Abstract
Uterine leiomyosarcoma has the characteristics of high malignancy, a poor prognosis, and a high recurrence rate. Surgery is the main treatment option, supplemented by chemotherapy and radiotherapy. We report on a patient with recurrent uterine leiomyosarcoma who was treated with high-intensity focused ultrasound combined with chemotherapy. Tumor growth was controlled and the patient’s survival time was prolonged. High-intensity focused ultrasound combined with chemotherapy may thus provide a new treatment strategy for patients with recurrent and surgically difficult uterine leiomyosarcoma.
Keywords
Introduction
Uterine leiomyosarcoma is a rare, extremely aggressive uterine cancer with a high recurrence rate. Safe and effective treatment is therefore required. High-intensity focused ultrasound (HIFU) is a noninvasive thermotherapy for the treatment of benign and malignant solid masses, and is regarded as one of the most promising and representative noninvasive therapeutic modalities for cancer. HIFU causes irreversible coagulation necrosis in the target region, eventually resulting in effective lesion resection by inducing thermal, cavitation, and mechanical effects. HIFU can also improve the sensitivity of tumor cells to chemotherapeutic drugs, and can thus complement the effects of chemotherapy. In this study, we report the case of a patient with recurrent uterine leiomyosarcoma who benefited from HIFU combined with chemotherapy, thus providing a reference for the treatment of patients with recurrent leiomyosarcomas who are unsuitable for surgery.
Case report
A 51-year-old woman was admitted to our hospital on 26 July 2013 for “postoperation of subtotal hysterectomy for 2 years, and abdominal distension for 3 months”. A palpable mobilizable mass about 10 × 10 cm was detected in the pelvic cavity during vaginal bimanual examination. Pelvic computed tomography (CT) showed an irregular and lobulated mass about 10.9 × 8.6 cm. Surgery was carried out on 30 July 2013, with removal of the cervical stump and resection of the tumor, with no residual tumor. The tumor was diagnosed pathologically as a uterine leiomyosarcoma (Figure 1). Immunohistochemical results showed estrogen receptor−, progesterone receptor+, Ki67 10%, smooth muscle actin++, desmin++, CD117−, CD34−, and DOG-1−. The patient received six cycles of chemotherapy with gemcitabine and docetaxel combined with radiotherapy. She underwent two further operations in February and December 2015 to remove recurrent pelvic tumors, with no visible masses remaining after surgery. The pelvic masses were all diagnosed as metastatic leiomyosarcoma. The patient unfortunately developed incomplete intestinal obstruction after surgery. In March 2016, she underwent pelvic magnetic resonance imaging (MRI), which revealed several recurrent nodules in the surgical area, of which the largest was about 3.4 cm in diameter.
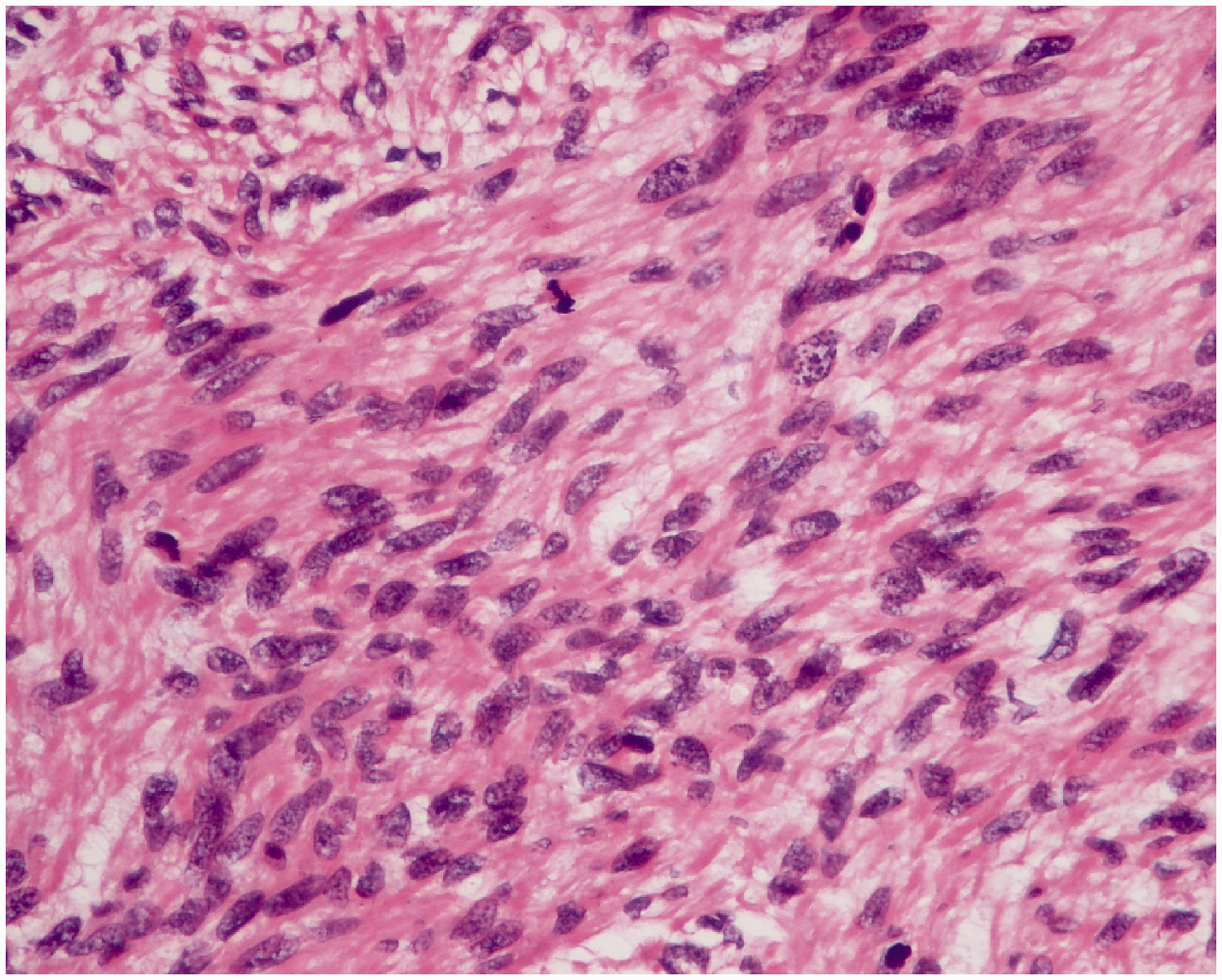
Uterine leiomyosarcoma cells with severe atypia and mitotic activity ≥10/10 high-power fields (hematoxylin and eosin × 200).
Considering the difficulty of reoperation, HIFU combined with gemcitabine 1 g and docetaxel 90 mg chemotherapy was initiated in April 2016. HIFU was administered using an FEPBY02 HIFU system (Yuande Biomedical Engineering Co. Ltd, Beijing, People’s Republic of China) with the following parameters: ultrasonic power 160250 W; emission time of T1 990 ms; interval time of T2 10 ms; 40 transmissions per therapeutic point with a distance of 2 mm between adjacent therapeutic points; and three therapeutic points comprised one unit with 10-s intervals between each unit. Real-time ultrasound monitoring during therapy was carried out using a GE Logiq 400CL (GE Healthcare Biosciences, Piscataway, NJ, USA). The total emission lasted 39 minutes and 36 s and the whole therapeutic schedule lasted 55 minutes and 31 s. We assessed the patient’s condition during treatment using MRI. After three cycles, pelvic MRI showed that the largest pelvic nodule had reduced in size. HIFU combined with chemotherapy was continued for a further two cycles. The tumor size (September 2016) remained constant (compared with August 2016), but the center of the tumor developed necrosis (Figure 2a and b). The patient was subsequently treated with six courses of HIFU alone, and the necrotic area of the mass enlarged (Figure 2c). However, the pelvic mass had increased in May 2017 (95 × 80 × 66 mm compared with 84 × 45 mm) and the lymph nodes adjacent to the right femoral artery were enlarged on MRI. Scattered proliferative foci (largest about 5 mm) were also detected in the middle lobe of the right lung on high-resolution CT. The enlarged mass and suspected lung metastasis indicated that the disease had progressed. The patient was therefore treated with four cycles of doxorubicin, ifosfamide, and mesna. However, the mass continued to enlarge (96 × 89 × 75 mm in October 2017 compared with 95 × 78 × 66 mm in August 2017). The patient was therefore given a further six courses of HIFU from July 2018, which slowed the growth of the tumor and brought the disease under control. However, the patient unfortunately died as a result of her poor physical condition in November 2018. The patient’s survival time from the diagnosis of recurrent uterine leiomyosarcoma in February 2015 was 63 months, which was longer than the median survival time for patients with uterine leiomyosarcoma, suggesting beneficial effects of HIFU combined with chemotherapy for recurrent uterine leiomyosarcoma (Figure 3).
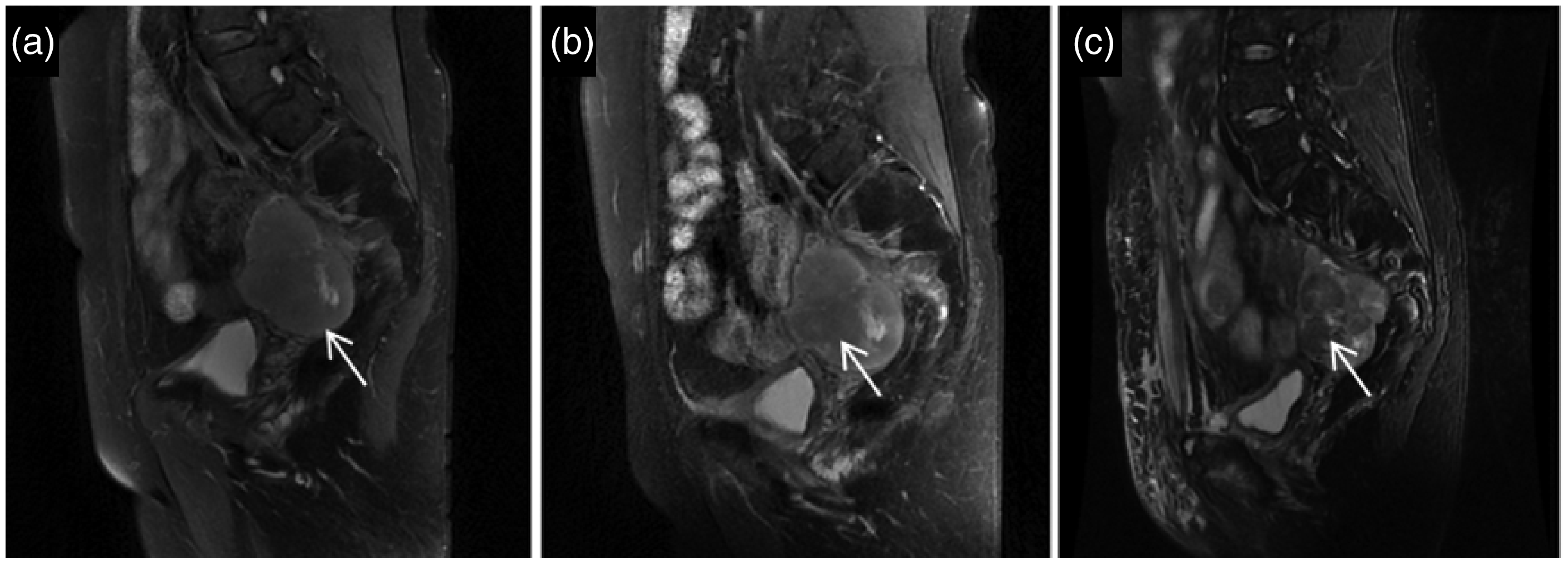
Pelvic magnetic resonance imaging. (a) Pelvic mass about 6.7 × 4.5 × 6.7 cm in sagittal T2 image before high-intensity focused ultrasound (HIFU) treatment. (b) Necrosis in the center of the mass with low signal in sagittal T2 image after HIFU combined with chemotherapy for five cycles. (c) The necrotic area was increased compared with the previous sagittal T2 image after six courses of HIFU alone.

Time–event axis. The patient survived for >5 years, which was considerably longer than the median survival time (10 months). HIFU, high-intensity focused ultrasound.
This study was approved by the Ethics Committee for Human Research of The Second Affiliated Hospital, Zhejiang University School of Medicine. Written informed consent was obtained from the patient’s relatives for publication of this case report and accompanying images.
Discussion
Uterine sarcomas are rare malignant tumors of the female reproductive system derived from uterine smooth muscle, endometrial stroma, and connective tissue, accounting for 2% to 6% of all malignant uterine tumors. Pathologically, uterine sarcomas comprise uterine leiomyosarcoma, endometrial stromal sarcoma, mixed epithelial and stromal tumors, and other sarcomas. Uterine leiomyoscarcoma is highly malignant with a poor prognosis and a recurrence rate of 53% to 71%, a 5-year survival rate of 15% to 25%, and a median survival time of only 10 months. 1 The diagnosis of uterine leiomyosarcoma is often difficult because of its non-specific clinical manifestations. Its diagnosis thus relies on histopathological criteria, including abundant leiomyoma cells, mild atypia with mitotic activity ≥10/10 high-power fields or moderate and severe atypia with mitotic activity ≥5/10 high-power fields, or invasion of muscular vessels, pathological mitotic figures, and coagulation necrosis. Uterine leiomyosarcoma is characterized by early metastasis and local recurrence, with the lung being the most common site of metastasis (74%), followed by the peritoneum (41%), bone (33%), and liver (27%). 2 The main treatment option for uterine leiomyosarcoma is surgery, supplemented by chemotherapy, radiotherapy, and hormone therapy, with personalized treatment required according to the pathological type and stage. However, surgery remains the main treatment for recurrent single uterine leiomyosarcoma. 3 Isolated metastatic foci are treated by surgical excision, followed by radiotherapy to the local recurrence site, and palliative systemic chemotherapy or hormone therapy. 4 However, leiomyosarcomas are not sensitive to radiotherapy and chemotherapy, and their treatment thus represents a considerable challenge.
HIFU is a new non-invasive thermotherapy with the advantages of high repeatability, uniform thermal diffusion, and conformal treatment based on the actual size and shape of the tumor. HIFU is mainly monitored by ultrasound (USg HIFU) and magnetic resonance (MRg HIFU) to orientate the lesions and evaluate its therapeutic effects. USg HIFU monitors the echo changes and regulates the ultrasonic energy release through real-time ultrasound to achieve safe and effective target tissue thermal ablation, while MRg HIFU can monitor temperature changes in the target area and surrounding tissue in real time to adjust ultrasonic power and energy irradiation. 5
Tumor cells are more sensitive to high temperature than normal cells. 6 HIFU increases blood flow to the target tissue and enhances the permeability of the tumor cell membrane via its thermal effects, thus accelerating the penetration and absorption of chemotherapeutic drugs. 7 At the same time, HIFU changes the molecular structure as a result of acoustic cavitation, radiometric force, shear stress, and acoustic stream/micro-flow, which can promote the uptake of the drug and the sensitivity of tumor cells to chemotherapeutic drugs. 8 HIFU combined with chemotherapy can therefore kill cells with a low blood supply and cells in the quiescent period, thus improving the effects of chemotherapy. 9 Paparel et al. 10 found that the Dunning tumor volume doubling time was significantly prolonged and the complete remission rate was about 30% in rats treated with a combination of HIFU and docetaxel compared with 0% in the control group. HIFU can destroy small blood vessels and capillaries, causing embolism of tumor blood vessels and irreversible injury of vascular endothelial cells. 11 Subcutaneous ovarian cancer tissue showed complete coagulation necrosis following HIFU in nude mice, due to the destruction of nourishing blood vessels and capillary networks in the tumor. 12
HIFU has achieved good clinical effects in patients with solid tumors such as liver, breast, and pancreatic cancers.13–15 Abdominal distension, abdominal pain, jaundice symptoms, anorexia, and ascites were all significantly relieved in patients with liver cancer after 3 months of HIFU, compared with before treatment, and CD4 and CD8 levels were significantly increased and immunity was improved after HIFU. 13 In a randomized clinical trial, the HIFU-treated breast cancer region showed full coagulative necrosis and blocked tumor vasculature. 16 HIFU reduced the tumor size and tumor blood flow in breast cancer patients undergoing neoadjuvant chemotherapy compared with the control group, and greatly improved the success rate of breast-conserving treatment. 17 HIFU for 7 to 9 days before radical surgery significantly improved the resection rate and R0 resection rate, and reduced the difficulty and risk of surgery in 30 patients with borderline resectable pancreatic cancer (resectable and locally advanced unresectable pancreatic cancer). 18 Low-energy pulsed HIFU within 24 hours of gemcitabine administration increased the survival time to above the 95% confidence interval in patients with unresectable pancreatic cancer, as well as reducing cancer pain by destroying the peripancreatic nerve and celiac plexus. 19 In relation to the female reproductive system, HIFU has been approved by the United States Food and Drug Administration for the treatment of uterine leiomyoma. 20 The diameter and volume of uterine leiomyomas were significantly reduced after HIFU treatment,21,22 and a non-perfused volume ≥90% was attainable in MRI-guided HIFU treatment for both uterine leiomyomas and adenomyosis, with no trade-off in patient safety. 23 Dysmenorrhea was relieved in patients with adenomyosis and obvious nonperfused areas were detected in the lesion by contrast-enhanced MRI after HIFU. 24
There have been no previous reports of HIFU for the treatment of uterine leiomyosarcoma. In the current case, the tumor relapsed quickly after the initial diagnosis of uterine leiomyosarcoma, and the disease progressed after repeated surgery, radiotherapy, and chemotherapy. Re-operation was difficult because of distant metastasis and the high risk of postoperative recurrence. We therefore administered HIFU combined with chemotherapy. HIFU resulted in liquefactive necrosis in the center of the tumor, leading to slowed tumor growth and prolonged patient survival. The patient received 17 courses of HIFU at approximately monthly intervals. She survived for >5 years, which was considerably longer than the median survival time of 10 months. 1 We therefore conclude that HIFU combined with chemotherapy was beneficial for the treatment of recurrent uterine leiomyosarcoma.
HIFU is accepted by clinicians and scientists as a safe and effective innovation, with great potential for tumor therapy. HIFU not only has direct effects on the tumor, but also has a synergistic effect with chemotherapy. In terms of the comprehensive treatment of tumors, HIFU has achieved good clinical results in combination with surgery, chemotherapy, and radiotherapy, thus providing a useful new treatment strategy. The current study is not intended to provide guidelines for the treatment of uterine leiomyosarcoma, but rather to indicate a new treatment option. However, further studies are needed to establish the optimal dosimetry, non-invasive deep precise temperature measurement, and time intervals for this treatment.
Footnotes
Acknowledgements
We are grateful for the support of Lirong Chen, Director of Pathology, at The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
