Abstract
Objective
To investigate the short- and long-term effects of neonatal single exposure to propofol anesthesia on neurocognitive function.
Methods
Institute of Cancer Research mice were intraperitoneally injected with fat emulsion or propofol at a dose of 50 or 100 mg/kg once at postnatal day 12. At 1, 4, 12, and 24 weeks following anesthesia, learning and memory abilities were evaluated using the novel object recognition and Morris water maze experiments. Histopathology and synaptic function of the hippocampus were assessed using hematoxylin and eosin and Golgi-Cox staining, and the expression levels of cyclic adenosine monophosphate–response element binding protein (CREB), brain-derived neurotrophic factor (BDNF), and postsynaptic density protein-95 (PSD95) in the hippocampus were detected using western blotting.
Results
There was no significant difference in total exploration time, recognition index, and time and crossings in the target quadrant between the control and propofol-exposed groups. No hippocampal pathological change and abnormal dendritic length or spine density were observed. Furthermore, no significant alterations in the expression levels of CREB, BDNF, or PSD95 were detected.
Conclusions
Neonatal single exposure to propofol anesthesia does not impair recognition function in either the short- or long-term.
Keywords
Introduction
Administering general anesthesia to neonates and infants who require surgical procedures is essential for ensuring comfort throughout the operation and supporting successful surgical outcomes.1,2 Among intravenous general anesthetics, propofol (PPF) is the most widely used drug in pediatric anesthesia, particularly in neonates, because it acts rapidly, has a short recovery time, and exhibits antiemetic effects suitable for induction and maintenance of anesthesia in clinical practice.3,4 In clinical environment, most neonates and infants receive PPF anesthesia only once or at long intervals. However, there is growing clinical concern about whether even “single exposure” or “long-interval” PPF use may affect short- and long-term cognitive function. 5 Moreover, existing research has not fully addressed these concerns.
Current evidence available on the neurocognitive risks of PPF in neonates is largely limited to repeated, short-interval exposures. It has been reported that multiple administrations of PPF in neonates over short intervals may result in long-lasting behavioral issues in adulthood. 6 Additionally, research has shown that repeated exposure to PPF notably impacts learning and memory, as demonstrated by novel object recognition (NOR) and Morris water maze (MWM) assessments. 7 During the neonatal stage, repeated PPF exposure may impair hippocampal synaptic plasticity and cognitive recognition capabilities in adult rats. 8 The signaling pathway involving cyclic adenosine monophosphate (cAMP)–response element binding protein (CREB) and brain-derived neurotrophic factor (BDNF) plays a crucial role in mediating damage to synaptic plasticity and excitability, contributing to cognitive deficits in adulthood following neonatal PPF exposure. 9 Additionally, studies have highlighted the role of postsynaptic density protein-95 (PSD95) in long-term synaptic toxicity and cognitive dysfunction resulting from neonatal exposure to PPF. 10
Although previous studies have extensively investigated the neurocognitive effects of repeated PPF exposure in neonates, the impact of a single exposure (i.e. at doses equivalent to those used in neonatal clinical practice) remains underexplored. This discrepancy is particularly concerning, as single exposure is the most common clinical scenario encountered by anesthesiologists and parents; however, evidence regarding their potential impact is lacking.11,12
Therefore, the present study aimed to investigate whether a single neonatal exposure to PPF at clinically-relevant doses produces short- or long-term cognitive deficits. We focused on two specific cognitive domains that have previously been linked to PPF exposure: (a) recognition memory (assessed via NOR) and (b) spatial learning and memory (assessed via MWM).
Materials and methods
Animals
Institute of Cancer Research (ICR) male mice were obtained from Pengyue Experimental Animal Breeding Co., Ltd., Jinan, China (certificate no. SCXK 20220006). Mice were maintained at 24°C under a 12-h light/12-h dark cycle, with unrestricted access to food and water. All animal experiments were approved by the Institutional Animal Care and Use Committee of Tongliao People’s Hospital, Tongliao, Nei Menggu Province, China (approval no. TLRMYY-20241036, approval date: 18 February 2024). The study followed international and national guidelines for the humane treatment of animals and complied with relevant legislation.
Experimental groups and drug treatments
Mice were randomly categorized into three groups (n = 10): (a) control group; (b) PPF 50 mg/kg (PPF50) group; and (c) PPF 100 mg/kg (PPF100) group. The control group received a single intraperitoneal injection of fat emulsion as the control solvent, whereas the PPF50 and PPF100 groups received a single intraperitoneal injection of PPF (50 or 100 mg/kg, respectively) on postnatal day 12. 13 The PPF dosage was determined based on the median effective dose (ED50) and 95% effective dose (ED95) values, which ranged from 3.5 to 6.8 mg/kg and 7.1 to 16.9 mg/kg, respectively. These doses were converted to mouse equivalents using the body surface area method, 9 yielding mouse equivalents ranging from 31.85 to 61.88 mg/kg (ED50) and 64.61 to 153.79 mg/kg (ED95). A heating pad was used to maintain the animal’s body temperature at approximately 37°C during anesthesia. This study was conducted according to the planned schedule (Figure 1). Mice were euthanized via intraperitoneal injection of pentobarbital sodium upon completion of the behavioral tests.

The schedule of the experimental procedure.
Behavioral test
NOR test
The NOR test was used to evaluate recognition memory and learning in rodents, using an open-field box with a length, width, and depth of 50 cm as the experimental setup. The assessment consisted of three stages: (a) habituation; (b) familiarization; and (c) testing. On day 1 (habituation), each mouse was allowed to freely explore the arena for 5 min without any objects present. On day 2 (familiarization), a single mouse was placed in the arena containing two identical sample objects (A and A) and allowed to explore for 5 min. After a 24-h interval, one of the objects from the previous phase was replaced with a novel object (A and B), and the mouse was allowed to explore the arena freely for 5 min. During the testing phase, the mice were placed in the center of the same box. Engagement with an object was considered exploratory behavior if the mouse oriented its nose toward it at a distance of ≤1 cm or actively touched or sniffed it. Actions such as sitting on or turning around the objects were not considered exploration. To ensure consistent scent across trials, the box and objects were sanitized with 70% isopropyl alcohol between each session. The primary outcomes were as follows: (a) total exploration time of both the objects during the testing stage and (b) recognition index, calculated as recognition index (%) = (Novel object B exploration time∕Total A + B exploration time) ×100. 14 A higher recognition index indicates better recognition memory.
MWM test
Hippocampal-dependent spatial learning and memory abilities were assessed using the MWM experiment. A round black pool with a diameter of 180 cm and a depth of 50 cm was filled with water to a height of 30 cm. The water temperature was maintained at 23°C ± 1°C. An invisible platform, with a diameter of 10 cm, was submerged 1 cm below the water surface and placed at the center of quadrant IV. Four starting points were set at equal distances from the edge of the pool in quadrants I, II, III, and IV. A high-definition camera was mounted above the pool to record the movement trajectories of the mice, which were analyzed using the EthoVision XT 14 system. Because mice prefer a dry environment and avoid water, they instinctively searched for the platform to escape the water. The MWM procedure consisted of a training phase followed by a testing trial. Over 4 consecutive days during the training phase, each mouse was placed at the center of each quadrant (I, II, III, and IV) facing the pool wall and given 60 s to locate the platform. If a mouse failed to locate the platform within this time, the experimenter guided it to the platform and allowed it to remain there for 15 s. Mice were always removed from the pool while on the platform. If a mouse jumped back into the water after reaching the platform, it was returned to the platform, dried with a towel, and then placed in its home cage. The escape latency (the time taken to locate the hidden platform) and swimming speed were recorded. After the final training trial, the testing phase commenced. The platform was removed, and each mouse was introduced into the pool at location (II), the point farthest from the target quadrant. The mouse was allowed to swim freely for 60 s. During the training and testing trials, the trajectories of the mice were tracked using a computerized video tracking system (EthoVision, Noldus, Netherlands). The recorded parameters included the following: (a) During training. Swimming speed and escape latency and (b) During testing. Latency to first entry into the target zone (the area where the platform was previously located), time spent in the target zone, and number of crossings over the original platform position. 15
Hematoxylin and eosin (H&E) staining
After the mice were euthanized, hippocampal tissue was promptly extracted on ice, subsequently immersed in 4% paraformaldehyde for fixation, and stored at 4°C to preserve structural stability. The fixed tissue was trimmed, rinsed overnight in running water, dehydrated through a graded series of alcohol solutions, embedded in paraffin, and coronally sectioned at a thickness of 4 μm. For dewaxing, the sections were sequentially treated with the following solutions: xylene (I) at 37°C for 10 min, xylene (II) at 37°C for another 10 min, absolute ethanol (I) for 10 min, absolute ethanol (II) for 10 min, 95% ethanol for 5 min, 90% ethanol for 5 min, 80% ethanol for 5 min, and 70% ethanol for 5 min. The sections were stained with H&E, sealed with neutral gum, and kept at room temperature for 2 h. After the neutral gum had dried, the samples were examined under a microscope (E200, Nikon, Japan), and the morphological characteristics of hippocampal neurons in each group were documented and saved for further analysis.
Golgi-Cox staining
After the mice were sacrificed, the brain tissue was rapidly harvested, placed on ice, and immersed in an impregnation solution prepared by mixing equal volumes of Solutions A and B. The solution was replaced with a fresh one the following day, and the tissue was stored in dark at room temperature for 2 weeks to allow adequate dye impregnation. The brain tissue was then transferred to Solution C (fixative/differentiation solution) and incubated at 4°C for 72 h. Using a cryotome, 100 µm sections were prepared. A drop of Solution C was applied to gelatin-coated microscope slides. After drying, the sections were briefly rinsed to remove any remaining Solution C and then stained according to the manufacturer’s protocol of the FD Rapid Golgi Stain Kit (FD, Catalog No. PK401A). Morphological analysis of the hippocampal CA1 region was performed using an L300D microscope (Nikon, Japan). 16
Western blotting
The hippocampal tissues were homogenized in radioimmunoprecipitation assay (RIPA) lysis buffer containing protease inhibitors, followed by centrifugation at 14,000 r/min for 15 min at 4°C to clarify the lysate. The resulting supernatant was collected as the total protein sample. After quantification, 30 μg of protein was loaded onto a 10% sodium dodecyl sulfate–polyacrylamide (SDS–PAGE) gel for separation. The proteins were then transferred onto a nitrocellulose membrane and blocked with 5% skim milk in Tris-buffered saline with Tween-20 (TBST) for 2 h at room temperature to prevent nonspecific binding. The membranes were incubated overnight at 4°C with the primary antibodies against CREB (1:1000 dilution, ab32515, Abcam, Cambridge, UK), BDNF (1:1000 dilution, ab216443, Abcam, Cambridge, UK), and PSD95 (1:2000 dilution, ab238135, Abcam, Cambridge, UK), followed by TBST washes. The membranes were then incubated at room temperature with horseradish peroxidase (HRP)–conjugated secondary antibodies for 2 h. The experiment was performed in triplicate. Protein bands were visualized using enhanced chemiluminescence reagent and analyzed with a gel imaging system (LAS4000, GE, USA). Semiquantitative densitometric analysis was conducted using ImageQuant LAS4000 V1.22 software, and target protein expression was normalized to that of β-actin (dilution. 1:1000, ab8226, Abcam, Cambridge, UK).
Statistical analysis
Statistical analyses were performed using Statistical Package for Social Sciences (SPSS 17.0) software. Differences in the escape latency and swimming speed in the MWM test among more than two groups were analyzed using two-way analysis of variance (ANOVA), whereas data from other behavioral experiments were analyzed using one-way ANOVA. The Bonferroni post hoc test was applied following ANOVA. The two-tailed Student’s t-test was used for multiple comparisons of data from Golgi-Cox staining and western blotting analyses. Data were presented as means ± SD. Statistical significance was set at a P value of <0.05.
Results
Effect of single neonatal exposure to PPF anesthesia on short- or long-term recognition memory ability
To determine whether a single neonatal exposure to PPF anesthesia induces abnormal behavior, we performed a series of short- and long-term behavioral assessments on mice exposed to PPF, beginning with the NOR test. The design of the NOR test is shown in Figure 2(a), and representative movement trajectories are presented in Figure 2(b). The preference for exploring the novel object reflects the animal’s recognition memory ability. As shown in Figure 2(c), the control, PPF50, and PPF100 groups exhibited comparable total exploration times for both objects across different time points following PPF anesthesia: (a) 1 week. F (2, 27) = 0.07398, P = 0.928; (b) 4 weeks. F (2, 27) = 0.02533, P = 0.9750; (c) 12 weeks. F (2, 27) = 0.06352, P = 0.9386; and (d) 24 weeks. F (2, 27) = 0.1068, P = 0.8991. Mice typically prefer to explore and sniff the novel objects. However, the result (Figure 2(d)) demonstrated no significant differences in the recognition index between the control and PPF groups across different time points: (a) 1 week. F (2, 27) = 0.5901, P = 0.5613; (b) 4 weeks. F (2, 27) = 0.5454, P = 0.5859; (c) 12 weeks. F (2, 27) = 0.5276, P = 0.5960; and (d) 24 weeks. F (2, 27) = 0.05841, P = 0.9434.

Effect of single neonatal exposure to PPF anesthesia on recognition memory in mice. (a) Experimental design of the NOR test. (b) Representative movement tracks of the three groups in the NOR test. (c) Total exploration time and (d) recognition index. n = 10 mice per group. Data are represented as mean ± SD from three independent experiments. No significant differences were observed in total exploration time and recognition index between the control and PPF groups. PPF: propofol; NOR: novel object recognition.
Effect of single neonatal exposure to PPF anesthesia on short-or long-term spatial learning and memory abilities
The effects of single neonatal exposure to PPF anesthesia on short- and long-term spatial learning and memory abilities were assessed using the MWM test. The design of the MWM test is presented in Figure 3(a). As training progressed, the control, PPF50, and PPF100 groups demonstrated a moderate increase in the swimming speed (Figure 3(b)) and a decrease in the escape latency across all tested time points (Figure 3(c)), indicating spatial learning ability over time. Two-way ANOVA of the swimming speed revealed no significant effects of training days across different time points: (a) 1 week. F (3, 116) = 2.205, P = 0.0912; (b) 4 weeks. F (3, 108) = 1.618, P = 0.1895; (c) 12 weeks. F (3, 112) = 1.008, P = 0.3920; and (d) 24 weeks. F (3, 102) =2.117, P = 0.1027. Additionally, the effect of groups on the swimming speed was nonsignificant: (a) 1 week. F (2, 116) = 1.932, P = 0.1495; (b) 4 weeks. F (2, 108) = 1.095, P = 0.3381; (c) 12 weeks. F (2, 112) = 0.4414, P = 0.6442; and (d) 24 weeks. F (2, 102) = 1.058, P = 0.3508. Similarly, the effect of the interaction between days and groups was nonsignificant: (a) 1 week. F (2, 116) = 1.932, P = 0.1495; (b) 4 weeks. F (2, 108) = 1.095, P = 0.3381; (c) 12 weeks. F (2, 112) = 0.4414, P = 0.6442; and (d) 24 weeks. F (2, 102) = 1.058, P = 0.3508. The escape latency data showed significant differences in training days across different time points: (a) 1 week. F (3, 106) = 29.58, P < 0.0001; (b) 4 weeks. F (3, 108) = 22.57, P < 0.0001; (c) 12 weeks. F (3, 107) = 24.37, P < 0.0001; and (d) 24 weeks. F (3, 103) = 30.09, P < 0.0001. However, the effects of groups on the escape latency was nonsignificant: (a) 1 week. F (2, 106) = 0.008734, P = 0.9913; (b) 4 weeks. F (2, 108) = 0.05170, P = 0.9496; (c) 12 weeks. F (2, 107) = 0.01133, P = 0.9887; and (d) 24 weeks. F (2, 103) = 0.2783, P = 0.7576. Similarly, the effect of the interaction between days and groups was nonsignificant: (a) 1 week. F (6, 106) = 0.02010, P > 0.9999; (b) 4 weeks. F (6, 108) = 0.02647, P > 0.9999; (c) 12 weeks. F (6, 107) = 0.02798, P > 0.9999; and (d) 24 weeks. F (6, 103) = 0.2370, P = 0.9634. The typical paths of mice on the 5th testing day are shown in Figure 3(d). In the memory retrieval test on day 5, there were no significant differences among the three groups in latency to first entrance to the target zone across different time points (Figure 3(e)): (a) 1 week. F (2, 27) = 0.2805, P = 0.7575; (b) 4 weeks. F (2, 27) = 0.3174, P = 0.7307; (c) 12 weeks. F (2, 27) = 0.09555, P = 0.9092; and (d) 24 weeks. F (2, 27) = 0.08078, P = 0.9226. Compared with the control group, the PPF50 and PPF100 groups demonstrated no significant reduction in the time spent in the target zone (Figure 3(f)): (a) 1 week. F (2, 27) = 0.06178, P = 0.9402; (b) 4 weeks. F (2, 27) = 0.3170, P = 0.7310; (c) 12 weeks. F (2, 27) = 0.1979, P = 0.8216; and (d) 24 weeks. F (2, 27) = 0.3651, P = 0.6975. Moreover, there were no significant differences in the number of target crossings among the three groups across different time points (Figure 3(g)): (a) 1 week. F (2, 27) = 0.05397, P = 0.9476; (b) 4 weeks. F (2, 27) = 0.2854, P = 0.7540; (c) 12 weeks. F (2, 27) = 0.5311, P = 0.5940; and (d) 24 weeks. F (2, 27) = 0.2727, P = 0.7634.

Effect of single neonatal exposure to PPF anesthesia on learning and spatial memory in mice. (a) Experimental design of the MWM test. (b) Swimming speed of the three groups during the training phase of the MWM test. (c) Escape latency of the three groups during the training phase of MWM test. (d) Representative movement tracks of the three groups in the MWM test on day 5 of testing. (e) Latency to the first entrance to the target zone on day 5. (f) Time spent in the target quadrant on day 5 and (g) number of crossings over the previous platform location on day 5. n = 10 mice per group. Data are presented as mean ± SD from three independent experiments. No significant differences were observed in swimming speed, escape latency, latency to the first entrance to the target zone, time spent in the target zone, or number of target crossings between the control and PPF groups. PPF: propofol; MWM: Morris water maze.
Effect of single neonatal exposure to PPF anesthesia on short- and long-term histopathological changes in the hippocampus
To assess whether single neonatal exposure to PPF anesthesia induced short- or long-term histopathological alterations in the hippocampus, the morphology of hippocampal subregions CA1, CA3, and dentate gyrus (DG) was assessed using H&E staining. As shown in Figure 4(a) to (d), neuronal cells in the control group were neatly arranged and densely distributed, with regular morphology, clear cellular structure, lightly stained cytoplasm, centrally located nuclei, and prominent nucleoli. The neuronal morphology in the PPF50 and PPF100 groups at 1, 4, 12, and 24 weeks was comparable to that in the control group. No apparent neuronal cytoplasmic vacuolation or nuclear irregularities were observed. The number of neurons was determined, revealing no statistically significant differences in the CA1, CA3, and DG subregions of the hippocampus in the PPF50 or PPF100 groups compared with that in the control group, as shown in Figure 4(e) to (g).
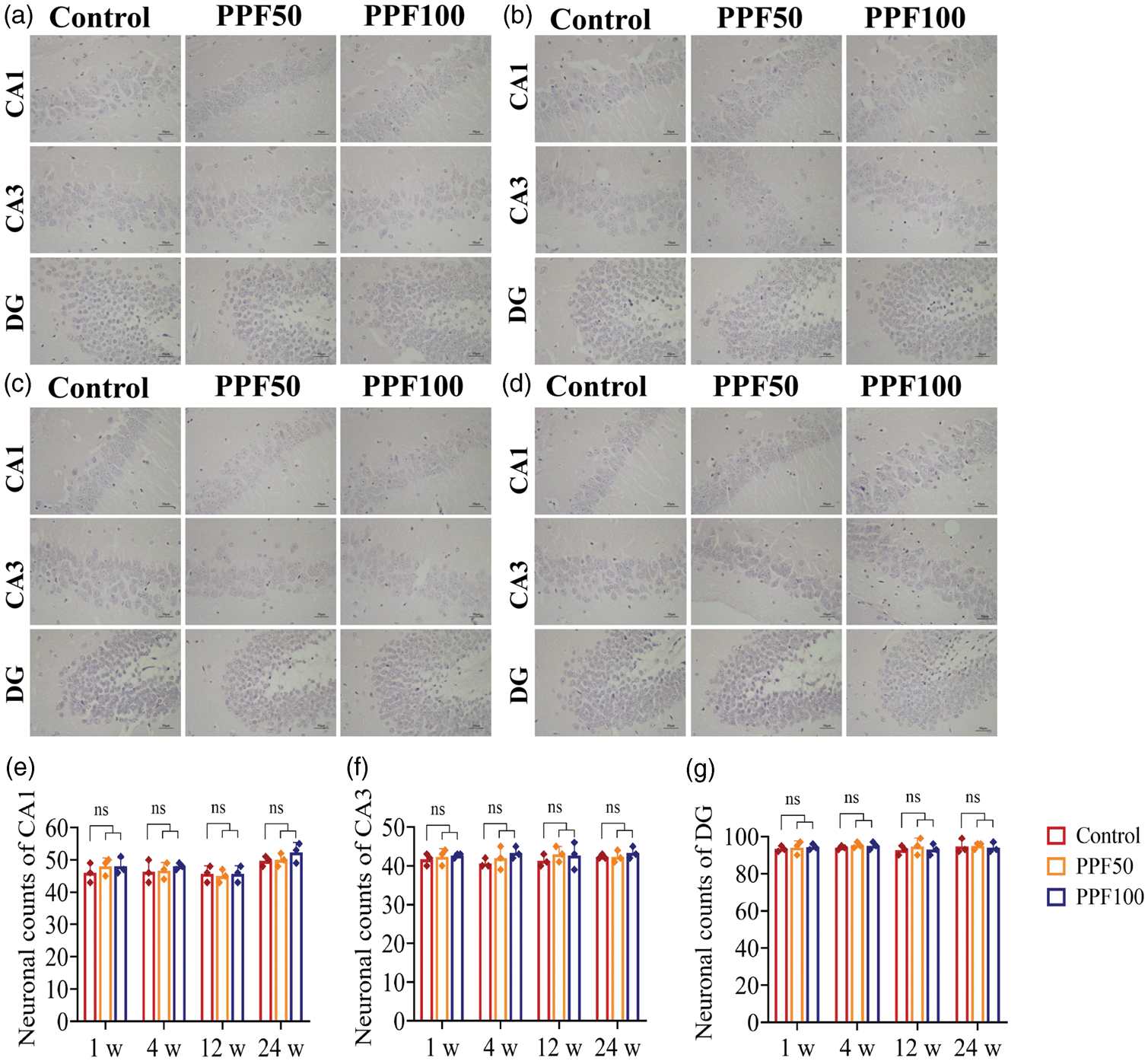
Effect of single neonatal exposure to PPF anesthesia on hippocampal pathology in mice.
Effect of single neonatal exposure to PPF anesthesia on short- and long-term synaptic plasticity in the hippocampal CA1 region
To determine whether single neonatal exposure to PPF anesthesia affects short- or long-term synaptic plasticity in the hippocampal CA1 region, we performed Golgi-Cox staining, which enables detailed visualization of neuronal dendritic branches and spines. The density of dendritic spines and total length of dendrites are key morphological indicators of synaptic plasticity. Microscopic images of the hippocampal CA1 region with Golgi-Cox staining, as shown in Figure 5(a), revealed no statistically significant differences in the dendritic spine density (Figure 5(b)) or total dendritic length (Figure 5(c)) between the PPF50 or PPF100 groups versus the control group at 1, 4, 12, or 24 weeks. These findings suggest that single neonatal PPF anesthesia exposure does not affect the key morphological correlates of synaptic plasticity in the hippocampal CA1 region in either the short- or long-term.

Effect of single neonatal exposure to PPF anesthesia on synaptic plasticity in the hippocampal CA1 region. (a) Representative images of hippocampal CA1 neurons after Golgi-Cox staining (×200), Scale bar = 50 μm. (b) Quantitation of dendritic spine density and (c) quantitation of total dendritic length. Experiments were performed in triplicate. Data are presented as mean ± SD. No significant differences were observed in length of dendrites and dendritic spine density between the control and PPF groups. PPF: propofol.
Effects of single neonatal exposure to PPF anesthesia on short- and long-term expression of CREB, BDNF, and PSD95 in the hippocampus
CREB, BDNF, and PSD95 are critical factors involved in the formation and protection of learning and memory and the facilitation of synaptic plasticity formation and maintenance. We determined the expression levels of CREB, BDNF, and PSD95 in the hippocampus using western blot analysis, and the representative protein bands are presented in Figure 6(a). As summarized in Figure 6(b), in the short- (1 or 4 weeks) and long-term assessments (12 or 24 weeks) following PPF anesthesia, compared with the control group, there was no significant difference in the expression of these proteins in the PPF50 or PPF100 groups (both, P > 0.05). Our findings indicate that single exposure to PPF did not affect the expression of proteins over short-term or long-term, further supporting that PPF did not impair learning, memory, or synaptic plasticity.

Effect of single neonatal exposure to PPF anesthesia on the expression of CREB, BDNF, and PSD95 in the hippocampus. (a) Representative western blot bands for CREB, BDNF, and PSD95 and (b) quantitative analysis of hippocampal CREB, BDNF, and PSD95 protein expressions in the control and PPF groups. Data are presented as mean ± SD from three independent experiments. No significant differences were observed in the expression levels of CREB, BDNF, and PSD95 of hippocampus between the control and PPF groups. PPF: propofol; CREB: cyclic adenosine monophosphate–response element binding protein; BDNF: brain-derived neurotrophic factor; PSD95: postsynaptic density protein 95.
Discussion
Studies have reported that the brains of developing individuals are vulnerable to PPF anesthesia exposure, revealing neurotoxic impacts, particularly on the hippocampus, which plays a crucial role in learning and memory.17,18 Investigations on animals and humans have suggested that recurring exposure to general anesthetics or high doses during the neonatal stage may result in considerable deficits in learning and memory later in life.19,20 Despite this knowledge, whether a single dose of PPF anesthetic can induce significant neurotoxic effects in newborn infants remains debatable. 21 In the present study, low and high clinically equivalent doses of PPF were administered at P12 in mice, a developmental stage equivalent to human neonates.
The NOR test, based on the innate preference of mice for new experiences, is a widely used behavioral assay for evaluating different facets of recognition memory. 22 The MWM test is a standard approach for assessing cognitive abilities in laboratory rodents and is particularly useful for identifying variations in learning and spatial memory capabilities. 23 The results suggested that the PPF50 and PPF100 groups demonstrated no statistically significant difference from the control in the recognition index measured using the NOR test. Similarly, in the MWM test, there were no differences in the swimming speed, escape latency, latency to the first entrance into the target zone, time spent in the target zone, and number of target crossings at any time point (1, 4, 12, and 24 weeks) following PPF exposure. Therefore, these behavioral outcomes reveal that PPF anesthesia did not significantly impair recognition memory, spatial learning ability, and spatial memory, either in the short- or long-term.
The hippocampus is a critical brain region involved in multiple cognitive and behavioral processes. 24 Repeated exposure to PPF can induce rapid neuronal degeneration, including apoptosis and necrosis, as well as immediate cellular responses such as neuronal injury and disrupted mitochondrial dynamics. 25 We examined the morphological changes of neurons in the hippocampus. H&E staining of the CA1, CA3, and DG regions revealed that single PPF exposure did not impair the short- and long-term hippocampal neuron morphology. Recent studies have suggested that synaptic plasticity could also be involved, which can be evaluated using the Golgi-Cox method. 26 In this study, Golgi-Cox staining revealed no significant differences in synaptic plasticity-related morphological indicators between the control and PPF-exposed mice in either the short- or long-term. Thus, single PPF exposure did not induce impairment of synaptic plasticity in the hippocampus.
It is well established that the transcription factor CREB is essential for cognition, serving as a key molecular target required for the establishment and maintenance of learning and memory.27,28 The expression of CREB-regulated genes is essential for long-term memory function and the mechanisms underlying synaptic plasticity. 29 Activation of CREB promotes the transcription of key proteins, particularly BDNF, which are necessary for activity-dependent plasticity. 30 BDNF functions as a major enhancer of synaptic plasticity and memory development. 31 Additionally, PSD95 is the primary scaffold protein located in the dendritic spine within the postsynaptic density of glutamatergic synapses. 32 It plays a crucial role in the arrangement of the PSD’s architecture by interacting with membrane receptors and ion channels, thereby supporting structural and functional synaptic plasticity. 33 The analysis reveals that CREB, BDNF, and PSD95 expression levels in the hippocampal tissues of the PPF-exposed mice remained unchanged compared with those in the controls. Furthermore, stable PSD95 level and the Golgi-Cox staining showed that single PPF exposure did not affect the hippocampal synaptic plasticity. Mechanistically, single PPF exposure did not alter the expression of hippocampal CREB, BDNF, or PSD95.
Additionally, compared with our study, variations in experimental design, including species or strain, developmental period, exposure paradigm, and outcomes, account for the discrepancies reported in previous studies of PPF neurotoxicity.8,34–36 Key factors contributing to these differences include the frequency of exposure (single vs. repeated), developmental stage (early neonatal vs. infantile), and dosage (supratherapeutic vs. clinical). Notably, repeated or high-dose exposure during earlier developmental stages produce significant effects. Our findings indicate that a single, clinically-relevant dose of PPF did not result in short- or long-term cognitive impairment in infantile P12 mice. These results offer a new perspective on neonatal PPF-induced neurotoxicity. However, the study has limitations, including the use of only male ICR mice, a limited dose range and cognitive assessments, the relatively short follow-up period, and a small sample size. Further research addressing these limitations is warranted.
Footnotes
Acknowledgments
We are grateful to Tongshen Liu for his valuable suggestions and assistance during the Golgi-Cox staining process.
Author contributions
SL L, T W, Y Z, and PF L conceived and designed the study. JY L and JY H participated in sample collection. M F and ZJ S conducted the data analysis. YH W, HR B, and ZJ S assisted in revising the manuscript. SL L, M F, Y D, and YF S drafted the original manuscript. T W, Y Z, and PF L reviewed and edited the manuscript. All authors have read and approved the final manuscript.
Data availability statement
The data in the study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
All authors declare that they have no conflict of interest.
Funding
This study was supported by the Natural Science Foundation of Shandong Province (ZR2017PH073).
