sporadically reported, to the best of our knowledge, there are no published descriptions of a patient with primary SS with both aseptic meningitis and mononeuritis multiplex as neurological manifestations. Herein, we report the details of an extremely rare case of aseptic meningitis followed by mononeuritis multiplex in a patient with primary SS.
Case presentation
A 38-year-old woman was admitted to Nihon University Itabashi Hospital because of a body temperature of 38°C, headache, and vomiting without photosensitivity, skin rash, oral ulcer, or arthritis. The patient had no intake of non-steroidal anti-inflammatory drugs, no evidence of prior infection (such as herpes infection), and no head trauma. Thirty-one days before developing the fever, she had received her third messenger RNA-based BNT162b2 vaccination against severe acute respiratory syndrome coronavirus-2. The patient showed jolt accentuation without Kernig’s sign or any other neurological abnormalities in terms of consciousness, mental status, cranial nerves, motor/sensory nerves, and coordinated movement. The day after the onset of headache, she began to feel sensory disturbances as neuralgia in both forearms. Although no abnormalities were revealed by brain computed tomography, lumbar puncture revealed that her cerebrospinal fluid (CSF) had an elevated cell count (6/µL), total protein (46 mg/dL; normal range <40 mg/dL), and initial pressure (260 mmH2O; normal range 70–180 mmH2O). Brain magnetic resonance imaging was not performed. Negative results were obtained in a bacterial/mycobacterial culture and in polymerase chain reactions for Mycobacterium tuberculosis, herpes simplex virus, varicella zoster herpes virus, and 15 other types of CSF microorganisms (including enterovirus). A smear test for Mycobacterium and a 6-week liquid culture of M. tuberculosis using the patient’s CSF were also negative.
Negative results were obtained in examinations including repeated blood culture, urine bacterial culture, polymerase chain reaction for severe acute respiratory syndrome coronavirus-2, cytomegalovirus antigenemia, and serum immunoglobulin (Ig)M antibodies including adenovirus, herpes simplex virus, toxoplasma, cytomegalovirus, and parvovirus B19. Although antibody to Epstein–Barr virus nuclear antigen was positive, anti-early antigen diffuse type and restricted type antibody was negative. Positive virus capsid antigen IgG and negative virus capsid antigen IgM were also detected, indicating no evidence of primary infection or reactivation of Epstein–Barr virus. A urinalysis showed no protein or occult blood, and a blood test showed a decreased white blood cell count (2400/µL) without the appearance of atypical lymphocytes, with hemoglobin 10.2 g/dL and platelet count 209,000/µL. We also observed normal aspartate aminotransferase, alanine aminotransferase, and serum creatinine, but slightly elevated lactate dehydrogenase (356 U/L; normal range 124–322 U/L) and C-reactive protein (4.20 mg/dL; normal range <0.2 mg/dL).
Because the patient was diagnosed with aseptic meningitis based on her elevated CSF cell count and total protein without the detection of any CSF microorganisms, we administered acyclovir for 5 days, starting 2 days after the appearance of sensory disturbance of the forearms. Additionally, we administered 8 days of glyceol, and acetaminophen (200 mg) was used as needed. The patient’s headache improved, and her C-reactive protein value became negative on day 7 of hospitalization. When screening for underlying diseases, the patient’s antinuclear antibody 80× (nucleolar and speckled pattern) and anti-Ro/SS-A antibody were highly positive (145 U/mL; normal range <10 U/mL), without rheumatoid factor, double strand deoxyribonucleic acid antibody, anti-Sm antibody, anti-ribonucleoprotein antibody, anti-IgM/IgG cardiolipin antibodies, anti-β2-glycoprotein I antibody, lupus anticoagulant, myeloperoxidase/proteinase-3-anti-neutrophil cytoplasmic antibodies, abnormal complements, or cryoglobulin.
The patient exhibited no xerostomia or xerophthalmia, but primary SS was suspected because of the presence of anti-Ro/SS-A antibody. The possibility of systemic lupus erythematosus was excluded by clinical and laboratory findings including urinalysis and a serum test. The patient’s case fulfilled the diagnostic criteria for primary SS issued by Japan's Ministry of Health, Labour and Welfare 5 based on her positive gum test result (5 mL/10 minutes; positive volume <10 mL) and positive technetium-99m salivary gland scintigraphy with anti-Ro/SS-A antibody. Although the Schirmer’s test was negative (right, 25 mm; left, 20 mm; 5 minutes), a fluorescein staining test result was positive (right, 3; left, 1 on cornea only). The patient refused to undergo a salivary gland biopsy.
Two weeks after the onset of aseptic meningitis and sensory disturbance of both forearms, the patient’s fever, headache, and vomiting had disappeared, but she presented left drop fingers. Drop fingers refer to the inability to actively extend the fingers and thumb. Neurologically, damage to the bilateral medial cutaneous nerves of the forearm and left posterior interosseous nerve paralysis were suspected. A manual muscle test showed weakness of the extensor digitorum muscle only; ganglia were not detected and there was no history of trauma. Magnetic resonance imaging showed no spinal cord compression or intramedullary abnormal signals. A nerve conduction velocity test (NCV) including a compound muscle action potential test for the left radial nerves showed poor derivation of amplitudes without any conduction block (Table 1). These findings led to a diagnosis of mononeuritis multiplex with axonal damage. The patient’s clinical symptoms and neurological findings, as well as the results of her NCV examination, confirmed the asymmetrical distribution of motor-sensory neuropathy. We therefore diagnosed the patient with mononeuritis multiplex rather than polyneuropathy occurring in Sjögren’s syndrome.
Nerve conduction study results.
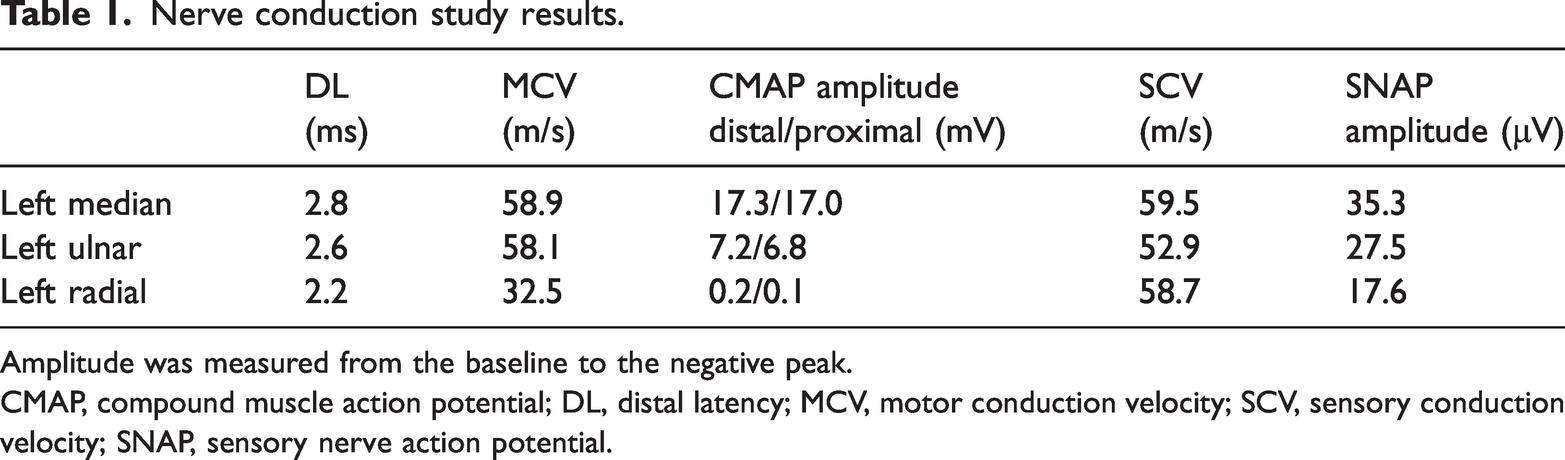
Amplitude was measured from the baseline to the negative peak.
CMAP, compound muscle action potential; DL, distal latency; MCV, motor conduction velocity; SCV, sensory conduction velocity; SNAP, sensory nerve action potential.
Notably, the NCV study excluding the radial nerves showed no abnormalities. In addition, no prolongation of the distal latency of the left radial nerve was observed on an NCV study, which is consistent with mononeuritis multiplex. Because highly active peripheral neuropathy was considered even though the patient’s aseptic meningitis had improved at this point, we first administered a set of methylprednisolone (mPSL) pulse therapy and subsequent intravenous Ig (IVIg) followed by oral glucocorticoids (Figure 1). Because the patient was scheduled to undergo gynecological surgery for a borderline tumor in the left ovary, we also administered 0.5 mg/kg of oral prednisolone after the first and second sets of mPSL/IVIg, followed by a first administration of intravenous cyclophosphamide (IVCY) for mononeuritis multiplex after the gynecological surgery.

Clinical course of the patient’s neurological symptoms. Sensory disturbance of the forearms appeared immediately after the emergence of aseptic meningitis symptoms; subsequently, drop fingers appeared. Two sets of mPSL pulse therapy and IVIg followed by IVCY were administered for the symptoms of mononeuritis multiplex. CRP, C-reactive protein; IVCY, intravenous cyclophosphamide; IVIg, intravenous immunoglobulin; mPSL, methylprednisolone; NCV, nerve conduction velocity.
The patient’s sensory disturbance was improved after the first mPSL administration, but continued regardless of additional mPSL or IVCY (Figure 1). When the patient visited as an outpatient after the IVCY treatment, the active range of motion of her metacarpophalangeal and proximal interphalangeal joints of the II to IV left fingers was improved by keeping the fingers in a horizontal position. Furthermore, although the compound muscle action potential amplitude of her left radial nerve was poorly evoked on motor nerve conduction velocity at the first study, it was detected after intensive immunotherapy; the sensory nerve action potential was unchanged by treatment. Her oral prednisolone was reduced to 7 mg/day.
Discussion
In the present report, we have detailed the appearance of mononeuritis multiplex after aseptic meningitis in a patient with primary SS. The pathological mechanism involved in the central nervous system in primary SS remains unclear, and some concerns need to be addressed. Kurotaki et al. 6 identified a high anti-Ro/SS-A antibody level in an aseptic meningitis patient with primary SS, suggesting that the local production of anti-Ro/SS-A antibody is related to the onset of meningitis. Furthermore, Akiyama et al. 7 reported a primary SS patient with aseptic meningitis as an initial manifestation. Interestingly, they noted that 70% of the published cases of aseptic meningitis or aseptic meningoencephalitis occur before or simultaneously to the onset of primary SS. In our case, the headache improved with the administration of acyclovir, despite the CSF polymerase chain reaction results suggesting that herpes viruses were unlikely to be involved. Regarding Epstein–Barr virus, it has been reported 8 that it is commonly found in the CSF in aseptic meningitis, although caution is required when considering it as a causative agent of aseptic meningitis. However, no reactivation of Epstein–Barr virus was suggested in our case because the patient was negative for anti-early antigen diffuse type and restricted type IgG.
Peripheral neuropathy, such as sensory neuropathy, has frequently been observed in primary SS. However, multiple mononeuritis appears to be relatively rare in peripheral neuropathy; it has been reported that 11 of 92 (12%) patients with primary SS with peripheral neuropathy had mononeuritis multiplex, 9 and the authors suggested the possibility of vasculitis in such cases of mononeuritis multiplex. Another research group noted that mononeuritis multiplex was observed in only 6% of cases of peripheral neuropathy in a series of primary SS patients. 10 Their report also described the existence of vasculitis in nerve biopsies. A case of drug-induced peripheral neuropathy involving acyclovir has also been reported. 11 However, sensory disturbance in the forearm appeared before acyclovir administration in our patient, indicating that this symptom does not correspond to drug-induced neuropathy. Furthermore, although chemotherapy for ovarian tumors can cause peripheral neuropathy, 12 our patient only underwent surgery. A previous report 13 showed that peripheral neuropathy other than mononeuritis multiplex is associated with histologically confirmed ovarian dysgerminoma. In our case, it was mononeuritis multiplex, but the possibility of peripheral neuropathy associated with ovarian tumor remains.
Considering pathogeneses that may be shared by aseptic meningitis and mononeuritis multiplex, it is difficult to suggest a common mechanism because the pathology of mononeuritis multiplex can be explained by vasculitis, whereas the pathology of aseptic meningitis is not fully understood. However, we can speculate about the induction of vasculitis by viral infection. Polyarteritis nodosa that shows mononeuritis multiplex is an acute symptom of hepatitis B infection. 14 Its mechanism of onset is the formation of an immune complex by hepatitis B surface antigen and anti-hepatitis B surface antibody, which deposits on the arterial wall to induce vasculitis. Given that aseptic meningitis is also caused by viral infection, it is conceivable that a similar onset mechanism of vasculitis is involved.
Mononeuritis multiplex is typically considered a form of vasculitis, and its treatment strategy is glucocorticoid-based management followed by treatment with an immunosuppressant such as cyclophosphamide. One study reported that, among 100 cases of vasculitic peripheral neuropathy, 54 were successfully treated with glucocorticoid and IVCY. 15 In addition, the 1-year survival was 90.3%, suggesting that glucocorticoid-based treatment is useful in vasculitic neuropathy.
The EULAR management recommendations for SS 16 provide a treatment strategy for peripheral neuropathy. The management of axonal peripheral neuropathy involves IVIg, mPSL, and cyclophosphamide as a rescue therapy, and a glucocorticoid, oral immunosuppressant, or rituximab and cyclophosphamide/plasma exchange as rescue therapies. In our patient’s case, we selected IVIg, mPSL, and IVCY to treat her mononeuritis multiplex. Although rituximab is not permitted for the treatment of primary SS in Japan, other immunosuppressants or plasma exchange may be candidate treatments if systemic vasculitis is suspected in cases such as that of our patient’s.
There are some limitations to the present report. It remains unclear whether our patient’s case fulfills the American College of Rheumatology/EULAR classification criteria (2016) for SS 17 because we were unable to perform a minor salivary gland biopsy to obtain pathological findings. Moreover, we did not determine a common pathogenesis of aseptic meningitis and mononeuritis multiplex in the current study. When diagnosing aseptic meningitis, enterovirus and varicella zoster virus are major causative viruses. 18 However, many viruses cannot be identified by standard tests. Furthermore, anti-inflammatory drugs and antibiotics have also been reported to cause aseptic meningitis, 19 and should be noted.
In summary, we have provided a report of aseptic meningitis as an initial manifestation of primary SS that was confirmed by Japanese diagnostic criteria, and the subsequent development of mononeuritis multiplex within the short term. The patient’s mononeuritis multiplex was successfully treated using a multidisciplinary strategy. We must pay close attention to neurological symptoms that are preceded by a diagnosis of primary SS.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231189121 - Supplemental material for Aseptic meningitis followed by mononeuritis multiplex in a patient with primary Sjögren’s syndrome
Supplemental material, sj-pdf-1-imr-10.1177_03000605231189121 for Aseptic meningitis followed by mononeuritis multiplex in a patient with primary Sjögren’s syndrome by Hideki Nakamura, Yutaka Tanikawa, Masahiro Nishihara, Masako Tsukamoto, Yosuke Nagasawa, Kumiko Akiya, Naotoshi Natori, Noboru Kitamura, Tadateru Takayama and Hideto Nakajima in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethical approval
The patient provided written informed consent for the publication of her data. No ethical approval was required for this report based on the journal’s instruction. The reporting of this study conforms to CARE guidelines (for CAse REports). 20
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
