Abstract
Objective
This study aimed to identify Helicobacter pylori virulence factors and examine their associations with clinical outcomes in Thai patients. Moreover, the association between these genotypes and gastric mucosa morphological patterns was investigated.
Methods
This retrospective study enrolled patients who underwent esophagogastroduodenoscopy at Suranaree University of Technology Hospital. The presence of the cagA and vacA genes was investigated by real-time polymerase chain reaction.
Results
The H. pylori-specific genes ureA and 16S rRNA were detected in all 698 gastric biopsy specimens. In total, 567 (81.23%) patients with H. pylori infection were positive for the cagA gene, 443 (63.46%) were positive for the vacA gene, and 370 (53.0%) were positive for both. The cagA genotype was significantly more common in patients with chronic gastritis and peptic ulcers (78.99% and 79.41%, respectively) than the vacA gene (51.48% and 55.88%, respectively) and combined genotypes (32.34% and 47.05%, respectively). Moreover, the cagA genotype was significantly more common in patients with type 4 or 5 gastric mucosa patterns (69.49% and 76.31%, respectively).
Conclusions
The cagA genotype is the main cause of serious inflammation of the gastric mucosa. The cagA gene is possibly an important factor explaining gastroduodenal disease outcomes in Thai patients with H. pylori infection.
Keywords
Introduction
Helicobacter pylori is a gram-negative bacterium that can colonize the human stomach. Infection by this bacterium is observed in approximately 25% of the population in developed countries, 70% to 90% of people in developing countries, and more than 50% of people in East Asia, particularly in Japan and China.1–3 In Thailand, the prevalence of H. pylori infection is high in the northeastern region. 4 Chronic infection by H. pylori is related to a wide variety of gastric mucosa damage ranging from chronic gastritis and gastric ulcer disease to gastric cancer and mucosa-associated lymphoid tissue (MALT) lymphoma.5–7 The varieties of clinical outcomes of H. pylori infection are more likely attributable to variations in the virulence of individual strains. Several virulence factors have been described thus far. However, maculating cytotoxin A (vacA) and cytotoxin-associated antigen (cagA) play major roles in the clinical symptoms of H. pylori infection.8,9 The cagA gene was proposed as a marker for the pathogenic island genome and it is an oncoprotein. 10 There is strong evidence that the cagA gene suppresses phagocytosis, reduces antimicrobial peptide levels, blocks the effector T-cell response, and induces tolerogenic dendritic cells. 11 A meta-analysis of a case–control study revealed that cagA-positive strains increase the risk that gastric precancerous lesions develop into gastric cancer. 12 Another virulence factor, vacA, was reported to be important in the induction of vacuoles and apoptosis in the gastric epithelium. 9 The vacA structure consists of two important regions: the signal region (s1 and s2) and the mid-region (m1 and m2). 13 Several studies suggested that cagA and vacA are useful markers of most virulent strains of H. pylori related to severe gastroduodenal disease.14–16 Although efforts in the past decade have been exerted to determine the association between the development of a specific disease and differences in the genotypes of H. pylori strains, the reports have not been conclusive. Therefore, the discovery of a marker for identifying patients at higher risk of gastroduodenal disease would be helpful for clinical practice. This study examined the association between the presence of the cagA and vacA genes of H. pylori and gastroduodenal diseases. Moreover, the association between these genotypes and the morphological patterns of the gastric mucosa was also investigated.
Materials and methods
Patients and specimens
Specimens were received from patients with H. pylori infection-associated diseases who underwent esophagogastroduodenoscopy (EGD) at Suranaree University of Technology Hospital (Nakhon Ratchasima, Thailand) from July 2021 to June 2022. The EGD procedures were conducted using an EVIS EXERA III (CV-190) endoscope (Olympus Corp., Tokyo, Japan). The whole stomach was reviewed by conventional gastrointestinal endoscopy. Then, gastric biopsy specimens were obtained using the site-specific biopsy method. 17 Biopsy samples from the antrum and corpus were obtained for the rapid urease test, and only test-positive samples were used to extract DNA for real-time polymerase chain reaction (PCR). Biopsy samples for histological examinations were subsequently selected by the pathologist. Giemsa staining (Thermo Fisher Scientific, Waltham, MA, USA) and hematoxylin and eosin (H&E, Thermo Fisher Scientific) staining were applied for H. pylori detection. All specimens were stored at −20°C until further evaluation by molecular techniques. Patients signed a written informed consent form prior to enrollment. The study methods were approved by the Ethics Committee for Research Involving Human Subjects, Suranaree University of Technology (approval number: EC63-39). Moreover, this study followed relevant STROBE guidelines, 18 and all patient details were de-identified.
DNA extraction
The extracted genomic DNA from gastric biopsies was obtained using a QIAamp DNA mini kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s guidelines. The concentration of the extracted DNA was investigated using a DS-11þ spectrophotometer (Denovix Inc., Wilmington, DE, USA), and the extracted DNA was stored at −20°C.
Real-time PCR
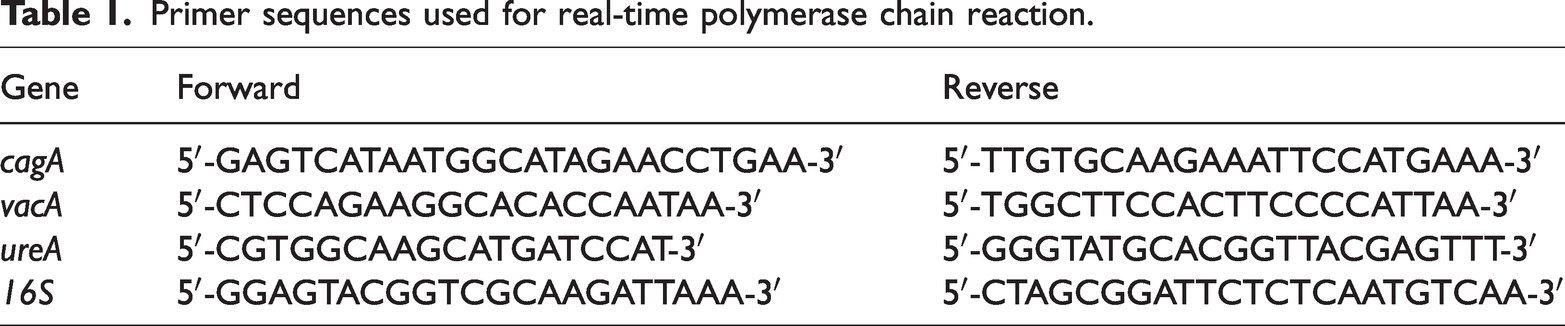
To investigate H. pylori infection, the presence of the 16S rRNA and ureA genes was examined. The H. pylori-positive cases were used to assess the presence of the cagA and vacA genes using real-time PCR. The primers for 16S rRNA, ureA, cagA, and vacA (Integrated DNA Technologies Inc., Coralville, IA, USA) are presented in Table 1. The extracted DNA from biopsy specimens was applied as templates for PCR. Real-time PCR was conducted according to the manufacturer’s instructions in a final volume of 20 µl consisting of the cDNA template, 5 pmol of each primer, and 2 × SYBR Green PCR Master Mix (Roche AG, Basel, Switzerland) using a Light Cycler VR 480 Instrument (Roche). The PCR conditions were as follows: initial denaturation at 95°C for 10 minutes followed by 40 cycles of denaturation at 95°C for 15 s and annealing at 60°C for 1 minute. The data were evaluated using Light Cycler 480 software, version 1.5 (Roche).
Primer sequences used for real-time polymerase chain reaction.
Evaluation and classification of gastric mucosa morphological patterns
All endoscopic investigations were recorded, and images of the examination site were captured. The gastric mucosa morphological patterns were evaluated by three endoscopists as described by Anagnostopoulos et al. 19 with some modifications. The gastric mucosa patterns were classified into five types as follows: type 1, normal gastric mucosa indicated by a honeycomb-type subepithelial capillary network pattern with a regular arrangement of collecting venules; type 2, mild chronic gastritis indicated by cone-shaped gastric pits; type 3, moderate-to-severe chronic gastritis indicated by rod-shaped gastric pits with sulci; type 4, ground-glass appearance; and type 5, dark brown patches with irregular borders and bluish margins. Types 1 and 2 indicate normal-to-mild inflammation and morphological features, and types 3 to 5 indicate moderate-to-severe inflammation with atypical morphological features. 20 Before reviewing the slides, the endoscopists were blinded to the results of H. pylori infection.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 20.0 (IBM Corp., Armonk, NY, USA). Comparisons between groups were performed using the χ2 and Pearson correlation tests. P < 0.05 was considered statistically significant.
Results
H. pylori virulence factors and clinical outcomes
The 16S rRNA and ureA genes were detected in all 698 (100%) gastric biopsy cases. In total, 567 (81.23%) H. pylori-positive patients were positive for the cagA gene, 443 (63.46%) were positive for the vacA gene, and 370 (53.0%) were positive for both genes. Among infected patients, 538 (77.07%) had chronic gastritis, 103 (14.75%) had duodenitis, 34 (4.87%) had peptic ulcers, 13 (1.86%) had duodenal ulcers, and 10 (1.43%) had gastric cancer. The cagA genotype was significantly more common in patients with chronic gastritis and peptic ulcer (78.99% and 79.41%, respectively; P = 0.024 and 0.038, respectively), whereas the presence of vacA and the dual positive genotype had no significant association with clinical outcomes (Table 2).
Prevalence of Helicobacter pylori virulence factors in gastroduodenal diseases. The virulence genes and each clinical outcome were compared with those in chronic gastritis.

*, P < 0.05.
H. pylori virulence factors and the severity of chronic gastritis
The 698 patients included 441 (81.97%) patients with the type 2 or 3 pattern, 59 (10.96%) patients with a type 4 pattern, and 38 (7.06%) patients with a type 5 pattern. cagA was significantly more common in patients with the type 4 (69.49%; P = 0.041) and type 5 (76.31%; P = 0.035) patterns. However, no significant associations were established between the vacA gene or combination genotypes and gastric mucosa pathological patterns (Table 3).
Prevalence of Helicobacter pylori virulence factors according to the severity of chronic gastritis. The virulence genes and each gastric mucosa morphological pattern type were compared with those in gastric mucosa morphological pattern types 2–3.

*, P < 0.05.
Discussion
H. pylori virulence factors and the inflammatory response of the host have been reported to be involved in gastric lesion development induced by the bacterium. Persistent infection by H. pylori causes the development of severe complications such as chronic gastritis, peptic ulcer, duodenal ulcer, gastric MALT lymphoma, and gastric cancer.5–7 Many studies have examined the effect of H. pylori virulence genes on clinical symptoms, whereas data about the effects of virulence genes on gastrointestinal diseases remain limited. Two main pathogenic factors have been identified in H. pylori, namely the cagA and vacA genes.8,9 The presence of these two virulence genes has been associated with a more aggressive phenotype of H. pylori strains, and these genes have been linked to severe disease outcomes.14–16 This study determined the prevalence of the cagA and vacA genes and combination genotypes in Thai patients with H. pylori-associated gastroduodenal diseases. Our study demonstrated that the presence of the cagA gene was associated with chronic gastritis and peptic ulcers. Meanwhile, the vacA gene and the combination genotype were not associated with any clinical outcomes. Moreover, this is the first report to evaluate the association between H. pylori virulence factors and gastric mucosa morphological patterns in gastroduodenal diseases. We revealed that the cagA gene was associated with type 4 and type 5 gastric mucosa patterns. These findings suggest that the cagA gene is an influential marker for chronic gastritis and peptic ulcer development, and it might be involved in the severity of the disease.
The prevalence of the cagA gene varies in different geographical regions. In Western countries, the prevalence of the cagA gene is 60% to 70%, 21 but the gene was found in more than 80% to 90% of patients in East Asian countries. 22 It has been demonstrated that most H. pylori strains in Thailand carry the cagA gene. 23 Our results are supportive of previous findings that Thai strains predominantly carried the cagA genotype. In addition, the prevalence of cagA-positive strains in this study was similar to that reported in the northeastern region of Thailand 24 and in other Asian countries, in which the prevalence rate of cagA-positive strains exceeded 80%. Moreover, this study revealed that H. pylori strains harboring the cagA gene are associated with chronic gastritis and peptic ulcers. The results were consistent with a previous report of Brazilian clinical isolates that found a significant association between cagA and peptic ulcers. 25 Therefore, the cagA gene might be an influential marker for identifying patients at higher risk of gastritis and peptic ulcer development.
The rate of vacA genotype positivity in this study was 63%, which corresponded to the findings of a cross-sectional study in northeastern Thailand. 24 Conflicting results have been obtained in studies on the association between the vacA gene and gastroduodenal disease in different geographical regions. Our study revealed no association between vacA positivity and gastroduodenal disease, in accordance with previous findings,26–28 suggesting that the vacA genotype is not a good marker of clinical outcomes in H. pylori infection. However, other studies recorded an association between gastroduodenal disease (e.g., atrophic gastritis, peptic ulcer) and the vacA gene in H. pylori-infected patients. 29 Although the presence of both genes is often observed simultaneously, which increases the pathogenicity of the strains, we found that combination genotypes did not affect the clinical outcomes of H. pylori-infected patients in Thailand. One important reason for the difference might be geographical differences in H. pylori strains. Several studies demonstrated that cagA-positive H. pylori strains are involved in gastroduodenal disease severity.8,30 Our results also indicated that H. pylori strains harboring the cagA gene were associated with inflammatory changes in the gastric mucosa, suggesting that the cagA gene could be useful in indicating severe disease in H. pylori-infected patients. Although this study included a large number of subjects, the number of patients in some groups (peptic ulcer, duodenal ulcer, and gastric cancer) remained small, and larger numbers of cases are needed.
Conclusions
The cagA gene is the major virulence factor present in H. pylori, and it is associated with severe gastroduodenal pathology that includes both chronic gastritis and peptic ulcer disease. The presence of this gene can be used as an indicator of the severity of the disease and risk of progression of gastric mucosa damage in Thai patients.
Footnotes
Author contributions
Conception and design: T. Tongtawee, W. Wattanawongdon
Acquisition of data: T. Tongtawee, W. Wattanawongdon, T.S. Bartpho
Analysis and interpretation of data: T. Tongtawee, W. Wattanawongdon, T.S. Bartpho
Writing and revision of the manuscript: T. Tongtawee, W. Wattanawongdon
Declaration of conflicting interests
The authors declare no conflict of interest regarding the publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Suranaree University of Technology, Thailand Science Research and Innovation, and the National Science, Research and Innovation Fund.
