Abstract
Congenital systemic candidiasis is a rare disease observed in both full-term and preterm infants. It can occur with or without congenital cutaneous candidiasis (CCC) and to date, only a few cases have been reported in the literature. We report here, a case of a full-term newborn who presented with diffuse skin eruptions at birth. Blood, urine, and skin scraping cultures were positive and the aetiological agent was Candida albicans. After six weeks of anti-fungal treatment with fluconazole, the newborn was cured. Early diagnosis is crucial in preventing complications caused by candidiasis in newborns.
Background
Unlike hospital-acquired systemic candidiasis, which is common in neonatal intensive care units worldwide, congenital systemic candidiasis is extremely rare and can occur in full-term and preterm infants.1,2 A meta-analysis conducted in 2020 found that fewer than 50 recognized cases had been reported in the literature over the past 54 years. 2 The disease may be localized (i.e., congenital cutaneous candidiasis, CCC) and present as an extensive skin rash which eventually results in widespread desquamation, or generalized (i.e., congenital systemic candidiasis) which usually lacks cutaneous involvement and presents with respiratory distress, meningitis, sepsis, and death.2,3 CCC has been estimated to occur in 0.1% of neonatal intensive care unit (NICU) admissions, with most cases occurring in preterm infants <37 weeks gestation. 4 We describe here, a case of a full-term neonate with congenital systemic candidiasis and CCC. The infant developed candidemia due to Candida albicans and the same yeast was isolated in blood, urine and skin cultures.
Case report
A female infant weighing 3.28 kg was born at 40 weeks gestation to a healthy 24-year-old woman with a history of two abortions; this was her third pregnancy. The baby was delivered by caesarean section because the mother had scoliosis, right hydronephrosis, active stagnation, and suspected intrauterine infection. The mother’s prenatal screening tests (i.e., gonorrhoea, chlamydia, syphilis, rubella, group B streptococcus, and complete blood count) were unremarkable and there was no evidence of C. albicans in her vaginal secretion in a sample taken six months previously. She had not received any antibiotics for a urinary tract infection (UTI) or any other bacterial infection in the previous six months.
Two hours before the birth, the mother developed a fever (38.0°C) after which amniotomy was performed and she was given IV oxytocin 2 mU/min and IV cefuroxime sodium 1.5 g. Her Apgar score was 8/9 at 1 and 5 minutes after birth, and her amniotic fluid was clear without meconium staining. Histopathological examination of the placenta and umbilical cord after birth showed severe acute chorioamnionitis and moderate umbilical vasculitis with extensive neutrophilic infiltration in the chorionic and subchorionic plates of the placenta. There was also extensive cellulose attachment in the villi. The baby was admitted to the NICU because of groaning and spitting.
At birth, the infant presented with a generalized erythematous, papular eruption over the face, trunk and extremities (Figure 1a). Three days later, pustules and desquamation were observed, and the palms and soles were also affected (Figures 1b, c, d, and e). Smears of skin scrapings were negative for Candida spp and the baby did not show presence of oral or perianal thrush. With the exception of transient respiratory distress, which was remedied by low-flow oxygen through a nasal cannula for two days, and a decreased transmittance chest X-ray, the infant’s physical examination was normal.
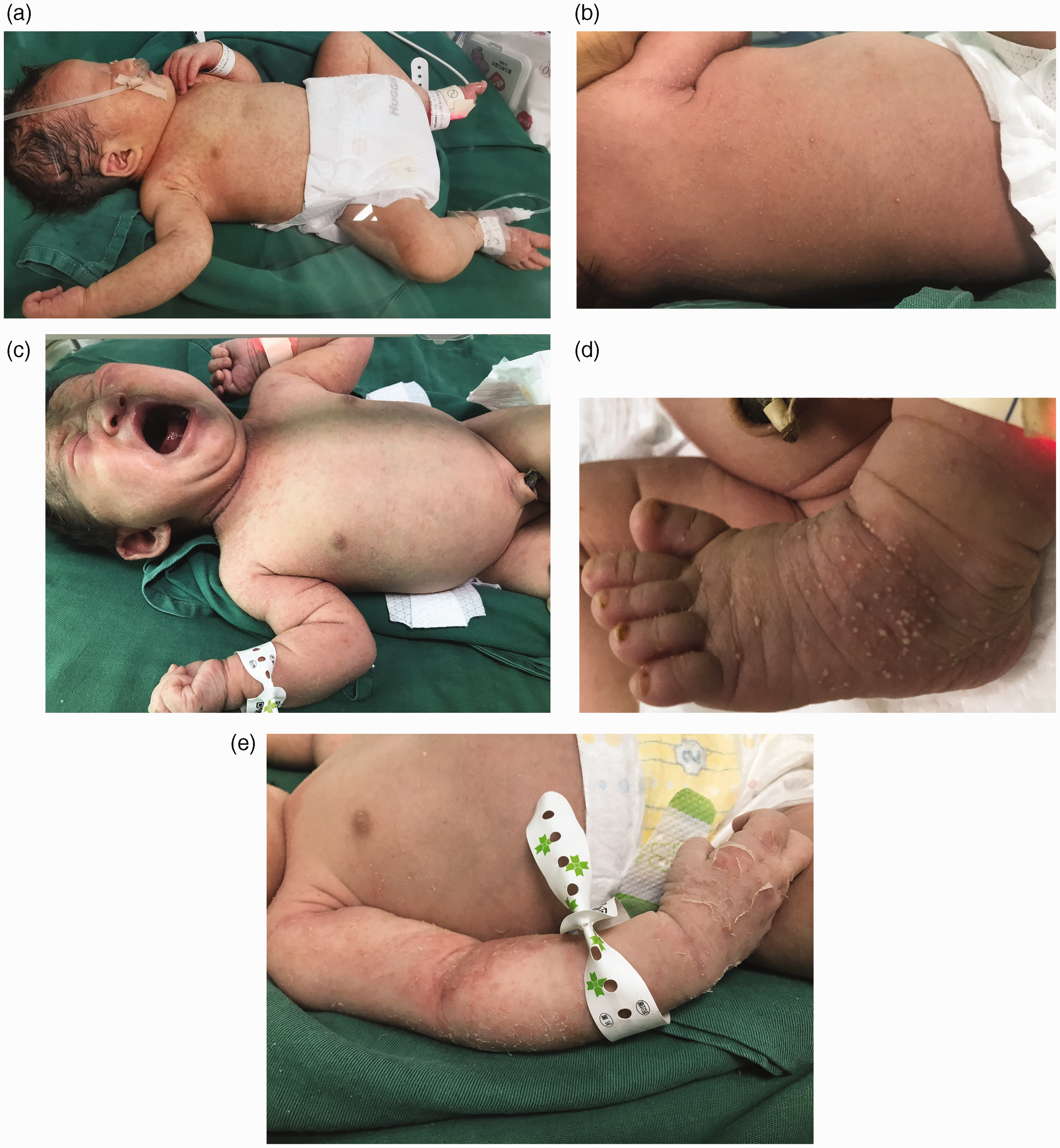
Full-term infant with congenital systemic candidiasis, presenting with generalized erythematous papules (a), pustules and desquamation, and involvement of the palms and soles (b, c, d, and e).
After 18 hr, blood analysis showed an increase in C-reactive protein (CRP) levels (68.2 mg/l) and leucocytosis with left shift, and so a lumbar puncture was performed. Cerebrospinal fluid (CSF) culture was negative, CSF white blood cell (WBC) count was 15 cells/mm3, CSF protein was 63.4 mg/dl and CSF glucose was 2.5 mmol/l. These results excluded a central nervous system (CNS) infection. The newborn was treated with empiric antimicrobial therapy (i.e., cefoperazone/sulbactam (80 mg/kg/d, q12 h) and penicillin (100,000 IU/kg/d, q12 h) for seven days (Figure 2). The differential diagnosis of the rash at birth included: congenital herpes simplex virus (HSV); congenital syphilis; streptococcal infection; Listeria monocytogenes infection; candidiasis.
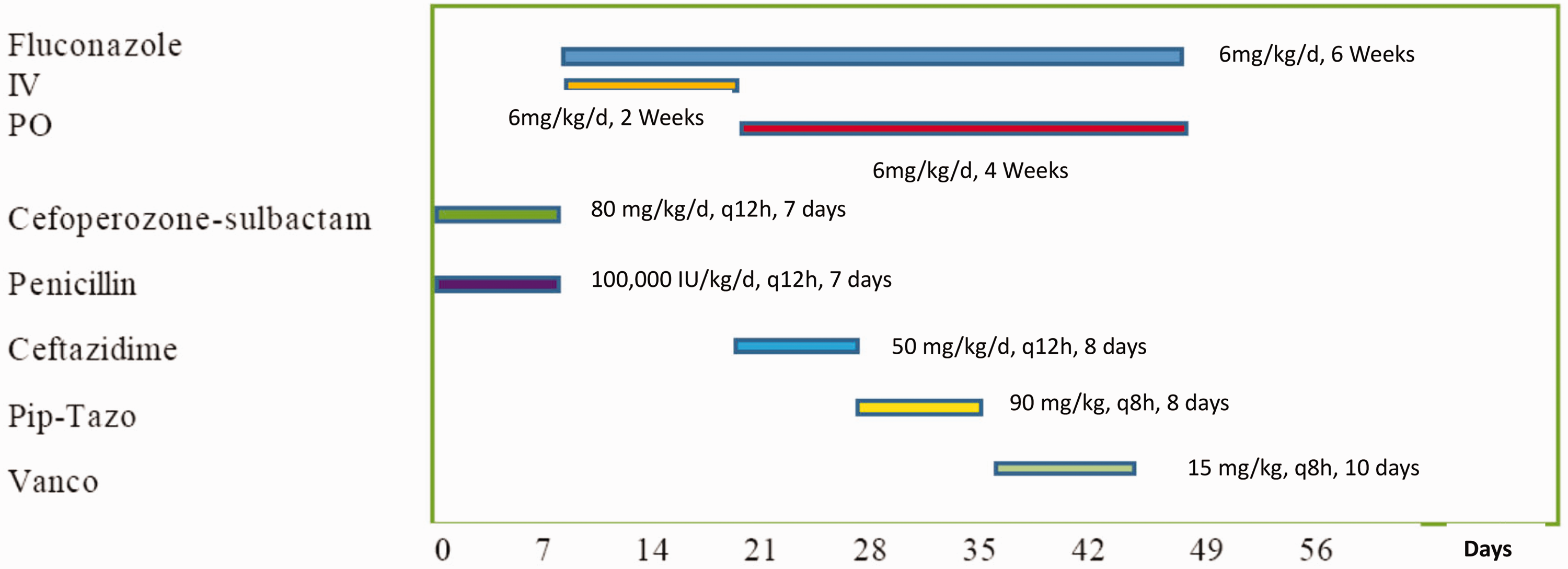
Timeline of antifungal and antibiotic treatment.
On Day 7, the skin rash had desquamated and excoriated lesions persisted. The infant performed well clinically, and her WBC count and CRP levels were within the normal range, so the antibiotics were stopped. However, on the same day we were informed that cultures from blood, urine and skin samples taken on admission were positive for C. albicans. However, the results of a serum (1–3)-β-D-glucan assay were negative. The infant was started on IV fluconazole (6 mg/kg/day) and topical skin antifungal therapy (miconazole nitrate). On Day 13, high-throughput DNA sequencing of blood samples did not show evidence of fungi, bacteria, or viruses and the patient’s urine cultures were negative. On Day 20, the infant showed an increase in acute phase reactants. Sepsis was suspected and the site of the infection was the urinary tract. Ceftazidime (50 mg/kg/d, q12 h) was started and continued for eight days. IV fluconazole was stopped and oral fluconazole (6 mg/kg/day) was initiated for four weeks. On Day 28, due to the persistence of the UTI (Escherichia coli and Enterococcus faecium) the infant required serial treatment with piperacillin/tazobactam (90 mg/kg, q8 h) 8 days. and on Day 35 vancomycin (15 mg/kg, q8 h, total 10 days). was started. Interestingly, her cutaneous lesions had resolved by Day 22.
Due to recurrent bacterial UTIs, we suspected that the baby had immune deficiency. We conducted blood immunity-related genetic analysis and immune function tests (i.e., quantitative immunoglobulin and T cell number and function analysis) which showed normal immune function and excluded chronic granulomatous disease/galactosemia. She was also suspected of having vesicoureteral reflux but voiding cystourethrography was not performed because of difficulties in achieving urine retention in a baby.
Following the 10-day course of IV vancomycin, two repeat urine cultures were sterile, two urine routine examinations were normal, and audiometry was normal. An ophthalmologic examination, brain magnetic resonance imaging (MRI) (Day 16), chest computed tomography (CT) (Day13), liver, renal, and bladder ultrasonography (Day 3) and 99mTc-DMSA static renal scan (Day 49) were normal. An echocardiogram (Day 3) showed a small atrial septal defect and patent foramen ovale (0.19 cm, 0.13 cm) without endocarditis. At two months, the infant was discharged and showed good development.
This case study was approved by Medical Ethics Committee of Beijing Friendship Hospital and signed informed consent was obtained from the patient’s legal guardian before publishing the anonymised data. The reporting of this study conforms to CARE guidelines. 5
Discussion
Congenital systemic candidiasis is extremely rare and caused by intrauterine or intrapartum infection with Candida spp. The condition has a variety of clinical manifestations, from diffuse erythematous rash with or without blisters and pustules to systemic disease involving invasive infections of blood, urine or lungs, and the skin may or may not be affected. 6 CCC typically presents at or within a few days of birth. 4 Typical pustules usually appear on the palms and soles of feet. The lesions vary in form, and different stages are often observed simultaneously. 7 Other clinical features of CCC can include onychomycosis, mucositis, and acute chorionic inflammation with yellow and white spots on the placenta and umbilical cord. 8
In this present case, the newborn had diffuse red macula at birth, and then gradually developed pustules and desquamation even on her palms and soles. Although the results from initial skin scrapings on Day 3 and (1–3)-β-D-glucan assay taken on Day 7 were negative, C. albicans was isolated from blood, urine and skin cultures taken on Day 7. These findings established a diagnosis of congenital systemic candidiasis, with invasive C. albicans and the infant was treated with IV and topical fluconazole.
Blood culture is considered the gold standard for detection of candidiasis, and has high specificity but poor sensitivity. 9 In addition, the small amount of blood used in infantile cultures makes it difficult to isolate the Candida spp. While the serum (1–3)-β-D-glucan assay was developed to overcome the limitations of blood cultures in diagnosing Candida infections, it only has a sensitivity of approximately 60%. 10 High-throughput DNA sequencing (HST) techniques have also been used to detect pathogens in blood and urine samples, thereby increasing accuracy. 11 In this particular case study, results from HTS of blood samples taken on Day 13 did not show evidence of fungi, bacteria, or viruses and the patient’s urine cultures were negative. Interestingly, her skin lesions persisted until Day 22. In addition to urinary tract candidiasis, our infant was also found to have repeated UTIs with different types of bacteria. However, her immune function was normal and her kidney ultrasound and DSMA scan showed normal kidneys. Our patient’s UTI was resolved following sensitive antibiotic treatment.
Untreated invasive Candida infections have been reported to be associated with high mortality rates, from 39% to 94%.2,12 Therefore, early diagnosis and treatment are critical, especially for high-risk infants, such as those who are <25 weeks gestation, have thrombocytopenia, and/or, have been exposed to third-generation cephalosporins or carbapenems within seven days before blood culture. 13 Once candidemia is suspected, empirical antifungal treatment should be considered immediately.2,13 CCC is an invasive infection of the skin, and even in the absence of positive blood, urine or CSF cultures, requires systemic treatment for a minimum of 14 days for both preterm and full-term infants. 4 Prompt treatment should start at the time of rash presentation. Delayed or systemic treatment less than 14 days is associated with dissemination to the blood. 4 Amphotericin B deoxycholate is often chosen as the first line IV treatment for invasive Candida infections because some Candida spp have been reported to be resistant to fluconazole. 2 In addition, the policy of starting with amphotericin is part of the neonatal antifungal stewardship to prevent the emergence of fluconazole resistance. 14 Moreover, in some countries, amphotericin B deoxycholate is less expensive than other antifungal drugs. 15 Since fluconazole has excellent enteral absorption, either IV or enteral formulations are considered suitable systemic antifungal treatment. 4 According to antifungal susceptibility testing, our infant was treated with fluconazole intravenously for two weeks and orally for four weeks (i.e., total treatment course of six weeks).
In conclusion, congenital systemic candidiasis with cutaneous involvement in a full-term infant is rare, and the clinical manifestations differ from those reported in premature infants. Its clinical symptoms are relatively mild, which may be related to immune maturity. However, the skin rash is obvious. Clinicians should be highly vigilant against diffuse erythema and pustules at birth, especially involving the palms and soles.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231158015 - Supplemental material for Congenital candidiasis in a full-term infant: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231158015 for Congenital candidiasis in a full-term infant: A case report by He-Hua Zhao, Zhe- Zhou, Lu Lu, Yong-Ze Zhao, Li-Jun Yang and Ying-Xue Ding in Journal of International Medical Research
Footnotes
Acknowledgements
The authors express their gratitude to all staff at the Department of Paediatrics.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research project received no specific grant support from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
