Abstract
Objectives
To observe changes in actin and dendritic spines in the hippocampus after propofol anesthesia, and to evaluate the role of these changes in subsequent learning impairment in both young (3-month-old) and aged (20-month-old) male rats.
Methods
The shuttle box test was used to evaluate learning from 1, 3, 7, or 14 days after anesthesia. Both F-actin content and dendritic spines in the hippocampal CA1 region were observed using immunofluorescent staining and western blot assays.
Results
The latency of the escape response was significantly prolonged until 7 days after anesthesia in the aged rats, and their learning curves were shifted. Dendritic spines were also decreased in the aged rats within 7 days after anesthesia, and F-actin content was significantly increased until 14 days after anesthesia.
Conclusions
Learning was inhibited after propofol anesthesia, especially in aged rats. The over-polymerization of actin and subsequent reorganization of dendritic spines in the hippocampus may be responsible for this learning impairment. Our findings suggest that synaptic plasticity may be an underlying mechanism of perioperative neurocognitive disorders.
Keywords
Introduction
Although perioperative neurocognitive disorders (PND) have been discussed for over a century, the underlying mechanisms remain unclear. Many factors may contribute to the occurrence of PND by inducing changes in synaptic plasticity, with subsequent learning and memory dysfunction. 1
For synaptic plasticity, two features are usually considered: synaptic structure and function. The morphology of dendritic spines in the postsynaptic membrane both reflects and takes part in synaptic plasticity, and is mainly regulated by dynamic transformations between G-actin and F-actin. 2 Functional plasticity occurs via long-term potentiation (LTP) and long-term depression (LTD), which are both fine-tuned by multiple molecules. Furthermore, interactions occur between the structural and functional aspects. For example, more actins are polymerized during LTP, 2 whereas G-actin and smaller spines are related to LTD. 3
Anesthetics can affect synaptic plasticity. For example, propofol has been reported to inhibit LTP 4 and modulate actin dynamics. 5 To date, however, there have been no reports regarding the status of actin dynamics after anesthesia, although this knowledge may improve our understanding of PND.
During our early investigations into the mechanisms underlying PND, we noted the differential expression of some actin-binding proteins, such as cofilin 1 (CFL1) and coronin-1a (CORO1A), in the rat hippocampus after propofol anesthesia, implying changes in actin dynamics. 6 Further electrophysiological and morphological observations confirmed our speculations; F-actin levels were increased both before and during LTP induction in the hippocampus of aged rats 14 days after propofol anesthesia, whereas LTP amplitude was decreased. 7
We therefore hypothesized that over-polymerized actin might contribute to PND by disrupting synaptic plasticity. In the present study, enhanced actin polymerization and reorganization of dendritic spines were observed; both were related to PND in aged rats after propofol anesthesia.
Methods
Animals
Male Sprague Dawley rats aged 3 months (young, n = 60) or 20 months (aged, n = 60) were purchased from Chengdu Dashuo Biotechnology Co., Ltd (Chengdu, China). All animal procedures were performed under standard conditions in Beijing Chao-yang Hospital of Capital Medical University, and all experiments conformed to the animal care protocols of the institution. Experimental protocols were approved by the review board of Beijing Chao-yang Hospital of Capital Medical University (approval number AEEI-2020-117). Young and aged rats were each divided using a table of random numbers into the following groups: control, training, anesthesia, and training after anesthesia. Rats in the control group received no treatment. Rats in the training group were trained through shuttle box. In the anesthesia group, rats received anesthesia and were further divided into four groups based on the time after anesthesia (1, 3, 7, and 14 days). Rats in the training after anesthesia group received shuttle box training after anesthesia and were further divided into four groups based on the time after anesthesia at which training was started (1, 3, 7, or 14 days). Rat euthanasia was performed by decapitation.
Anesthesia
All rats were housed individually at a constant room temperature (20°C–22°C) and humidity (50%–60%) with a 12-hour light/dark cycle. They were freely fed with a standard diet and tap water. To induce anesthesia, propofol (100 mg/kg) was injected intraperitoneally. Anesthesia status was maintained for 3 hours with propofol (50 mg/kg/hour through the caudal vein). In the control group, rats underwent the same procedure but the propofol was replaced by an equivalent volume of saline.
Rats who underwent propofol anesthesia with no response to the tail pinch test were chosen for the following experiments. The rats were placed in a temperature-controlled incubator (28°C) until the end of anesthesia. The arterial blood pressure and blood gases of propofol-treated rats were measured and maintained within normal ranges during anesthesia. All rats were then housed before further experiments were performed.
Behavioral experiments
The shuttle box test was used to assess learning and memory. Rats were placed in the electric shock area of the test box. Stimulations of lights and/or a buzzing noise were given for 5 s, and electrical stimulation (30 V, 50 Hz) was then given for another 5 s. An active avoidance reaction in rats was defined as fleeing to the safe area within 5 s of the light or buzz stimulations, whereas a passive avoidance reaction was defined as when this reaction was induced only by the electric shock. A failure to react was defined if neither active nor passive avoidance reactions occurred within the stimulation period. Generally, active avoidance reactions developed after several rounds of training. The latency of the escape response (LER) was recorded as the time from the start of the electrical stimulus until the rat reached the safe area.
Training for the rats started at 1, 3, 7, or 14 days after anesthesia, between 8 a.m. and 12 p.m., and continued for 7 days. Protocols were controlled using a computer. The LERs were recorded and compared among groups on day 7 of the training protocol.
Immunofluorescent staining
After training and testing, rats were sacrificed and their brains were removed, fixed in 4% paraformaldehyde, and dehydrated in sucrose solution (20%–30%). The tissue was embedded in optimal cutting temperature compound and cut into 10-μm sections. The sections underwent antigen retrieval with citric acid retrieval solution and heating for 10 minutes before being blocked with 1% bovine serum albumin (Sangon Biotech, Shanghai, China). Thereafter, the sections were incubated with primary antibodies against postsynaptic density protein (PSD95; 1:100; Affinity, Changzhou, China; AF7839), microtubule-associated protein 2 (MAP2; 1:200; Affinity; AF4081), and F-actin (1:25; Abcam, Cambridge, UK; ab205) at 4°C overnight, followed by incubation with Cy3- or fluorescein isothiocyanate (FITC)-conjugated secondary antibodies (1:200; Invitrogen, Carlsbad, CA, USA or Abcam) for 60 minutes at room temperature. After being counterstained with 4′,6-diamidino2-phenylindole (DAPI; Aladdin, Shanghai, China), the sections were observed and images of the CA1 region of the hippocampus were captured using a fluorescent microscope (Olympus Optical Co., Tokyo, Japan).
Western blot
Proteins were extracted from the hippocampal CA1 region using radioimmunoprecipitation assay lysis buffer (Beyotime, Shanghai, China). After determining the protein concentration using a bicinchoninic acid protein assay kit (Beyotime, Shanghai, China), proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA). The membranes were then blocked with 5% skim milk and incubated with primary antibodies against PSD95 (1:500; Wanleibio, Shenyang, China; WL05046), MAP2 (1:1000; Wanleibio; WL04217), F-actin (1:1000; Abcam; ab205), and glyceraldehyde 3-phosphate dehydrogenase (1:1000; Wanleibio; WL01114) at 4°C overnight. The membranes were then washed with Tris-buffered saline with Tween and incubated with horseradish peroxidase-conjugated secondary antibodies (Beyotime, Shanghai, China) at 37°C for 45 minutes. Sensitive X-ray film was used to visualize the target bands.
Statistical analysis
Protein fluorescent densities were evaluated by one-way analysis of variance followed by the Bonferroni post hoc test using SPSS Statistics for Windows, Version 17.0 (SPSS Inc., Chicago, IL, USA). A P-value of less than 0.05 was considered significant.
Results
Among the groups, there were no significant differences in rat weights or the numbers of rats in each group who completed all experiments (young rat groups: 12/12 in all groups; aged rat groups: 11/12 in rats who started shuttle box training 1 and 14 days after anesthesia, 10/12 in rats who started shuttle box training 3 days after anesthesia, and 12/12 in all other groups).
Behavioral experiments
Our data indicated that learning was impaired in aged rats. Furthermore, the LERs of aged rats were prolonged until 7 days after anesthesia and were back to normal levels within 14 days (P < 0.05) (Figure 1). For young rats, similar changes were detected within 3 days after anesthesia only. Moreover, the LERs of aged rats were significantly longer than those of young rats at every time point (Figure 2).
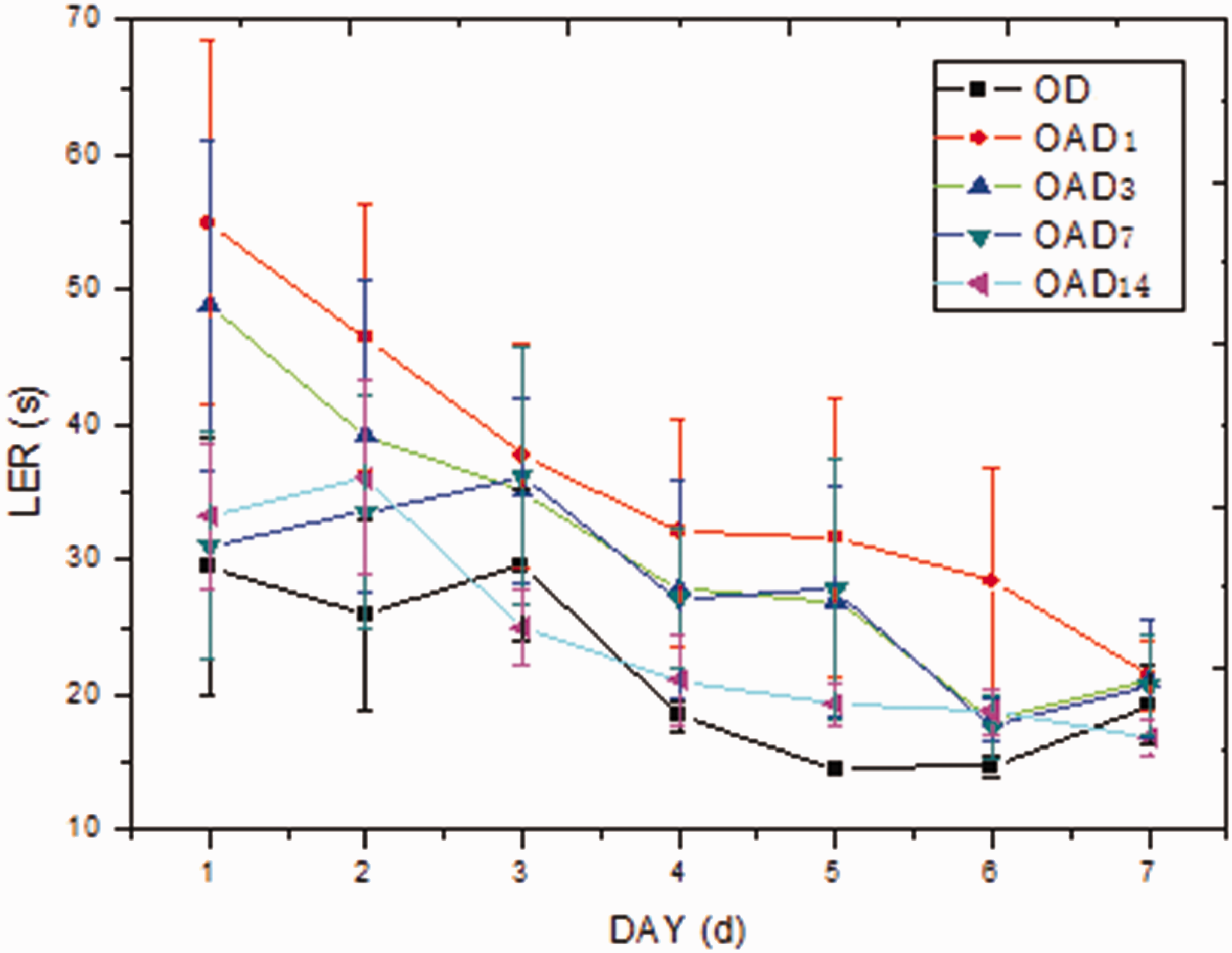
Learning curves of aged rats. Compared with that of OD, the curves of OAD1, OAD3, and OAD7 were shifted upward and LERs were prolonged (P < 0.05).

Comparison of LERs between the young and aged rats. The LERs of aged rats were longer than those of the young rats at every time point (*P < 0.05).
Dendritic spines in the CA1 area of the hippocampus
Immunofluorescent staining
After shuttle box training, more spines were observed in CA1 hippocampal neurons in all rats. For the young rats, fewer spines and a depressed increase in spines after training were seen within 3 days after anesthesia compared with controls. In the aged rat groups, however, both of these changes were noted until 7 days after anesthesia (Figure 3a).

PSD95 expression in aged rats. (a) Immunofluorescence showing dendritic spines labeled with PSD95 (red, arrows) in the CA1 region of the hippocampus of aged rats. DAPI is shown in blue. Scale bar = 50 μm. (b) Western blot of PSD95 expression (*P < 0.05).
Western blot
Western blot results showed similar expression changes of PSD95 to those from immunofluorescent staining (Figure 3b).
F-actin in the CA1 area of the hippocampus
Immunofluorescent staining
Before anesthesia, more F-actin was observed in young rats than in aged rats. Without anesthesia, F-actin content increased slightly after shuttle box training in all rats. However, F-actin content was significantly upregulated after anesthesia; this upregulation continued until day 7 after anesthesia in the young rats (P < 0.05, Table 1). In the aged rats, significantly increased F-actin content was observed until day 14 after anesthesia (P < 0.05, Table 1 and Figure 4a).
F-actin densities in the rat hippocampus.
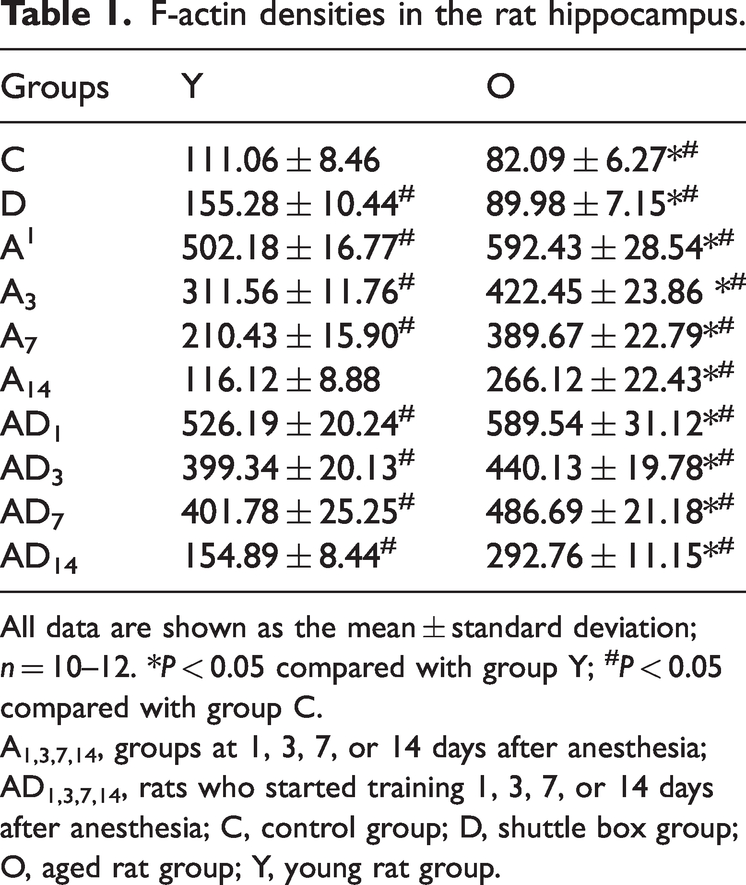
All data are shown as the mean ± standard deviation; n = 10–12. *P < 0.05 compared with group Y; #P < 0.05 compared with group C.
A1,3,7,14, groups at 1, 3, 7, or 14 days after anesthesia; AD1,3,7,14, rats who started training 1, 3, 7, or 14 days after anesthesia; C, control group; D, shuttle box group; O, aged rat group; Y, young rat group.

F-actin and MAP2 expression in aged rats. (a) Immunofluorescence showing the distributions of F-actin (red, arrows) and MAP2 (green) in the CA1 region of the hippocampus of aged rats. DAPI is shown in blue. Scale bar = 50 μm. (b) Western blot of F-actin expression (*P < 0.05).
Western blot
Western blot results showed similar expression changes of F-actin to those from immunofluorescent staining (Figure 4b).
Discussion
Advanced age is an accepted cause of PND, 8 which was further confirmed by the present study. All rats developed active avoidance reactions after training; however, learning was impaired in aged rats, which presented as upward-shifted learning curves. Moreover, the reactions of aged rats appeared to be declined because their LERs were longer than those of young rats at every time point. A delayed recovery of reaction was also observed in aged rats until 14 days after anesthesia, whereas those of the young rats returned to normal within 7 days. Together, these findings suggest that old age increases susceptibility to PND.
One difference between young and aged synapses is the morphology of dendritic spines. Spines in the aged brain are usually shorter in length and decreased in density compared with those in young brains; this is believed to contribute to cognitive impairment. 9 In the present study, there were differences in spines between young and aged rats both before and after anesthesia, which might explain the impaired learning and memory capability of aged rats to some degree. This finding also suggests an important role of dendritic spine morphological changes in PND.
Decreased, shortened, and contracted dendritic spines in the hippocampus after anesthesia indicates inhibited synaptic plasticity. It is generally accepted that LTP and LTD are associated with the long-term enlargement and shrinkage, respectively, of dendritic spines. 10 Moreover, biochemical states inside spines can be dynamically modulated by synaptic activity, leading to the reorganization of cytoskeletal architecture, and thus the structural and functional plasticity of the spine. 11 In some studies, propofol was reported to decrease dendritic spine density and restrict spine development, which was suggested to be involved in cognitive dysfunction during and after anesthesia. 12 The changes in dendritic spines in the present study may also be responsible for the impairments in learning and memory that were observed in the behavioral experiments. We therefore postulate that modified spine shapes is a possible morphological feature of PND in the hippocampus.
The actin cytoskeleton is the principal architectural component of the dendritic spine; its dynamic remodeling, or the balance between F-actin and G-actin, plays a major role in the structural plasticity of dendritic spines. 2 The appropriate polymerization of actin is the base of LTP, 2 and over-polymerized actin is associated with neural degeneration and cognitive dysfunction. 13 Furthermore, previous studies have reported the propofol-induced reorganization of actin. 5 In an earlier investigation, we identified increased F-actin levels in the aged rat hippocampus after propofol anesthesia; this finding was closely related to LTP depression, indicating the important role of actin over-polymerization in synaptic dysfunction after propofol anesthesia. 7
Many molecules regulate actin dynamics, and some of these molecules may be affected by propofol. For example, we have previously reported that phosphorylated CFL1 is upregulated during LTP induction in elderly rats after propofol anesthesia; this may be associated with increased F-actin and depressed LTP. 7 CFL1 is a key regulator of actin depolymerization, and is inactive when phosphorylated. 14 We therefore hypothesize that CFL1 inactivation may play a role in learning and memory impairment in aged rats after propofol anesthesia; however, further study is required.
There are limitations to the present study. First, more evidence for the changes in actin and any possible causes are required. Second, combining the present findings with an electrophysiological exploration would lead to more convincing results. Third, the mechanisms underlying actin reorganization in the current study remain unclear. Overcoming these limitations will be the main objective of our further investigations. We aim to identify the role of actin dynamics in PND, provide evidence for PND mechanisms, and provide clues for clinical treatments in future studies.
In summary, our study revealed that aged rats were less resistant to propofol-induced cognitive dysfunction than young rats. The differences in findings from rats of different ages were partially associated with the reconstruction of dendritic spines and aberrant F-actin dynamic equilibrium in the hippocampus; this may be a possible mechanism of PND.
Footnotes
Author contributions
XZ was the project investigator for the funding, designed the research protocol, and collected and analyzed data. JL performed experiments and collected data.
Data Availability
The data used to support the findings of this research are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (81000473).
