Abstract
Objective
Aldo-keto reductase family 1 member B10 (AKR1B10) is a protein that is produced and secreted by a significant number of breast cancers. However, a potential confounder to the use of AKR1B10 as a tumor marker is its elevation in patients given cytotoxic chemotherapy. We therefore conducted a prospective study to analyze AKR1B10 levels in patients with breast cancer receiving neoadjuvant cytotoxic chemotherapy.
Methods
The study enrolled 10 patients from November 2015 to July 2017. All patients had locally advanced, but non-metastatic, breast cancer, and they received neoadjuvant chemotherapy followed by surgery. Serum AKR1B10 levels and tumor imaging were assessed before, during, and after chemotherapy.
Results
No increase in serum AKR1B10 levels was noted in patients receiving chemotherapy whose levels were elevated at diagnosis.
Conclusion
The findings are complex, but the overall data suggest that AKR1B10 is suitable as a tumor marker in patients with elevated levels at the time of diagnosis.
Keywords
Introduction
An estimated 284,200 new invasive breast cancers were diagnosed in the United States in 2021, approximately 34% of which required adjuvant chemotherapy. 1 Through decades of efforts, the therapeutic targets of breast cancer, such as estrogen receptor (ER), have been identified and characterized, 2 but highly useful serum markers remain unavailable.3,4 Clinically available serum markers in breast cancer include carcinoembryonic antigen (CEA) and cancer antigen 27.29 (CA27.29, mostly used in North America)/CA15.3 (mostly used in Europe), but their sensitivity and specificity are considered low.5,6 Serum CA27.29 levels can also be increased by pregnancy, lactation, and inflammatory diseases, as well as ovarian, lung, and prostate cancers.5,7 The American Society of Clinical Oncology (ASCO) has not recommended a tumor marker for screening and staging of breast cancer or for monitoring treatment response or recurrence after breast cancer therapy. 7 Serum cytokeratin fragment 21-1 (CYFRA 21-1), a serum marker of lung cancer, 8 is elevated in patients with metastatic breast cancer. 9 However, it is not yet recommended by ASCO as a serum marker of breast cancer, likely because of its elevation in other conditions including several other malignancies. 7 Therefore, there is a need to identify and validate novel serum markers of breast cancer. The uses of such a marker could include screening, prognosis, tumor burden assessment in patients with metastasis, treatment response assessment, and/or recurrence detection.
Aldo-keto reductase family 1 member B10 (AKR1B10) is primarily expressed in the human colon and small intestine, but it upregulated in hepatocellular carcinoma 10 and non-small cell lung cancer. 11 Our recent studies revealed that AKR1B10 is overexpressed in primary, metastatic, and recurrent cancers of the breast. 12 In 50 pairs of primary breast cancers and matched normal tissues, AKR1B10 protein expression was increased by 2- to 10-fold in 19 (38.0%) tumors and by >10-fold in 23 (46.0%) tumors (maximum increase: 546.7-fold) compared with that in matched normal tissues. AKR1B10 expression in primary breast cancers was confirmed by tissue microarray studies. In the YTMA-23 array (Yale Tissue Microarray-23, Yale University at New Haven), AKR1B10 was expressed in 184 of 220 (83.6%) breast tumors. Furthermore, we found that AKR1B10 was expressed in ductal carcinoma in situ (71.4%, n = 28), metastatic cancer cells in lymph nodes (78.0%, n = 50), and recurrent breast cancer (87.5%, n = 32). 12 An extremely important finding from our laboratory is that AKR1B10 is a secretory protein 13 and a potential serum marker of breast cancer. 12 We found that AKR1B10 protein is secreted by cultured breast cancer cells. Its secretion was stimulated by temperature, ATP, Ca2+, Mg2+, and lysosomotropic NH4Cl, but it was not affected by the protein synthesis inhibitor cycloheximide or classical protein secretion pathway inhibitor brefeldin A. In cultured cells, AKR1B10 protein is present in secretory lysosomes and secreted together with the lysosomal marker cathepsin D. Therefore, AKR1B10 is secreted through a lysosome-mediated secretion pathway. 13
It should be noted that a potential confounder to the use of AKR1B10 as a tumor marker is prior reports of its elevation in patients given cytotoxic chemotherapy. Hashimoto et al. 14 obtained bladder cancer specimens from 10 patients before and after chemotherapy and measured AKR1B10 mRNA levels using real-time PCR. In addition, they conducted an immunohistochemical examination of AKR1B10 expression in 57 patients with bladder cancer before and after chemotherapy. They noted that AKR1B10 mRNA expression was significantly higher in the post-chemotherapy group than in the pre-chemotherapy group (P < 0.001). In addition, both Loffler-Ragg et al. 15 and Ebert et al. 16 reported the induction of AKR1B10 in colon cancer cell lines treated with the proteasome inhibitor bortezomib. However, these studies only investigated cell lines or tissues, but they did not monitor serum AKR1B10 changes. Therefore, we analyzed serum AKR1B10 levels in patients with breast cancer as they underwent treatment with cytotoxic chemotherapy to determine if prior data in other tumor types are valid in clinical breast cancer. Serum AKR1B10 levels in the neoadjuvant setting provided an ideal model to test this hypothesis because serum AKR1B10 can be monitored before, during, and after treatment. In addition, the correlation of changes in tumor size with serum AKR1B10 levels was examined to determine the confounding impact of cytotoxic chemotherapy on serum AKR1B10 levels.
Materials and methods
Ethics statement
IRB approval (approval number: 12–152; approval date: 22 February 2012) of the prospective protocol was obtained from the Springfield Committee for Research Involving Human subjects (SCRIHS, Springfield, IL, USA), and written informed consent was obtained from all subjects before any study-related activities. Serum was obtained through two protocols, namely The Tumor Bank of Simmons Cancer Institute at SIU School of Medicine and SCRIHS protocol 11–118, which was specifically created to prospectively collect serum for this AKR1B10 study. We followed the REMARK guidelines for reporting the findings of tumor marker prognostic studies. 17
Human subject enrollment
This study enrolled patients from May 2016 to June 2017. All patients had locally advanced, but non-metastatic, breast cancer, and they were scheduled to receive neoadjuvant chemotherapy followed by surgery. Patients underwent AKR1B10 serum level measurements and tumor imaging before, during, and after chemotherapy. All patient details were de-identified for this manuscript. Eligible patients who consented to enter the study were enrolled consecutively.
Serum sample collection
A standard operating procedure for collecting serum specimens was used. In brief, approximately 10 mL of whole blood was collected in glass collection tubes with no additives using a standard Vacutainer (Becton Dickinson, Franklin Lakes, NJ, USA). The blood was allowed to sit for 15 minutes, after which the tubes were centrifuged at 1890 × g for 15 minutes at room temperature. Serum was immediately transferred into clean polypropylene tubes (100 µL per tube) using an Eppendorf pipette, encoded with a number, and stored at −80°C. Serum samples that were improperly processed were excluded from study use.
Serum AKR1B10 measurement
AKR1B10 protein levels in serum were measured by sandwich enzyme‐linked immunosorbent assay as reported previously. 13
Tumor mass assessment
Tumor dimensions were quantified during chemotherapy by MRI software in the Memorial Medical Center Radiology Department. The Response Evaluation Criteria in Solid Tumours were applied to determine the tumor burden.
Statistical analysis
Descriptive statistics were examined for all variables. Spearman’s rank correlation coefficient was used to assess the relationship between AKR1B10 expression and continuous or ordinal variables or serum AKR1B10 levels. The Wilcoxon rank‐sum test or Kruskal–Wallis test was used to analyze categorical variables. Kaplan–Meier plots were produced to examine the relationship between AKR1B10 expression and patient survival, and the log‐rank test was used for statistical analysis. Results were considered statistically significant at P < 0.05.
Results
Patient characteristics
Table 1 summarizes the characteristics of the patients. The 10 patients ranged in age from 37 to 65 years (mean, 47 years). Nine patients were White, and the remaining patient was Asian. The Eastern Cooperative Oncology Group (ECOG) performance status was 0 for six patients and 1 for one patient, whereas it was not determined for the other three patients. The locations of mammary tumors was evenly divided between the right and left breasts. The tumor grade was grade 2 in one patient and grade 3 in nine patients. Six tumors were ER-positive, whereas five tumors each were progesterone receptor (PR)-positive and HER2-positive. The ER scores of the five ER-positive tumors ranged 5% to 95% (mean, 49.2%). The PR scores for the PR-positive tumors ranged 20% to 99% (mean, 55.25%). An attempt was made to assign molecular subtypes to the tumors based on marker information, and four, three, two, and one tumor was categorized into the luminal B, basal, luminal A, and HER2 subtypes, respectively. All tumors were invasive ductal carcinomas by histology. Nine patients had nodal involvement, and one had distant metastatic spread. The clinical tumor stages were IIIA, IIIB, III, IIB, II (further staging not specified), and IV in four, one, one, two, one, and one tumor, respectively.
Patient and tumor characteristics.

Treatment
Chemotherapy consisted of dose-dense doxorubicin (60 mg/m2)/cyclophosphamide (600 mg/m2) given intravenously every 2 weeks for four cycles followed by 12 weeks of weekly paclitaxel 80 mg/m2 in patients with hormone receptor-positive, HER2-negative disease. Four patients with HER2-positive tumors received four cycles of neoadjuvant pertuzumab, trastuzumab, and docetaxel, whereas the remaining patients received four cycles of doxorubicin and cyclophosphamide followed by three cycles of paclitaxel and nine cycles of docetaxel with concurrent pertuzumab and trastuzumab.
Surgery
The surgical treatment after neoadjuvant chemotherapy varied. Five patients underwent bilateral mastectomies, two patients underwent unilateral mastectomies, and three patients underwent lumpectomies. Post-operatively, patients received radiotherapy and treatment with either an aromatase inhibitor or tamoxifen for a minimum of 5 years.
Tumor measurements
As presented in Table 2, tumors ranged from 2.2 to 11.8 cm in size before treatment. All tumors responded to treatment, although the magnitude of response varied. Five patients achieved complete remission. Tumor size could not be accurately assessed before treatment in one patient, but it measured 5.9 cm after treatment. The remaining four patients experienced tumor reduction ranging 26.3% to 74.6%. The absolute size reductions ranged from 1 to 10.6 cm.
Tumor measurements before, during, and after treatment.

The longest axis is listed for each patient at the various timepoints in the table.
ND, Not determined
AKR1B10 levels
Table 2 outlines AKR1B10 levels before, during, and after treatment. Four patients had baseline AKR1B10 levels lower than 4.5 ng/mL, and they were considered negative for AK1B10 at baseline. Two patients had borderline levels (between 4.5 and 5 ng/mL), and their serial serum measurements displayed no significant change during treatment. The remaining four patients had AKR1B10 levels that declined from baseline after treatment. The extent of the decline ranged from 1.6 to 9.6 ng/mL.
Graphical analysis
Figure 1 illustrates the AKR1B10 levels in our patients before, during, and after treatment in comparison to those in 49 patients reported in our previous study. 18 Based on our prior study, the optimal diagnostic cutoff of serum AKR1B10 was 3.456 ng/mL, which yielded an area of the curve of 0.9426 ± 0.0324 (95% confidence interval [CI] = 0.8790–1.006; P < 0.0001), sensitivity of 90.63% (95% CI = 74.98–98.02), and specificity of 93.88% (95% CI = 83.13–98.72). To increase the sensitivity, we considered AKR1B10 levels exceeding 5 ng/mL as positive. The mean level in our current cohort was 6.35 ng/mL before treatment, and the level subsequently declined to 5.32 ng/mL during treatment and 4.68 ng/mL at the end of treatment. Figure 2 illustrates the changes in AKR1B10 levels for each patient. In five patients, AKR1B10 levels increased, albeit without significance. AKR1B10 levels declined in the remaining patients. Among the five patients whose AKR1B10 levels increased during treatment, three patients had normal levels at the beginning of therapy, whereas the remaining two patients had borderline levels. Only two of the five patients had significantly elevated AKR1B10 levels (>5 ng/mL) during treatment. As noted previously, all evaluable patients, including those with borderline or low AKR1B10 levels, had a positive response to therapy. Figure 3 presents a scatter plot illustrating the relationship between AKR1B10 levels and tumor size. Although no line was obtainable by logistic regression, the data revealed that AKR1B10 levels declined by the end of treatment in all patients irrespective of their pre-treatment levels.
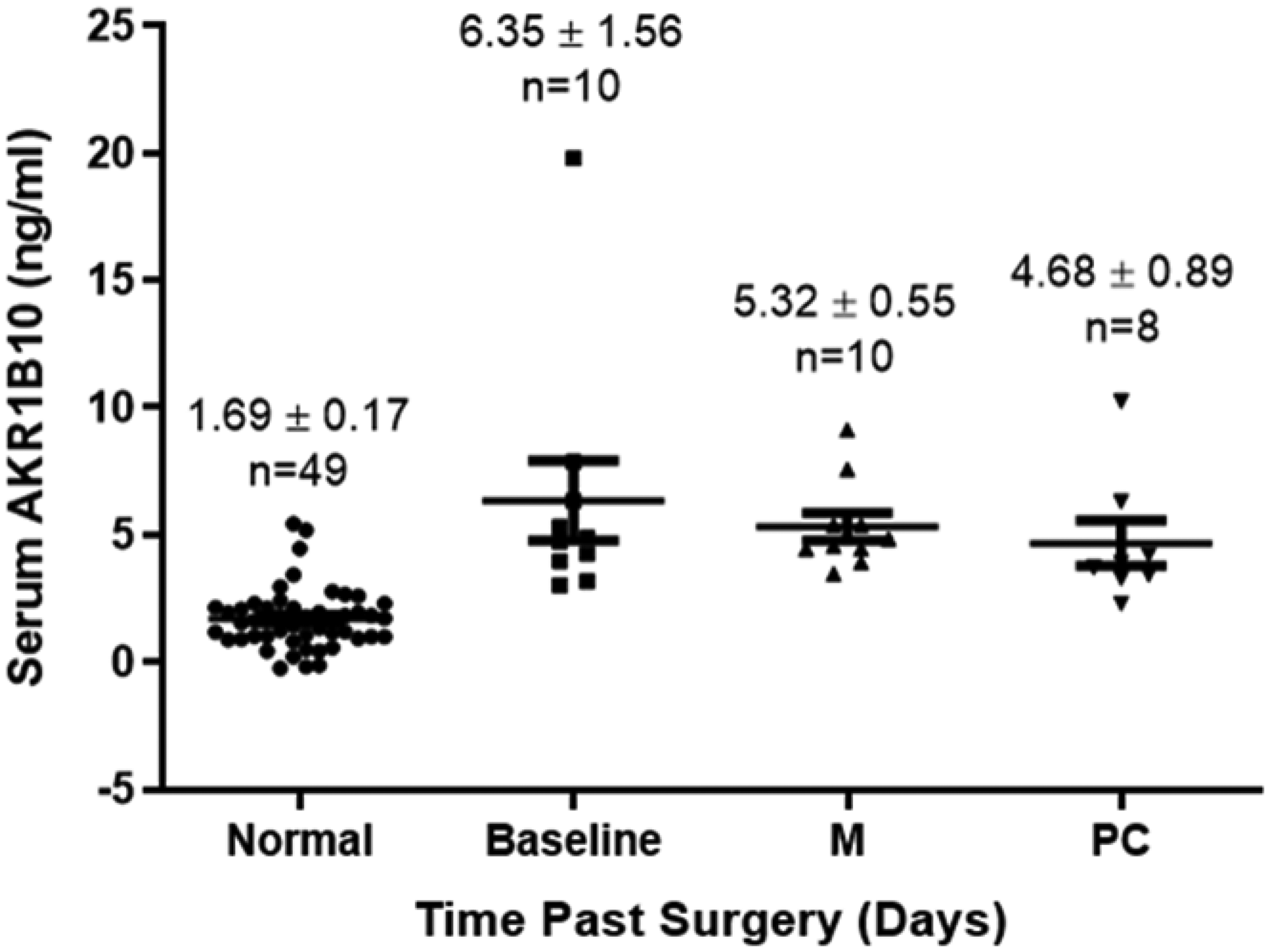
AKR1B10 values at baseline, during treatment, and after treatment in our current group of patients and a previous group of healthy controls (left).

Dynamic changes in AKR1B10 levels during and after treatment in each patient.

Analysis of AKR1B10 changes versus percent changes in tumor size. The percent change in AKR1B10 from baseline is plotted on the x-axis, whereas the percent change in tumor size from baseline is plotted on the y-axis.
Correlations
Baseline AKR1B10 levels and changes in AKR1B10 levels during treatment were not correlated with a variety of parameters, including pre-treatment tumor size, post-treatment tumor size, change in tumor size, percent change in tumor size, age at diagnosis, ECOG performance status, laterality, tumor grade, ER status, PR status, HER2 status/score, ER positivity rate, PR positivity rate, tumor subtype (luminal A, luminal B, basal, HER2), T stage, nodal stage, M stage, and overall stage. Baseline AKR1B10 levels were not correlated with BMI. However, BMI was negatively correlated with changes in AKR1B10 levels (P = 0.021); however, a single outlier patient affected this analysis. Once that patient was removed, no further significant association was noted.
Discussion
Our results revealed a complex set of findings. AKR1B10 levels can temporarily increase insignificantly in patients with normal or borderline levels. Its levels can increase during therapy before declining by the end of therapy. However, AKR1B10 levels declined following treatment in patients whose levels were elevated before treatment. The decline was continuous without a confounding increase during therapy. Chemotherapy has been noted to cause transient increases in tumor marker levels. Kim et al. 19 noted that CEA and CA19-9 levels surged in 18% of patients with gastric carcinoma undergoing treatment. Peak levels were obtained a median of 2.8 weeks following therapy, and the elevation persisted for a median of 9.1 weeks. The median time to the CA19-9 peak was 2.3 weeks, and its levels remained elevated for a median of 7.1 weeks. Interestingly, all patients in the study who experienced this surge were noted to experience clinical benefits from the chemotherapy regimen. The authors concluded that the temporary surge signaled a release of tumor markers from dead malignant cells, heralding a positive response to therapy. Likewise, Yoshida et al. 20 noted that 5.1% of patients treated with proton beam radiotherapy for hepatocellular carcinoma developed an alpha-fetoprotein spike at a median of 1 month.
Our own data suggest two separate conclusions. We observed no spike of AKR1B10 levels in patients whose levels were elevated before treatment, suggesting that the marker was activated by chemotherapy. We may have also missed the increase because serum was only collected during the middle of treatment. However, in patients whose AKR1B10 levels were borderline or low, its levels might spike at the midpoint of therapy, signaling a release of enzymes and heralding a positive response to therapy. This phenomenon might also be ascribed to standard deviations of serum AKR1B10 measurements while the readings were low. As such, the validity of AKR1B10 as a potential tumor marker is reinforced in patients with serum levels greater than 5 ng/mL, and these findings strengthen our previous work. Our data directly address the confounding issue of the effects of cytotoxic chemotherapy on AKR1B10 levels and present original and novel findings. Further study with an expanded cohort of neoadjuvant chemotherapy is warranted to solidify our findings.
Footnotes
Acknowledgement
We gratefully acknowledge the assistance of Kissindra Moore, who helped format the manuscript.
Author contributions
Krishna Rao and Deliang Cao conceived of the study, supervised the work, and wrote the manuscript. Zhe Cao performed the assays. Kathy Robinson recruited patients and served as study coordinator in her capacity as head of the clinical trials office. Kristin Delfino performed statistical analysis.
Data availability
Any reasonable request for de-identified data will be reviewed and such data may be made available.
Declaration of competing interests
The authors have no competing interests to declare.
Funding
This work was funded by the Team Science Grant award from the Simmons Cancer Institute at Southern Illinois University of Medicine.
