Abstract
Objective
To propose a treatment approach for primary spontaneous pneumothorax (PSP) in male patients with a smaller incision and less pain.
Methods
We retrospectively studied 29 patients with PSP who underwent areola-port video-assisted thoracoscopic surgery (VATS) and 21 patients who underwent single-port VATS. The areola-port VATS technique was performed as follows. First, an arc incision was made along the lower edge of the areola, and a 5-mm-diameter thoracoscope was placed. The bullae were completely removed, and the absence of air leaks and other bullae was confirmed. A drainage tube was placed in the chest with negative pressure and then quickly pulled out, and the reserved suture line was knotted.
Results
All patients were male, and their mean age was 19.07 ± 2.43 years. The mean intraoperative hemorrhage volume and postoperative pain score were significantly lower in the areola-port than single-port group. The mean operative time and mean postoperative hospital stay were also shorter in the areola-port group, but without statistical significance. The incidence of complications and the 1-year postoperative recurrence rate were 0% in both groups.
Conclusion
Our method is clinically feasible and inexpensive, has a traceless effect, and is especially suitable for adolescents.
Keywords
Introduction
Spontaneous pneumothorax occurs when gas enters the pleural cavity and the pressure within the pleural cavity increases or negative pressure develops. The lung is consequently forced to compress, blocking venous return to the heart and producing different degrees of cardiopulmonary dysfunction. Asphyxia and death can occur in severe cases. Spontaneous pneumothorax is a common thoracic surgical emergency.1,2 Several clinical data studies have shown that the incidence of primary spontaneous pneumothorax (PSP) is higher in male than female patients, especially among adolescents.3–5 Worldwide, 30% to 60% of cases of PSP relapse after conservative treatment. Surgery is the first-choice treatment for PSP because an operation can quickly relieve symptoms and effectively reduce the recurrence rate. 6 With the popularity of thoracoscopic technology, thoracoscopic-assisted bulla resection has become the standard operation for the treatment of PSP.7–9 Many traditional thoracoscopic-assisted bulla resection techniques are currently utilized, such as the single-pore method and porous method 10 ; however, the surgical scars caused by conventional thoracoscopic-assisted bulla resection are obvious and may result in an inferiority complex in some patients, especially adolescents. To improve patients’ postoperative quality of life, our hospital uses thoracoscopic-assisted bulla resection via the areola to treat PSP in male patients. Our hypothesis was that this method can effectively treat PSP with less postoperative pain and less scarring. We retrospectively evaluated patients who underwent thoracoscopic-assisted bulla resection via the areola in our hospital to demonstrate the clinical feasibility of the method.
Methods
We analyzed all patients with pneumothorax admitted to Jining First People’s Hospital from January 2019 to March 2022, among whom 36 patients underwent thoracoscopic-assisted bulla resection via the areola. Among them, we retrospectively studied 29 patients with PSP. The control group comprised 21 patients who underwent single-port thoracoscopic bulla resection in the conventional lateral decubitus position. The reporting of this study conforms to the STROBE guidelines. 11
This study was a retrospective analysis and was eligible for exemption from ethical review. All patients in the study provided written informed consent, and all patients’ details have been de-identified.
All patients underwent thoracoscopic-assisted bulla resection via the areola approach. All patients were male and had unilateral pneumothorax. No patients’ condition was complicated by massive pleural effusion or hemothorax. The inclusion criteria were as follows: confirmation of bullae by preoperative high-resolution computed tomography examination or a history of repeated pneumothorax attacks; no history of thoracotomy on the affected side and no obvious thoracic adhesion on imaging examination; exclusion of tuberculosis, lung cancer, lung infection, and other pulmonary diseases; adequate pulmonary function to tolerate a surgical operation and no other serious complications resulting in an inability to tolerate an operation; and full communication with the patients and their families before surgery with acquisition of written informed consent.
Both groups of patients underwent general anesthesia with double-lumen endotracheal intubation, were placed in the supine position, and were administered contralateral ventilation with pure oxygen. The skin within the surgical field was routinely disinfected, and sterile surgical towels were placed. An arc incision of about 2 cm was made along the lower margin of the areola on the affected side. A 5-mm-diameter thoracoscope and a linear stapler were inserted into the incision to observe the internal conditions and determine the location of the lesion (Figure 1). If the intrathoracic adhesion was severe or the lesion was difficult to remove because of its location, the operative wound was enlarged or the incision was lengthened. We carefully explored the whole lung, especially the apex, and identified the location of all bullae. Next, we completely removed the bullae along the base and, if necessary, removed a small amount of healthy lung tissue to reduce the risk of recurrence (Figure 2). After re-examining the surface of the lung and confirming that no bullae were visible, we injected water into the thoracic cavity. The lungs were then inflated with pure oxygen to determine whether air leakage occurred on the surface of the lung. After confirming the absence of air leakage on the lung surface, we repeatedly rubbed the parietal pleura with gauze to generate an aseptic inflammatory response, thus promoting the formation of adhesion between the parietal pleura and visceral pleura. Next, a 12-French drainage tube was placed in the chest cavity, the lung was completely inflated with pure oxygen, the muscle layer was sutured, and a suture line was reserved. The drainage tube was connected to a negative-pressure aspirator, and negative-pressure suction was continued for about 3 to 5 seconds. The drainage tube was then quickly pulled out, and the reserved suture line was knotted simultaneously. Finally, we placed pressure on the incision for 4 to 5 minutes and closed the skin layer with an absorbable suture line (Figure 3).

An arc-shaped incision about 2 cm long was made along the lower edge of the areola on the affected side.
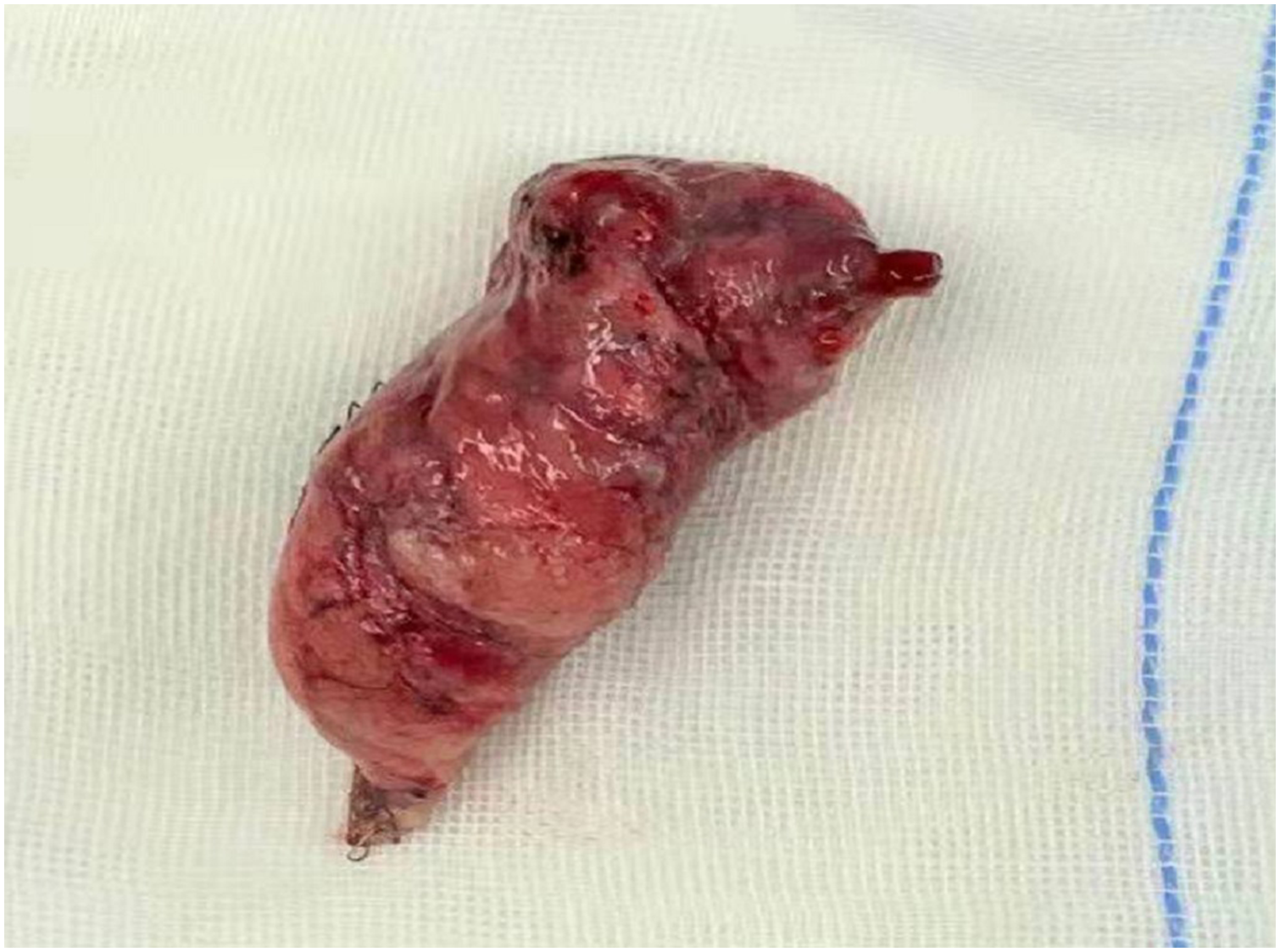
The lung bullae after resection.

Postoperatively, the incision can be hidden in the lower margin of the areola.
All patients underwent X-ray examination on the first postoperative day, and their pulmonary condition was assessed. We divided the patients’ pulmonary conditions into three grades based on the lung compression ratio (R): completely dilated (R ≤ 1%), basically dilated (1% < R ≤ 5%), and incompletely dilated (R > 5%). The patients were able to be discharged when R ≤ 5%, and at least two experienced attending thoracic surgeons were involved in the decision to discharge. If the lung did not expand completely (R > 5%), the patient remained hospitalized for observation. We did not place a chest drainage tube intraoperatively; therefore, if the patient’s symptoms of pneumothorax worsened, a chest tube was placed to relieve the symptoms postoperatively. Both groups of patients underwent pain assessment on the first postoperative day using a numeric rating scale in which 0 indicated no pain, 1 to 3 indicated mild pain, 4 to 6 indicated moderate pain, and 7 to 10 indicated severe pain. Both groups were given the same analgesic therapy after surgery. All patients were followed up 1 year after surgery to identify any recurrence.
Statistical analysis
Perioperative data were recorded for all patients. All
Results
Twenty-nine male patients with unilateral PSP were enrolled in the study, and all patients were successfully treated by thoracoscopic-assisted bulla resection via the areola. The patients’ ages ranged from 16 to 25 years (mean, 19.07 ± 2.43 years). The location of PSP onset was on the left in 17 (58.6%) patients and on the right in 12 (41.4%) patients. All lesions were located in the upper lobe.
All patients in both the study and control groups were able to get out of bed and undergo an X-ray examination on the first postoperative day. All patients’ lung compression status on the first postoperative day is shown in Table 1. The maximum volume of compressed lung among all patients was ≤5%, and that in most patients was ≤1%.
Patients’ lung condition on first postoperative day (n = 29).

Data are presented as n (%). R: lung compression ratio.
Table 2 shows the related postoperative factors in all patients. The mean operative duration in the study group and control group was 23.69 ± 2.26 and 24.00 ± 1.82 minutes, respectively, with no significant difference; the mean intraoperative hemorrhage volume was 2.48 ± 1.91 and 5.71 ± 1.22 mL, respectively (
Related postoperative factors in all patients.

Data are presented as mean ± standard deviation. VATS: video-assisted thoracoscopic surgery.
According to our research results, we strongly believe that thoracoscopic-assisted bulla resection via the areola without a drainage tube can be effective in treating PSP on either the left or right side. In addition, it can significantly reduce patients’ pain.
Discussion
Our study confirms that thoracoscopic-assisted bulla resection via the areola approach can effectively treat PSP in male patients with less intraoperative bleeding, less postoperative pain, and less scarring. PSP is a common emergency in the field of thoracic surgery, and it usually manifests as sudden unilateral or bilateral chest tightness and chest pain accompanied by dyspnea. It is a life-threatening disease in adolescents and adults with weak constitution. 12 At present, the diagnosis of PSP is mainly based on chest X-ray examination. High-resolution computed tomography examination is necessary for patients undergoing surgical treatment because it can help to determine the location of pulmonary bullae. 13 Many studies have shown that 30% to 60% of cases of PSP relapse after conservative treatment. Surgery is considered in cases of recurrent disease or persistent air leakage after chest tube placement, especially with the popularity of video-assisted thoracoscopic surgery (VATS).6,14–16
The operative methods for the treatment of PSP vary. In three-port VATS, an endoscopic hole is established in the seventh or eighth intercostal space along the midaxillary line, the main operating hole is established in the third or fourth intercostal space along the anterior axillary line, and an auxiliary operating hole is established in the sixth or seventh intercostal space along the posterior axillary line. Advantages of this method include its ease of operation and the ability to thoroughly check the surface of the lung tissue intraoperatively. This allows for identification of the maximum number of pulmonary bullae and can help to ensure that all visible pulmonary bullae have been surgically removed. Limitations of the technique include the need for more incisions, more damage to the intercostal nerve, postoperative pain, and surgical scarring, which may induce low self-esteem in some young patients. Single-port VATS was subsequently developed to reduce the surgical trauma and relieve patients’ pain. In this method, one operation hole is established in the third or fourth intercostal space along the anterior axillary line, through which the thoracoscope and all instruments are inserted to enter the chest. Compared with the previous method, single-port VATS in less invasive, produces significantly less postoperative pain, has a shorter length of stay, and provides significantly improved patient satisfaction. 17 Some studies have suggested that fiberoptic bronchoscopy can be used instead of VATS. Although fiberoptic bronchoscopy can further reduce the size of the surgical incision and further relieve patients’ pain, such bronchoscopes are rarely used clinically because of their soft texture and the difficulty controlling their direction intraoperatively.14,18
We propose the use of thoracoscopic-assisted bulla resection via the areola to treat PSP in male patients to further alleviate pain, reduce the psychological impact of the surgical scar, and improve postoperative quality of life. Because the chest is entered through the areola, it may cause great damage to the female breast and even affect the production of breast milk; therefore, it is not suitable for the treatment of PSP in women. We used a 5-mm-diameter thoracoscope and a stapler to enter the thoracic cavity; therefore, an arcuate surgical incision with a length of about 2 cm can ensure a sufficient operating space for the procedure. Analysis of our perioperative data showed that there was no need to place a chest tube after the operation; thus, the patients did not need to bear either the pain of tube placement or the secondary pain of tube removal. This is an additional advantage of this surgical method over the traditional surgical method.8,19,20
Studies have shown that areola-port VATS can significantly reduce pain and intraoperative bleeding compared with single-port VATS. We believe that there are three main reasons for this: the smaller incision, no intraoperative placement of a chest tube, and the relatively fixed location of the incision, which is conducive to the anatomy.
Because the arcuate surgical incision is located below the areola of men, the incision will be more aesthetic after transdermal suture closure with absorbable thread. Moreover, because the areola is darker in color than the surrounding skin, the incision is further hidden postoperatively, contributing to an aesthetic outcome (Figure 1). Surgery is only a part of the overall treatment; we must also pay attention to the patients’ psychological state, especially in adolescents. Because the postoperative scar is difficult to detect using this surgical technique, it is unlikely to cause an inferiority complex and will be more conducive to healthy emotional development.21–23
In this study, we compared areola-port VATS with single-port VATS and found that areola-port VATS was associated with significantly less intraoperative blood loss and postoperative pain. We consider that the reason for less bleeding is that the incision is smaller and the surgical damage is therefore less severe; additionally, the postoperative pain is less severe because there is no need to insert a chest tube. However, if air leakage occurs on the lung surface, pneumothorax will develop. If the patient has no obvious symptoms and the lung compression ratio does not exceed 20%, treatment is not required; otherwise, closed thoracic drainage must be performed. None of the patients in the present study underwent closed thoracic drainage after surgery. We considered that the cause of pneumothorax might be delayed application of pressure after removal of the chest tube, allowing a small amount of gas to enter the chest cavity. All patients were followed up for 1 year, and none developed recurrence.
Our technique has three main limitations. First, it is only applicable to lesions on the surface of the lung and cannot be used to reach lesions deep in the lung. Second, our method is not feasible for patients who have recurrent pneumothorax or severe adhesion in the chest. Third, this was a retrospective study, preventing equal numbers of patients in the two groups. Further study is still needed because of the limited sample size.
Conclusions
Thoracoscopic-assisted bulla resection via the areola approach may be a safe and reliable treatment method for PSP in male patients with significantly reduced intraoperative blood loss and less postoperative pain. In particular, the postoperative scar can be covered by the areola, which can effectively reduce patients’ sense of inferiority. We believe that our method fully complies with today’s minimally invasive surgery concept and the trend of rapid recovery and that it is worthy of application and promotion.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231154394 - Supplemental material for Thoracoscopic treatment of primary spontaneous pneumothorax without a drainage tube in male patients
Supplemental material, sj-pdf-1-imr-10.1177_03000605231154394 for Thoracoscopic treatment of primary spontaneous pneumothorax without a drainage tube in male patients by Honghao Fu, Defeng Jin and Yutao Wei in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Luchang Zhang and Zhaoqing Cui for assisting with the thoracoscopy.
Author contributions
Yutao Wei and Honghao Fu designed the research. Honghao Fu, and Defeng Jin performed the thoracoscopy. Defeng Jin collected the data. Honghao Fu analyzed the data and wrote the paper.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by an Institute of Chest Disease Research Grant from the Institute of Chest Diseases, Jining First People’s Hospital.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
