Abstract
Background
This study investigated the clinicopathological differences among breast cancer patients with distinct hepatitis B virus serological profiles.
Methods
Data were collected from 5004 female patients newly diagnosed with breast cancer at the First Affiliated Hospital of Chongqing Medical University between 2012 and 2020. A comparative analysis was conducted to determine intergroup differences in clinicopathological characteristics among breast cancer patients with distinct hepatitis B virus serological profiles.
Results
Of the 5004 patients, 8.0% were infected with hepatitis B virus, 55.6% had prior hepatitis B virus infection, and 13.8% were positive only for hepatitis B surface antibody (HBsAb). Hepatitis B virus–infected patients demonstrated a higher incidence of liver enzyme abnormalities (14.1%) and a lower incidence of Ki67 ≥ 20% (54.2%) than HBsAb(+) patients (61.0%). HBsAb(+) patients showed lower progesterone receptor and hormone receptor positivity than hepatitis B virus–negative patients (51.7% vs. 57.2% and 65.5% vs. 70.4%, respectively). Logistic regression analysis indicated that hepatitis B virus–infected patients had a higher incidence of liver dysfunction (odds ratio: 1.535) but a lower incidence of Ki67 ≥ 20% (odds ratio: 0.777). Patients with hepatitis B virus infection had lower hormone receptor (odds ratio: 0.829) and human epidermal growth factor receptor 2 (odds ratio: 0.788) positivity.
Conclusions
The hepatitis B virus serological status influences the clinicopathological features of breast cancer, particularly Ki67, hormone receptor, and human epidermal growth factor receptor 2 expression. Routine hepatitis B virus screening and monitoring are crucial during breast cancer diagnosis and chemotherapy.
Keywords
Introduction
In recent years, breast cancer (BC) has become the most common cancer among women, accounting for 11.6% of new cancer cases worldwide. 1 BC imposes a significant global medical burden. China is undergoing a cancer transition, with an estimated 357,200 new cases of female BC in 2024. The burden of BC continues to rise.2,3 China is also a high-prevalence region for hepatitis B virus (HBV) infection, and the medical burden caused by HBV in the Western Pacific region has not been fundamentally improved.4,5 There may be a link between BC and HBV. The epidemiological trend of BC in China shows a temporal and spatial overlap with the high prevalence of HBV infection. The coexistence of BC and HBV infection is as high as 7.9%, while the rate of BC and previous HBV infection reaches 55.1%. 6 Some studies have suggested that BC tissue serves as an additional reservoir of HBV infection. 7 HBV X-interacting protein (HBXIP) is a cancer protein involved in accelerating cancer progression. It was originally identified as a binding partner of the HBV X (HBX) protein. 8 High HBXIP levels are associated with BC progression, 8 especially in estrogen receptor (ER)–positive BC.9,10 Several studies have shown that HBXIP expression is upregulated in patients with HBV infection and is also associated with HBV replication.11,12 Furthermore, some studies have reported HBx-mediated upregulation of HER2 in HBV-related liver cancer, but it remains unknown whether it also has an effect on BC.13,14 Therefore, the relationship between BC and HBV warrants further study.
BC is a hormone-dependent, heterogeneous malignant tumor with diverse histological manifestations, molecular characteristics, and biological behaviors. Given its diverse pathological features, BC should be classified according to its clinical and pathological characteristics. Individualized treatment strategies based on such classification are essential for improving BC prognosis.15,16 However, only a few studies have evaluated the association between HBV infection status and clinicopathological features of BC. In this study, we conducted a retrospective case–control study to determine the clinicopathological characteristics and intergroup differences among women with newly diagnosed BC who exhibited distinct HBV serological profiles.
Materials and methods
Study population
Data from 5004 BC patients were obtained from the China Database for Concomitant Disease of Breast Cancer (2012–2020) in the Breast Cancer Center of the First Affiliated Hospital of Chongqing Medical University. Exclusion criteria included male patients; non-breast primary malignant tumors; incomplete hormone receptor (HR) and Ki67 results; unclear pathological diagnosis; Paget’s disease or special BC types without HR results; and incomplete serological indicators of HBV (Figure 1).

Study design flowchart, exclusion, and grouping.
Data collection
Experienced health workers recorded basic information of the patients, such as age and anthropometric parameters including height and weight, in accordance with standard procedures. Each BC patient enrolled in the study was diagnosed pathologically and immunohistochemically by the Pathology Center of Chongqing Medical University. Patients diagnosed elsewhere were required to have their pathological slides confirmed. Clinicopathological features included age at diagnosis, primary tumor size, degree of lymph node involvement, ER status, progesterone receptor (PR) status, human epidermal growth factor receptor 2 (Her-2) status, p53 mutation status, and Ki67 percentage. These data were obtained from electronic medical records and original pathology reports. HR positivity was defined as ER positivity, PR positivity, or positivity for both based on immunohistochemical staining.
In China, routine serological tests for pathogens are performed during hospitalization for all individuals undergoing surgical treatment. HBV serological marker tests included the detection of hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (HBsAb) using time-resolved fluoroimmunoassay as well as the detection of hepatitis B e antigen (HBeAg), hepatitis B e antibody (HBeAb), and hepatitis B core antibody (HBcAb) using enzyme-linked immunosorbent assay. All tests were conducted by the Laboratory Department of the First Affiliated Hospital of Chongqing Medical University, according to the manufacturer’s instructions and quality control procedures. All data were stored in the computerized medical record system of the Breast Cancer Center of Chongqing. HBV infection status was determined using a combination of HBsAg, HBsAb, HBeAg, HBeAb, and HBcAb indices. Patients positive for HBsAg were included in the HBV infection group, while those positive for HBcAb but negative for HBsAg were classified as previously HBV-infected. Patients positive only for HBsAb were considered immune to HBV. Patients negative for all HBV serological markers were considered to have no history of HBV infection. The study aimed to investigate intergroup differences in clinicopathological features among BC patients with different HBV serological groups.
Statistical methods
Microsoft Excel 2016 and IBM SPSS Statistics 25 were used for data recording and analysis. Continuous variables were described as medians (interquartile ranges) or means ± standard deviations. The Shapiro–Wilk or Kolmogorov–Smirnov methods were used for normal detection. Non-normally distributed continuous variables were analyzed using nonparametric tests. Categorical variables were described as numbers (proportions) and analyzed via the chi-square test with Bonferroni correction. Logistic regression was used to analyze the associations between HBV serological types and BC clinicopathological characteristics. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated, and a two-tailed p-value <0.05 was considered to indicate statistical significance.
Results
Among the 5004 female BC patients at initial diagnosis included in the study, the mean age was 50.8 ± 10.9 years. The median age of the HBV infection group (N = 400, 8.0%) was 49.0 (43.0–55.8) years, while that of the previously infected HBV group (N = 2781, 55.6%) was 51.0 (45.0–60.0) years. The median age of individuals only positive for HBsAb (N = 692, 13.8%) was 50.0 (44.0–58.0) years, and that of individuals negative for HBV serological markers (N = 1131, 22.6%) was 49.0 (43.0–56.0) years. The Kolmogorov–Smirnov test showed that the p value was less than 0.05, and the variables did not follow a normal distribution. Statistically significant differences in age were observed among these groups. The median body mass index (BMI) of the HBV infection group was 23.125 (21.5–25.1) kg/m2, while that of the previously infected HBV group was 23.296 (21.6–25.1) kg/m2. The median BMI of individuals only positive for HBsAb was 23.125 (21.2–24.8) kg/m2, and that of individuals negative for HBV serological markers was 23.233 (21.5–25.1) kg/m2. The Kolmogorov–Smirnov test showed that the p value was less than 0.05, and the variables did not follow a normal distribution. There was no significant difference in BMI among the groups (Table 1).
Age and BMI of breast cancer patients with different types of HBV serological markers.

BMI: body mass index; HBV: hepatitis B virus; HBsAg: hepatitis B surface antigen; HBcAb: hepatitis B core antibody; HBsAb: hepatitis B surface antibody.
p < 0.05.
In the single-factor analysis of intergroup differences, the incidence of elevated liver enzymes in the HBV infection group was 14.1%, which was significantly higher than those in the other three groups (9.3%, 8.6%, and 9.3%, respectively). In the HBV infection group, the incidence of Ki67 ≥ 20% was 54.2%, which was significantly lower than that in the only HBsAb(+) group (61%; p < 0.05). Within the only HBsAb(+) group, the positivity rates of PR and HR were 51.7% and 65.5%, which were significantly lower than those in the HBV serological marker negative group (57.2% and 70.4%), respectively (p < 0.05). No significant differences were observed in lymph node metastasis, tumor size, ER status, HER2 expression, or p53 mutation across all groups. After applying the Bonferroni method, the incidence of elevated liver enzymes in the HBV infection group was statistically different from that in the other three groups (Table 2).
The baseline characteristics of breast cancer patients with different types of HBV serological markers.

ER: estrogen receptor; HBV: hepatitis B virus; Her-2: human epidermal growth factor receptor 2; HR: hormone receptor; LN: lymph node; PR: progesterone receptor; HBsAg: hepatitis B surface antigen; HBcAb: hepatitis B core antibody; HBsAb: hepatitis B surface antibody.
Indicated statistical difference compared with negativity for all group (p < 0.05).
Indicated statistical difference compared with the only HBsAb(+) group (p < 0.05).
Indicated statistical difference compared with the HBcAb(+) and HBsAg(−) group (p < 0.05).
In the multivariate analysis, using the HBV serological marker negative group as the reference and adjusting for age and BMI, we observed statistically significant clinicopathological characteristics in the HBV infection group. These included Ki67 ≥ 20% (OR: 0.777 (0.599–0.983)) and impaired liver function (OR: 1.535 (1.041–2.264)). In the group with a history of HBV infection (HBcAb (+), HBsAg (−)), statistically significant clinicopathological features included HR positivity (OR: 0.829 (0.697–0.986)) and Her-2 positivity (OR: 0.788 (0.646–0.961)). In addition, in the only HBsAb(+) group, HR positivity (OR: 0.750 (0.596–0.944)) and Her-2 positivity (OR: 0.725 (0.553–0.949)) were statistically significant. No other indicators showed significant differences. The likelihood ratio test suggested that the significance of model fitting information was 0.000 (p < 0.05), and the model could fit the data adequately. The goodness-of-fit significance of the model was 0.517, and the model fit was reasonable (Table 3).
The ORs and 95% CIs in BC clinicopathological features of different hepatitis B virus serological marker groups after adjustment for age and BMI.
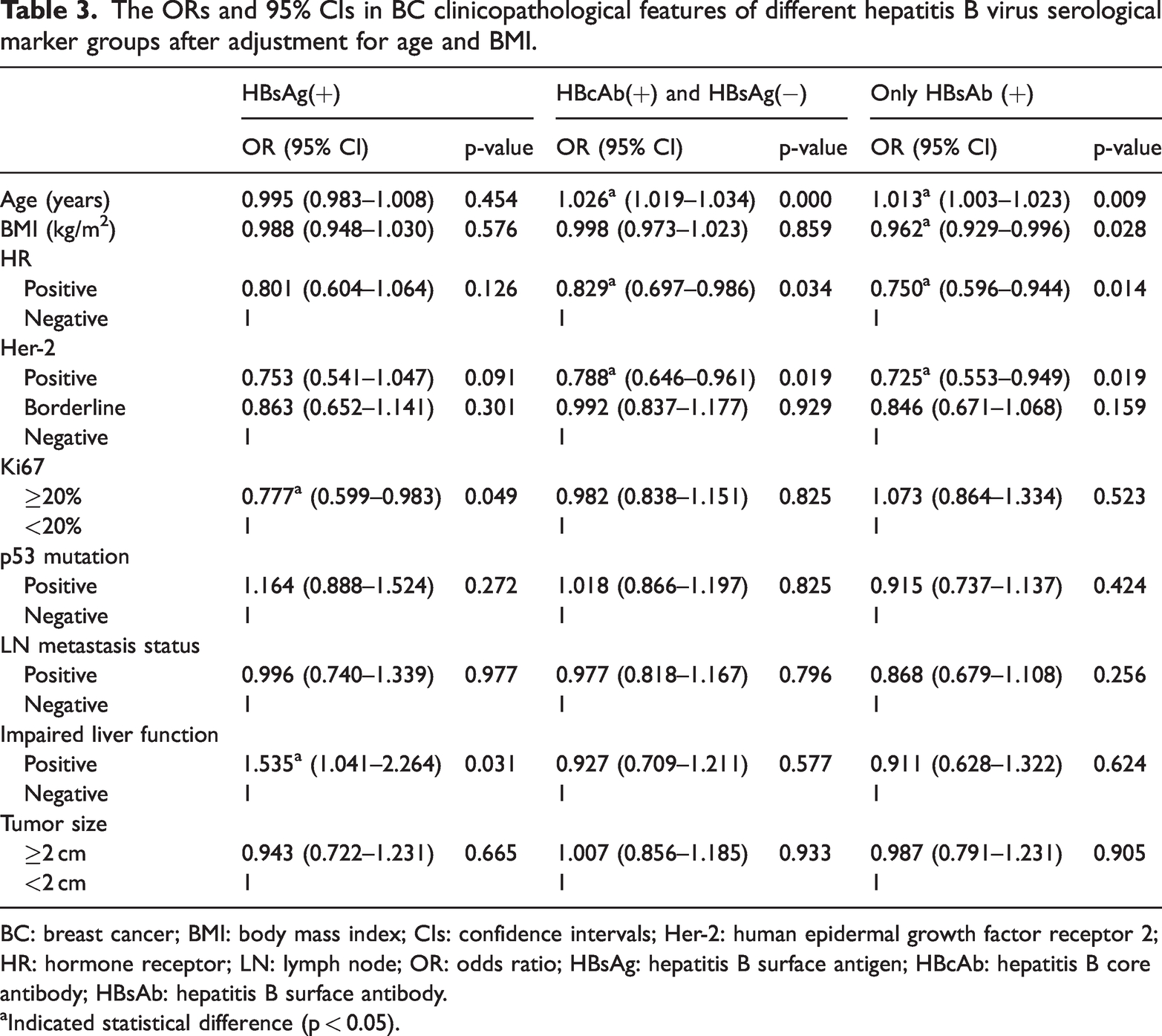
BC: breast cancer; BMI: body mass index; CIs: confidence intervals; Her-2: human epidermal growth factor receptor 2; HR: hormone receptor; LN: lymph node; OR: odds ratio; HBsAg: hepatitis B surface antigen; HBcAb: hepatitis B core antibody; HBsAb: hepatitis B surface antibody.
Indicated statistical difference (p < 0.05).
Discussion
Some viral infections are believed to be associated with the development of BC, but previous studies have primarily focused on human papillomavirus, cytomegalovirus, Epstein–Barr virus, and hepatitis C virus (HCV).17–20 HBV is highly prevalent in the Western Pacific region. Previous studies have shown that the rates of HBV infections and past HBV infections are 5.0% and 17.6%, respectively, in the routine physical examination population—both lower than those observed in BC patients. 6 The pathogenic mechanisms of HBV are similar to those of HCV; hence, it is important to investigate the association between HBV and BC. HBV is widely recognized as a hepatophilic virus and is closely related to the occurrence of primary hepatocellular carcinoma. Some studies have explored the association between HBV and extrahepatic cancers.21,22 Based on the clinical and pathological features of BC, developing treatment plans has become the cornerstone of BC treatment. This study examined the clinical and pathological characteristics of BC patients with distinct HBV serological features.
The diagnosis of HBV has long relied on serological markers, with each marker carrying distinct clinical significance. 23 We first divided patients with BC into four groups based on their HBV serological markers. The HBsAg(+) group was considered as currently infected with HBV, while the HBcAb(+) and HBsAg(−) group was considered to have a history of HBV infection. The HBsAb(+) group was considered immune to HBV. Patients negative for all HBV serological markers were considered to have no history of HBV infection. HbsAg is a surface protein on the outermost layer of the HBV capsid. Although noninfectious, HbsAg stimulates antibody production and serves as a marker of HBV infection. This study included 400 HBsAg(+) BC patients, representing 8.0% of the total study population. Elevated liver enzymes reflect the degree of liver function damage, whose severity is influenced by the HBV genotype and viral components. In patients with HBV infection, virus-induced liver damage disrupts various cellular processes, including signaling, apoptosis, transcription, and DNA repair. 24 In this study, liver function impairment in HBsAg(+) patients was significantly higher than that in other groups. It is therefore necessary to actively detect liver function indicators and use liver-protecting drugs to support treatment for HBsAg(+) BC patients. Ki67 is widely used as an immunohistochemical nuclear localization marker in surgical pathology. Nuclear immunoreactivity for Ki67 indicates active cell cycling, and the proportion of Ki67-positive tumor cells provides an estimate of the tumor growth fraction. In BC, Ki67 helps determine the proliferation rate of tumor cells and prognosis.25,26 In this study, consistent with common clinical practice, a cutoff value of 20% was used for Ki67 expression. 27 After correcting for age and BMI, the incidence of Ki67 ≥ 20% in the HBsAg (+) group was lower than that in the HBV serological marker negative group (OR: 0.777 (0.599–0.983)). After HBV infection, the immune system may enter a chronic stress state, altering immune cell function and cytokine secretion. 28 This includes imbalances in T lymphocyte subsets and Th1/Th2 cells,29,30 which reduce antitumor immune function and create conditions favorable for the occurrence and development of BC. This may be attributable to the fact that the BC microenvironment is in a state of chronic HBV-related stress, which is more likely to induce BC with low proliferation rates.28–31 This may also be due to other pathological features that determine the malignancy of BC jointly.
HBV is a partially double-stranded circular DNA virus that infects humans and is classified as a subtype of retrovirus due to its similarity to reverse transcriptase. After entering the human body, if HBV is not recognized and eliminated by the immune system, its nucleic acid enters the cell nucleus and integrates into the human genome, forming a covalently closed circular DNA (cccDNA) 32 that persists for a long term in hepatocyte nuclei. Conventional nucleic acid–based antivirals act outside the cell nucleus and cannot interfere with cccDNA, making chronic HBV extremely difficult to eradicate. HBcAb is considered a marker of HBV infection and is commonly used in Europe and the United States to assess the status of HBV infection. 33 In this study, 2781 HBcAb(+) or HBsAg(−) individuals with a history of HBV infection were included, representing 55.6% of the total BC study population. Although past HBV infection in these individuals did not result in active liver disease, the integration of HBV DNA is widespread. 34 Approximately two-thirds of invasive BC patients have ER(+) and more than half of patients are PR(+). Hormones bind to their HR complexes, which mediate regulatory functions. ER and PR play key roles in the development and progression of BC. 35 If ER or PR is positive, HR is considered positive. Clinically, BC patients often need endocrine therapy based on whether HR is positive. To avoid the interference of ER and PR on HR in the multivariate logistic regression model, only HR was included. After adjusting for age and BMI, BC patients with past HBV infection had a lower risk of HR positivity than the HBV serological marker negative group (OR: 0.829 (0.697–0.986)). Estrogen is normally inactivated in the liver. Liver function is impaired by HBV infection, and hormone inactivation is weakened. 36 Prolonged estrogen exposure may contribute to reduced HR sensitivity. Similar findings were observed in the HBsAb-positive group. HBsAb is considered a protective antibody that binds specifically to HBsAg, clearing HBV and preventing reinfection. 37 Since 1992, China has included HBV vaccination in its national immunization program, including newborn immunization program. With improved public health awareness, some individuals—particularly those with better health awareness and financial resources—receive the HBV vaccineregularly, monitor the serological indicators of HBV, and obtain booster doses when necessary. 38 In this study, 692 patients with only HBsAb (+) were included, representing 13.8% of the total BC study population. After adjusting for age and BMI, the HBsAb (+) group had a lower risk of HR positivity (OR: 0.750 (0.596–0.944)) than the HBV serological marker negative group. Individuals with prior HBV infection or vaccination may be more vulnerable to reduced endocrine therapy if they develop BC.
The tumorigenic mechanism of the HER2 oncogene involves inhibiting apoptosis and promoting proliferation. It also increases tumor cell invasiveness and promotes tumor angiogenesis and lymphangiogenesis. In this study, using a multiple regression model, we found that compared with the all-negative group of HBV serological markers. The risk of Her-2 positive was lower in patients with a history of HBV infection group (OR: 0.788 (0.646–0.961)) and only the HBsAb(+) group (OR: 0.725 (0.553–0.949)). Most cancers undergo genetic rearrangements during tumor evolution, ultimately driving cancer progression. The mechanism of HBV in the development of primary liver cell carcinoma has made substantial progress, with some HBV-mediated genetic rearrangements occurring up to 20 years before the diagnosis.39,40 Some studies suggest that BC tissue may serve as another reservoir of HBV infection. 7 BC associated with HBV infection may be influenced by HBV and its gene integration over extended periods. Some studies indicate that the HBV vaccine may help reduce cancer risk.41,42 Evidence suggests that in animal BC models, combining HBsAg with other cancer antigens resulted in enhanced antitumor activity. 43 Previous HBV infection status is even a favorable prognostic factor for intrahepatic cholangiocarcinoma. However, further epidemiological and animal studies are required to determine whether the HBV vaccine provides additional immune benefits beyond reducing liver cancer risk.41,44 Her-2 gene expression in BC patients with a history of HBV infection or HBsAb(+)-only group may be affected by HBV and its inactivated vaccine. Chemotherapy and targeted drug therapy are key treatments for malignant breast tumors. It is worth mentioning that chronic HBV infection patients may experience HBV reactivation (HBVr) during chemotherapy, targeted therapy, or immunosuppressive therapy, which may lead to severe liver failure. Some studies report that the risk of HBcAb(+) patients experiencing HBVr during chemotherapy is as high as 14% in hematological system tumors,41,45 and BC patients with a history of HBV infection also face this risk. BC patients previously infected with HBV should receive strengthened health management. Although this is a cross-sectional case–control study, serological markers of past HBV infection are time-dependent and reflect disease characteristics at a specific point in time, potentially making them more informative than other cross-sectional studies.
HBXIP is an oncogenic protein that accelerates cancer progression.8,10 It is an 18-KDa conserved protein originally identified through its interaction with HBX, which reduces HBVr along with HBsAg and HBeAg synthesis. This effect is believed to be mediated by inhibition of HBX activity on endogenous viral core promoter/enhancer elements. 46 HBx can also interact with a variety of cellular signaling molecules, activating PI3K/Akt, MAPK, and other signaling pathways. The PI3K/Akt signaling pathway regulates cell proliferation, survival, and metabolism; its activation promotes cell proliferation and survival while inhibiting apoptosis. 7 The MAPK signaling pathway is involved in cell growth, differentiation, and stress response, and its abnormal activation can result in excessive proliferation and tumorigenesis. 47 Dysregulation of these signaling pathways may contribute to the initiation and progression of BC.
This was a cross-sectional study, and long-term survival data were not available for each group. As a central tertiary medical institution, most of our BC patients were from urban areas, and their income levels and lifestyle habits may differ from those of the broader Chongqing region and the general population. The data in this study are observational; therefore, a direct causal relationship between HBV infection and BC pathological characteristics cannot be established. While collecting Her-2 data, we could not obtain the results of Her-2 gene amplification determined by the fluorescence in situ hybridization method. We therefore relied on the negative, borderline, and positive classification criteria. At the same time, most indicators were derived directly from clinical data, with a relative lack of supporting basic experiments. These represent important limitations of this study. If feasible, we intend to conduct multicenter studies and animal model experiments in the future to further enhance the validity of our study findings.
Conclusions
Among women newly diagnosed with BC, the incidence of HBV infection and past HBV infection was higher, and different HBV serological profiles were associated with distinct clinicopathological characteristics, particularly HR, Ki67, and Her-2 indices. Women with HBV infection were more likely to have BC with relatively low proliferation. Women with a history of HBV infection or immunity to HBV may be more likely to develop HR(−) and Her-2 (−) BC. The pathological indicators of BC associated with different HBV serological markers have further research value.
Supplemental Material
sj-xlsx-1-imr-10.1177_03000605251384085 - Supplemental material for Clinicopathological characteristics of initially diagnosed breast cancer patients with different serologic types of hepatitis B virus
Supplemental material, sj-xlsx-1-imr-10.1177_03000605251384085 for Clinicopathological characteristics of initially diagnosed breast cancer patients with different serologic types of hepatitis B virus by Zhao-Xing Li, Zhi-Yu Jiang, Lei Hu, Li-Yuan Mu, Xiao-Chun Cheng, Xiu-Quan Qu, Bai-Qing Peng, Jiang-Yu Tu, Pu Qiu, Yu-Xuan Huang, Dong-Li Liu, Kai-Nan Wu and Ling-Quan Kong in Journal of International Medical Research
Footnotes
Author contributions
All authors made significant contributions to the research. Zhao-Xing Li is mainly responsible for the conception, drafting, writing and revision. Zhi-Yu Jiang is responsible for conceptualization, research, and design. Lei Hu is responsible for data collection. Li-Yuan Mu and Xiao-Chun Cheng were responsible for data analysis and interpretation, while Xiu-Quan Qu, Bai-Qing Peng, and Jiang-Yu Tu were responsible for data verification. Pu Qiu, Yu-Xuan Huang, and Dong-Li Liu were responsible for the study’s ethical issues and literature collection. Kai-Nan Wu and Ling-Quan Kong supervised the revision and review of the paper. Additionally, all authors have agreed on the journal to which the article has been submitted and agree to be accountable for all aspects of the work.
Data availability statement
All raw data were stored in the computer medical record system of Chongqing Breast Cancer Center and the Health Management Center of the First Affiliated Hospital of Chongqing Medical University. A backup of the data for this study is available in the supplementary information file, which can be accessed by contacting the corresponding author upon reasonable request to protect the privacy of study participants.
Declaration of conflicting interests
All authors declare that there is no actual or potential competing interests.
Ethics approval and informed consent
This study was approved by the Ethics Review Committee of the First Affiliated Hospital of Chongqing Medical University and was conducted in accordance with the principles of the Declaration of Helsinki (Approval No. k2024-227-01). This is a retrospective study that utilized anonymized/de-identified data. According to local/national guidelines, written informed consent of the participants is not required. The need for informed consent was waived by IRB.
Funding
All authors declared that this study received no funding.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
