Abstract
Objective
We compared efficacy and adverse outcomes following totally intravenous anesthesia (TIVA) versus combined intravenous–inhalation anesthesia (CIVIA) on hemodynamics and postoperative recovery following laparoscopic gynecological (LG) surgery at high altitudes.
Methods
We enrolled 80 ASA I or II patients scheduled for LG surgery and randomly assigned them to TIVA with propofol (group P, n = 40) or CIVIA with isoflurane (group I, n = 40). Mean arterial pressure, heart rate, pulse oxygen saturation, and partial pressure of end-tidal carbon dioxide were measured at various time points. Outcome measures were interval to spontaneous ventilation, eye opening, return to consciousness, extubation, operating room duration. Intraoperative awareness and postoperative nausea/vomiting were assessed at follow-up.
Results
No differences in hemodynamic parameters were detected in either group. Group P had a significantly shorter postoperative anesthetic recovery time and lower incidence of postoperative nausea/vomiting.
Conclusion
TIVA is superior to CIVIA for GL surgery at high altitudes.
Keywords
Introduction
Human habitation in high-altitude environments (1500–3500 meters) is no longer uncommon. Initial responses to diminished inspiratory oxygen pressure include decreased exercise performance and increased ventilation (reduced arterial PaCO2). The arterial oxygen saturation is usually unchanged, although the oxygen pressure is markedly reduced. During acclimatization, oxygen delivery to tissues is enhanced through adjustments in the respiratory, cardiovascular, and hematological systems to meet cellular oxygen demand. 1 Because of these physiological changes, anesthetics delivered at high altitude require an understanding of high-altitude medicine, specifically the altitude’s impact on anesthesia. 2
Gynecological laparoscopy is one of the most commonly performed surgical procedures. It is often the treatment of choice for ectopic pregnancies, 3 endometriosis, 4 and ovarian cystectomy. 5 Laparoscopy has been shown to be safer and more cost-effective for these procedures as well as a having a shorter recovery time than traditional laparotomy. A laparoscopic technique is favored by surgeons and patients. 6 Although TIVA and CIVIA are commonly applied during gynecological laparoscopic surgery, there are no reports that have compared these two anesthetic methods during gynecological laparoscopic surgery at high attitude. Therefore, to investigate the optimal method of anesthesia delivery under these conditions, we performed a prospective study of the effects of TIVA and CIVIA on intraoperative hemodynamic parameters and postoperative recovery. The study was performed at People’s Hospital of Linzhi Area, which is located at an altitude of more than 3000 m above sea level.
Methods
Patient information
After gaining approval from the Institutional Ethics Committee of People’s Hospital of Linzhi Area (Ethics Committee reference number: 20101220) and obtaining written informed consent, 80 American Society of Anesthesiologists (ASA) physical status I–II patients who were scheduled to undergo gynecological laparoscopic surgery at People’s Hospital of Linzhi Area (located in Linzhi, Tibet Autonomous Region, China) between January 2011 and December 2011 were enrolled in the study. All participants were lifelong residents or had been continuous residents at high altitude for more than 3 years. They were 18–56 years old (height 151–167 cm, weight 44–71 kg). None of the patients reported a history of hypertension or cardiovascular disease. Focused physical examinations of the heart, lung, liver, and kidney were unremarkable in all participants, as were the electrolyte levels. The patients were randomized, by a computed algorithm, to receive either totally intravenous anesthesia (TIVA) with propofol (group P) or combined intravenous–inhalation anesthesia (CIVIA) with isoflurane (group I). No statistically significant differences in baseline information or duration of surgery were observed between the two groups.
Anesthetic methods
All patients were intravenously (IV) administered midazolam 0.1 mg/kg and atropine 0.01 mg/kg 30 min prior to surgery. The mean arterial pressure (MAP), heart rate (HR), pulse oxygen saturation (SaO2), and partial pressure of end-tidal carbon dioxide (PETCO2) were recorded using a BRD-2000B multi-function monitor (Shenzhou, Guandong, China). Sodium lactate 300–500 mL was infused preoperatively. Patients in both groups underwent induction with atracurium 0.05 mg/kg IV, propofol 2 mg/kg IV, and fentanyl 3 µg/kg IV. After endotracheal intubation, patients were mechanically ventilated with a fraction of inspired oxygen (FiO2) of 50%, tidal volume 8–10 mL/kg, respiratory rate 12–15/min, inhalation/expiration ratio 1:2, PETCO2 26–30 mmHg, and abdominal pressure 12–14 mmHg. Intraoperatively, the tidal volume and respiratory rate were adjusted to achieve a PETCO2 of 35–45 mmHg. In the P group, anesthesia was maintained with propofol 3–10 mg/kg/h and remifentanil 0.1–0.2 µg/kg/min IV. Patients in group I received 1–3% isoflurane and remifentanil 0.1–0.2 µg/kg/min IV. A total of 10–15 mg of atracurium was administered IV intraoperatively to facilitate surgical exposure.
The patient was placed in a Trendelenburg position, and her head was elevated to a 15°–20° angle relative to the feet. TIVA or CIVIA was administered continuously for 5 min prior to insufflation, continued throughout the procedure, and maintained for 5 min following removal of the laparoscopic trocars. The Narcotrend index was adjusted to 40–50 to monitor the anesthetic depth during the surgery. Upon completion of the operation, neostigmine 2 mg IV and atropine 1 mg IV were administered to antagonize the effects of residual muscle relaxants. Extubation was performed when the patient exhibited awareness, cough and swallowing reflexes, and spontaneous ventilation with a normal tidal volume. She was then transferred to the recovery room for further observation.
Outcome measures
MAP, HR, SaO2, and PEFO2 were measured in all patients at the following time points: 30 min before induction, immediately before insufflation, 5 and 15 min after insufflation, 5 min after desufflation, 5 min after extubation. The anesthesia stop time and the time to spontaneous breathing, eye opening, return to consciousness, extubation, and time in the operating room were recorded. Eye opening in this study was defined as 2 in the Observer’s Assessment of Alertness/Sedation Scale, and return to consciousness was defined as 1. An observer blinded to the anesthesia grouping assessed the patients 24 h postoperatively regarding intraoperative awareness, postoperative nausea/vomiting, and other complications.
Statistical analysis
All data were expressed as the mean ± standard deviation. Student’s t-test and the χ2 test were performed using SPSS15.0 software (SPSS, Chicago, IL, USA).
Results
Demographic characteristics of participants
Demographic characteristics of participants
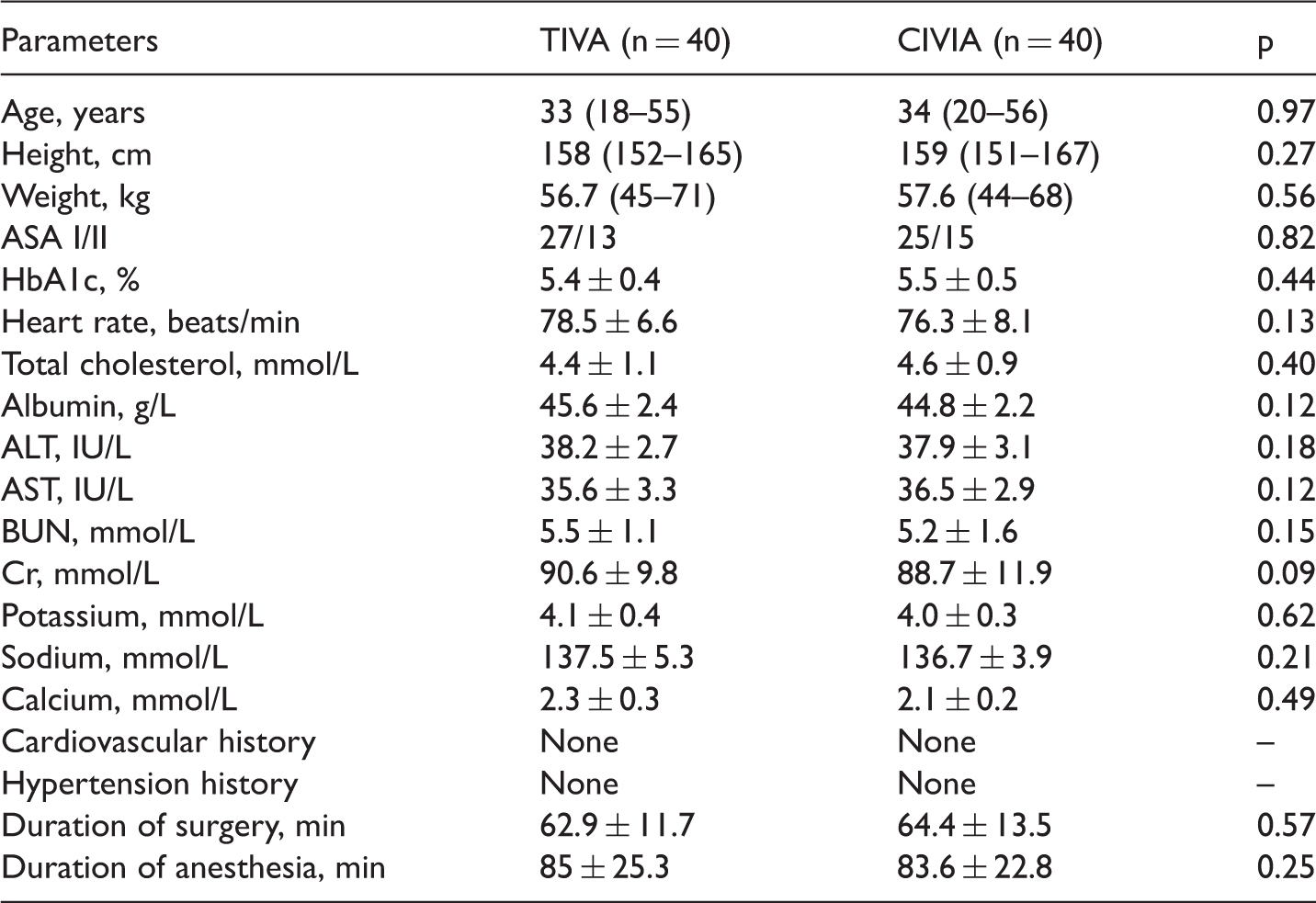
ASA, American Society of Anesthesiologists; HbA1c, hemoglobin A1c; ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Cr, creatinine.
Values are given as the number and range or mean ± SD.
There were no significant differences in any of the parameters between the TIVA and CIVIA groups
Comparison of MAP, HR, SaO2, and PETCO2 at various points in time
MAP, HR, SaO2, and PETCO2 at multiple time points for the TIVA and CIVIA groups

MAP, mean arterial pressure; HR, heart rate; SaO2, pulse oxygen saturation; PETCO2, partial pressure of end-tidal carbon dioxide; BI, before induction; BP, before insufflation; AP, after insufflation; P, TIVA with propofol (n = 40); I, CIVIA with isoflurane (n = 40).
Results are given as mean ± SD.
*P < 0.05, **P < 0.01: compared with prior to induction.
Postoperative recovery
Recovery status and intraoperative/postoperative complications in TIVA and CIVIA groups

P, TIVA with propofol (n = 40); CIVIA with isoflurane (n = 40); SBR, spontaneous breathing recovery; EO, eye opening; OR: return to consciousness; ET, extubation; RT, time in operating room; IA, intraoperative awareness; PNV, postoperative nausea and vomiting.
Results are given as mean ± SD.
P < 0.05, **P < 0.01: compared with group I.
Postoperative Adverse Events
Intraoperative awareness was not observed in either group. In contrast, the incidence of postoperative nausea and vomiting within 24 h of surgery was significantly lower in group P patients than in group I patients (P < 0.05) (Table 3).
Discussion
Lingzhi Hospital is located in the Qinghai–Tibet Plateau region at an altitude of 3000 m above sea level. The geographic and climatic environment of this region affects human physiology and poses a unique challenge in perioperative anesthesia management. The pressure of inspired oxygen at high altitudes may lead to an increased incidence of anesthesia-related complications compared with that at lower altitudes7,8 A high-altitude environment generally causes humans to acclimatize. Particularly, there are respiratory, cardiovascular, and hematological adjustments that improve the transport and utilization of O2. Genes coding for proteins involved in oxygen transport, growth of blood vessels, and erythropoiesis are activated. 8 The catalytic activity and expression of certain isoenzymes of cytochrome P450 at high altitude are modified, leading to changes in relative drug metabolism and pharmacokinetics. 9 Additionally, patients receiving anesthetics at high altitude who have fasted during the preoperative period require more vigorous hydration than their counterparts at low altitude. 10 Therefore, recovery from anesthesia must be carefully managed.
To facilitate early postoperative recovery, high-altitude anesthesia should focus on restoring PaO2 and PaCO2 to preoperative levels rather than reference levels. 7 No standards for PaO2, PaCO2, and PETCO2 have been established for high altitudes, however, and the reference ranges for PaCO2 and PETCO2 levels in individuals residing at sea level are not generalizable to those at high altitude. Some clinical investigations have demonstrated that PETCO2 can indirectly reflect PaCO2.11,12 Under normal conditions, PETCO2 is lower than PaCO2 by 3–6 mmHg. 13 At an altitude of 3000 m, the atmospheric pressure of our hospital is 532 mmHg and the PaCO2 is 34 mmHg, resulting in a calculated PETCO2 of 28–31 mmHg. 2 In this study, preoperative PETCO2 in all patients was found to be 26–32 mmHg, thereby confirming the predicted values, which should be the PETCO2 reference value for high-altitude patients (ASA I–II). Therefore, during laparoscopy at high altitude, PETCO2 should be monitored continuously and respiratory parameters adjusted to maintain PETCO2 at pre-anesthesia levels to prevent hypercapnia.
After induction of anesthesia, MAP and HR were decreased in all patients, secondary to the sympatholytic effects of anesthetics. PETCO2 was reduced after induction and before insufflation, likely due to hyperventilation. SaO2 was elevated in all patients after insufflation until after extubation because of mechanical ventilation with high FiO2. The HR in all patients had decreased 15 min after desufflation. This effect is likely attributable to increased peritoneal pressure. Nonetheless, hemodynamic parameters were not significantly different between the two groups at any time point during the surgery.
Group P patients had a faster postoperative recovery and a lower incidence of nausea and vomiting during the first postoperative day. Based on this analysis, both TIVA and CIVIA can be used for gynecological laparoscopic surgery. In this study, however, TIVA provided not only more rapid postoperative recovery from anesthesia and high-quality awareness, it lowered the incidence of postoperative nausea and vomiting. Numerous studies have corroborated the point that at lower altitudes TIVA allows the most rapid recovery from anesthesia with the fewest postoperative side effects.14–17 Our results demonstrated that TIVA displayed the same advantages at high altitude. Furthermore, TIVA is not a respiratory tract irritant and does not contaminate the air in the operating room.
Puri et al. 18 found significantly lower baseline heart rates (72.0 ± 9.83 vs. 88.0 ± 12.1 bpm; p < 0.04) and an attenuated heart rate response in patients (in Leh, India, which is 3505 m above sea level) to the stresses of endotracheal intubation and surgical incision at high altitude, compared with those at low-altitude. The baseline heart rate (73.6 ± 7.8 bpm) and the heart rate response to the stresses of insufflation in our study were similar to those in the study by Puri et al. 18 Compared with another, similar study conducted at low altitude (Beijing, China), our study shows a lower baseline heart rate (73.6 ± 7.8 vs. 76 ± 4 bpm) for participants at high altitude. Whether there is a significant difference remains unknown, however, as we were not able to calculate the statistical difference. It seems, however, that the heart rate response and postoperative recovery time (11.20 ± 4.30 vs.10.4 ± 7.4 min) in our study were similar to those in the Beijing study. 17
The limitation of this study is that neither newcomers to high altitude nor persons with a history of cardiovascular and/or pulmonary disease were recruited for this study. Those people experience less adaptation to the high altitude than do high-altitude residents and may display different responses to anesthesia management. 8 Further study is required to investigate the effects of TIVA and CIVIA on those individuals.
In conclusion, it is important to know that, when administering anesthetics at high altitude for laparoscopic gynecological surgery, TIVA has several advantages over CIVIA, including its shorter recovery time and fewer postoperative complications.
Footnotes
Acknowledgements
This project was sponsored by National Natural Scientific Foundation of China (No. 81271390). We thank Dr. Peihua Cao for assistance with the paper’s revision.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
