Abstract
Objective
To investigate whether S-ketamine affects the Surgical Pleth Index (SPI) during video-assisted thoracoscopic surgery.
Methods
Eighty-four patients undergoing video-assisted thoracoscopic lung lobectomy were enrolled. They were randomly assigned to an S-ketamine group (group S) and an equivalent normal saline group (group N). SPI values were recorded; and pain score on a numerical rating scale (NRS), the consumption of opioids, rescue analgesia, and post-operative nausea and vomiting (PONV) were evaluated.
Results
The SPI and heart rate of the S-ketamine group were significantly lower 30 minutes after the start of surgery and at the end. The NRS score was lower in the S-ketamine group 6 and 12 hours postoperatively, but there were no differences in mean blood pressure or the NRS score 24 and 48 hours postoperatively. Rescue analgesia was required less frequently by the S-ketamine group, but the incidence of PONV did not differ between the groups.
Conclusions
S-ketamine was associated with lower intraoperative SPI 30 minutes after the start and at the end of surgery. It also reduced opioid use intraoperatively and the NRS scores 6 and 12 hours postoperatively.
Keywords
Introduction
Patients generally experience severe pain during video-assisted thoracoscopic surgery, 1 and until now there has been no effective way of monitoring this. 2 The Surgical Pleth Index (SPI), calculated using the pulse-wave amplitude and interval between heart beats, can be used to monitor the hemodynamic responses of patients, 3 and this might reflect sympathetic activity. Although the SPI, which has a scale from 0 to 100, reflects surgical stimuli, it is of no value for the assessment of pain in conscious individuals. 4 Furthermore, a previous study showed that both vasopressor use and the requirement for opioids were lower when a combination of Entropy monitoring and SPI guidance of anesthesia was used instead of standard care. 5
A systematic review and meta-analysis revealed that S-ketamine, which provides both analgesia and sedation, could be used to relieve acute postoperative pain.
6
In addition, a study by Mastrodonato
Although thoracoscopic surgery is relatively non-invasive, some patients experience intense pain during this procedure. Therefore, in the clinic, S-ketamine is used by anesthesiologists as an auxiliary analgesic drug during 10 and after surgery. 11 However, it is unclear whether it affects SPI. Therefore, we aimed to determine whether S-ketamine reduces SPI during video-assisted thoracoscopic surgery.
Methods
Ethics approval
The study was approved by the ethics committee of the First Affiliated Hospital of Suzhou University (approval no.: 2020127; Chairs: Professors Fuhai Ji and Qinyun Wang). The study was registered with the Chinese Clinical Trial Registry (ChiCTR2000040012, date of registration: 18/11/2020). Written informed consent was obtained from all the participants and the trial was performed in accordance with the CONSORT guidelines, 2010.
Study design
The inclusion criteria were as follows: planned thoracoscopic pulmonary surgery under general anesthesia, including wedge resection, segmentectomy, partial pneumonectomy, lobectomy, and unilateral radical resection of lung cancer; American Society of Anesthesiologists (ASA) classification I to II; age 18 to 65 years; body mass index (BMI) 19 to 29 kg/m2; and planned stay in hospital ≥48 hours. The exclusion criteria were as follows: ASA ≥III; allergy to S-ketamine, history of mental illness or chronic pain; communication disorder; emergency or trauma surgery; unscheduled visit to the intensive care unit; and refusal to participate in the study. Patients were randomly allocated to either an S-ketamine group (group S) or a normal saline group (group N).
Randomization and assigned groups
Randomization was performed using computer-generated random numbers and the participant allocation ratio was 1:1. One hour before the induction of anesthesia, the group assignments were concealed in sequentially numbered opaque envelopes. Participants allocated to group S were administered 0.1 mg/kg S-ketamine during the induction of anesthesia, followed by a continuous infusion of 0.1 mg/kg/hour until the end of surgery. Participants allocated to group N were administered the same volume of normal saline (0.9%) during the induction of anesthesia, and this was continuously infused at the same rate until the end of surgery.
All the participating patients, researchers, clinicians, and nurses were blinded to their treatment allocation. The test solutions were prepared by an anesthesiologist who was not involved in the study.
Anesthesia and analgesia protocol
After entering the operating room, patients were monitored using invasive blood pressure (BP) monitoring (radial arterial BP), electrocardiography, pulse oximetry, and end-tidal CO2 monitoring. The SPI was measured using a Datex-Ohmeda S/5 ADU (GE Healthcare, Madison, WI, USA) monitoring system. The type of double-lumen bronchial intubation was selected according to the surgical site and the thickness of the participant’s trachea.
Anesthesia was induced using intravenous midazolam (1 to 3 mg), sufentanil (0.3 to 0.6 μg/kg), propofol (2.0 to 3 mg/kg), and cisatracurium (0.2 mg/kg). The total amount of sufentanil administered was 0.4 to 1 μg/kg, according to the nature of the surgical procedure. Endobronchial intubation was performed once the injectable agents had taken effect. Anesthesia was maintained with 1% to 3% sevoflurane or propofol (4 to 6 mg/kg/hour), remifentanil (0.01 to 0.1 μg/kg/minute), and cisatracurium (0.05 to 0.1 mg/kg/hour); at a bispectral index (BIS) of 40 to 60. The settings were: one lung ventilation of 6 to 8 mL/kg tidal volume for the ideal body mass, respiratory rate of 12 to 16 breaths/minute, positive end-expiratory pressure of 3 to 5 mmHg, the lowest possible oxygen concentration to maintain an SpO2 of >90%, and an intraoperative end-tidal carbon dioxide of 35 to 45 mmHg. Before starting the surgery, 5 mL of 0.375% ropivacaine was injected at each incision site.
All the participants were transferred to the Post-anesthesia Care Unit after the surgical procedure had been completed. The postoperative analgesia protocol consisted of patient-controlled analgesia (PCA) using sufentanil, ondansetron, and 30 mg of intravenous ketorolac tromethamine every 12 hours. The PCA device was set to deliver a background infusion of 2 μg/hour sufentanil. If the pain score was >3 on the NRS, an additional 50 mg of flurbiprofen axetil was administered as rescue analgesia.
Outcomes
The primary outcomes were the SPI when the participants entered the operating room (baseline), at intubation, at the beginning of the surgery (skin incision), 30 minutes after the start of surgery, and at the end of the surgery.
The secondary outcomes were the NRS scores 6, 12, 24, and 48 hours postoperatively, the durations of surgery and anesthesia, the amount of sufentanil used, the time to extubation, hemodynamic parameters (including mean blood pressure (MBP) and heart rate (HR), the need for rescue analgesia, the requirement for flurbiprofen axetil, and post-operative nausea and vomiting (PONV). The intensity of pain following surgery was estimated on the ward using a 10-cm NRS (0 for no pain and 10 for the most severe pain imaginable).
Statistical analysis
We performed a power analysis using Prism 9.0 (GraphPad, La Jolla, CA, USA), with a β value of 0.1 and an α value of 0.05. In a preliminary experiment, we obtained a standard deviation for the SPI 30 minutes after the start of surgery of 11 and a difference between groups S and N of 8. These yielded a required sample size of 40 per group. Therefore, to allow for dropouts, we aimed to recruit 42 patients per group for the present study. If a patient refused to continue their participation in the trial or the surgery could not be completed as expected, they would be excluded.
The data were analyzed using GraphPad Prism 9.0 software. Normally distributed datasets are summarized as the mean ± standard deviation, and comparisons were performed using the unpaired Welch’s
Results
Participant characteristics
We initially enrolled 84 patients. Four of the patients were excluded: in group S, one patient declined further participation during the observation period and one was excluded owing to conversion to thoracotomy; and in group N, two patients were excluded owing to major intra-operative hemorrhage. Therefore, data from 80 participants were studied (n = 40 per group) (Figure 1).

CONSORT diagram of patient recruitment.
There were no differences in baseline characteristics (age, body mass, height, BMI, sex, ASA, smoking, or hypertension) of the two groups (Table 1), and the durations of surgery and anesthesia did not differ. The total amount of sufentanil used was lower in group S (
Baseline characteristics of the two groups of participants.
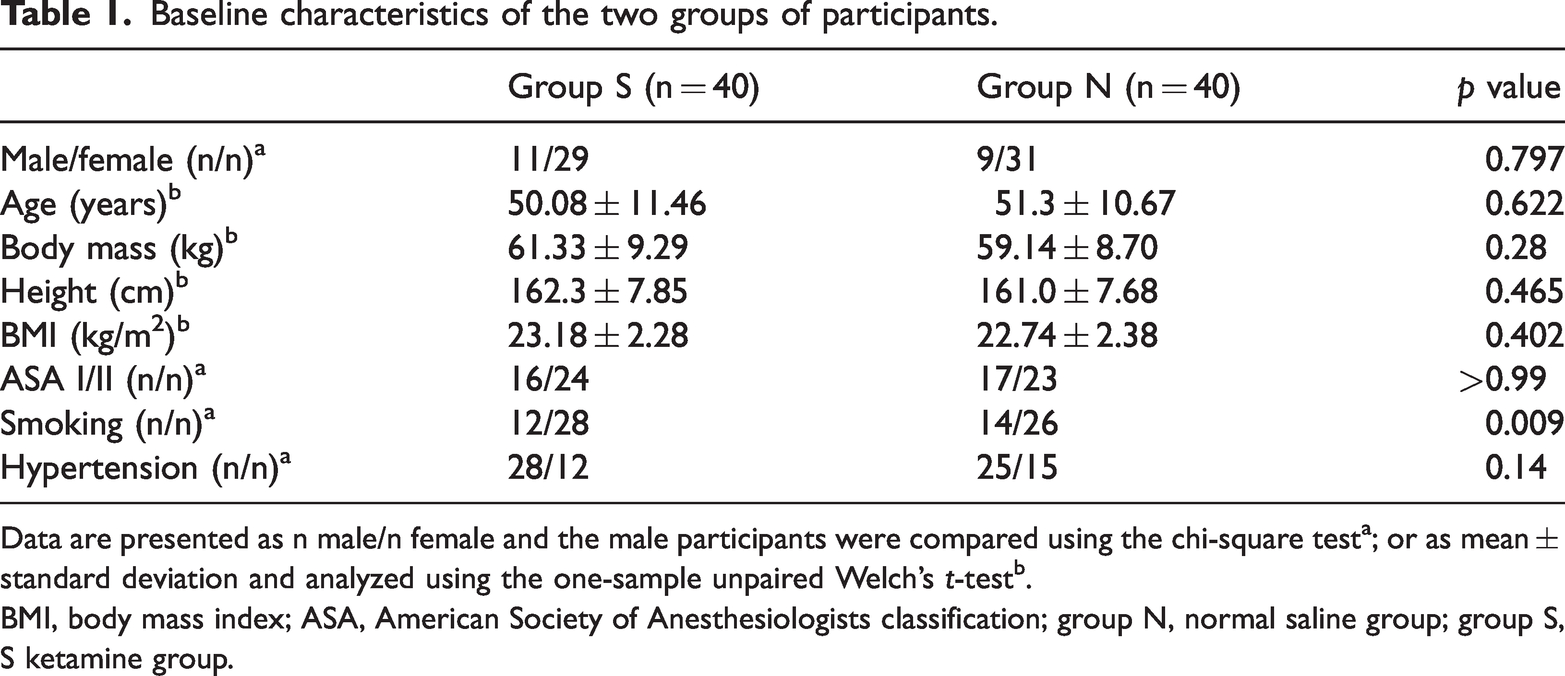
Data are presented as n male/n female and the male participants were compared using the chi-square testa; or as mean ± standard deviation and analyzed using the one-sample unpaired Welch’s
BMI, body mass index; ASA, American Society of Anesthesiologists classification; group N, normal saline group; group S, S ketamine group.
Secondary outcomes.

Data are presented as mean ± SD and analyzed using the one-sample unpaired Welch’s
group N, normal saline group; group S, S ketamine group; PONV, postoperative nausea and vomiting.
SPI
The SPI did not significantly differ between groups S and N at baseline (74.85 ± 10.75 and 75.95 ± 10.86, respectively), at intubation (52.83 ± 10.54 and 54.98 ± 14.69, respectively) or at skin incision (49.73 ± 10.24 and 49.6 ± 9.04, respectively). However, group S had lower SPI values 30 minutes after the start of surgery (32.28 ± 9.50

SPI values at various time points. The two groups were compared using Welch’s
Hemodynamic parameters
There was no difference in the MBP of the two groups, but the HR of the S-ketamine group was lower than that of the normal saline group, both 30 minutes after the start of surgery (66.33 ± 10.0

MBP and HR of the participants at each time point. a. MBP. b. HR. Welch’s
NRS pain score
At the end of surgery, a 28F tube was inserted for drainage. The NRS score was lower in the S-ketamine group 6 hours (1.45 ± 0.74
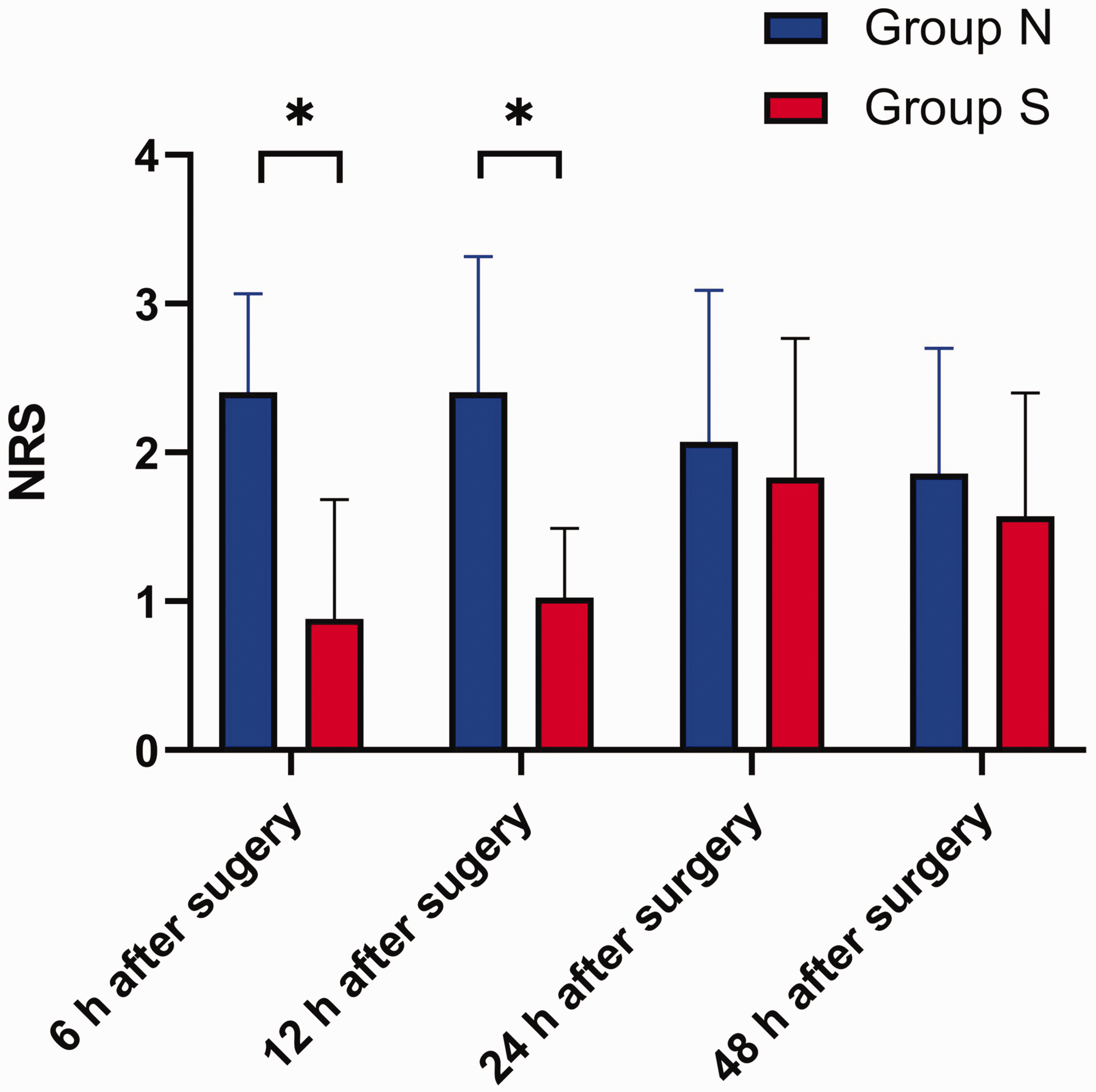
NRS score in the S-ketamine group at various postoperative time points. Welch’s
Discussion
In the present study, we found that the NRS score was lower in the S-ketamine group than in the control group 6 and 12 hours postoperatively, but there was no difference between the groups 24 and 48 hours postoperatively. The incidence of rescue analgesia and the total amount of flurbiprofen used over 48 hours were lower in the S-ketamine group. The administration of S-ketamine reduced the SPI and sufentanil dose required. The HR of the S-ketamine group was lower both 30 minutes after the start of surgery and at the end of surgery, but there was no difference in the incidence of PONV between the groups.
Minimally invasive video-assisted thoracoscopic surgery is widely used in the clinic, but patients can experience acute or chronic pain following lung surgery. 12 Paravertebral or intercostal nerve blocks are now used perioperatively, 13 but some patients still report intercostal pain. Furthermore, substantial postoperative opioid consumption 14 can lead to nausea, vomiting, respiratory depression and a delay in discharge. To improve the recovery of patients, S-ketamine 6 has been used by anesthesiologists to reduce the requirement for perioperative and postoperative analgesia, but the precise mechanism whereby S-ketamine has this effect has yet to be elucidated. In spinal surgery, S-ketamine has been shown to reduce opioid consumption and pain 15 in chronically opioid-dependent patients, and in major lumbar fusion surgery, S-ketamine has been shown to reduce oxycodone consumption over the first 48 hours post-surgery. 16 In the present study, the NRS score of the S-ketamine group was lower than that of the control group 6 and 12 hours postoperatively, but there was no difference in the NRS score 24 and 48 hours postoperatively. S-ketamine has also been shown to reduce the use of opioid intraoperatively, postoperative pain, the incidence of rescue analgesia, and the total amount of flurbiprofen used within the first 48 hours postoperatively, all of which are consistent with the present results. The explanation for the lack of difference in NRS at the later time points may be that patients feel pain most frequently during the first 12 or 24 hours postoperatively; participants in the present study were administered 5 mL ropivacaine injection at each incision and a PCA pump postoperatively. Most of the participants had a single incision and one small drainage tube was placed.
S-ketamine reduced the SPI and intraoperative sufentanil use, and led to stable hemodynamics during surgery. The SPI, which is on a scale from 0 to 100, with 0 representing the lowest response and 100 representing the highest response, calculated using the heart beat interval (HBI) and the photoplethysmographic amplitude (PPGA), 17 objectively reflects pain intensity during general anesthesia. SPI is calculated using the following formula: SPI = 100 − (0.33 × HBInorm + 0.67 × PPGAnorm). S-ketamine, an isomer of ketamine, has been shown to reduce opioid requirement during the perioperative period 18 , which is consistent with the present findings. However, it is unclear whether S-ketamine has any influence on SPI. SPI, which reflects the relationship between the intensity of surgical stimulation and anti-nociception during general anesthesia, is a reliable means of measuring autonomic responses to noxious stimuli. 19 Surgery stimulates the sympathetic nervous system, increasing HR, and leading to a change in SPI. In the present study, although there was no difference in the MBP of the S-ketamine and normal saline groups, the HRs of participants in the group N 30 minutes after the start of surgery and at end of surgery were faster than those in group S, which suggests that S-ketamine suppresses the relevant stimuli. Therefore, opioid drugs could be used more rationally by clinicians as part of a multimodal analgesia regimen.
Previous studies have shown that the SPI correlates with the NRS score, 20 but it can be affected by age 21 , 22 and BMI. 23 The SPI can be used to guide intraoperative analgesia, 24 and such SPI guidance significantly modifies intraoperative opioid use, with no changes in postoperative cortisol concentrations or postoperative pain levels. 25 In addition, in the present study, the S-ketamine group used less opioid and their NRS scores were lower during the first 12 hours postoperatively.
There were two principal limitations to the present study. First, we did not take into account the influences of age or BMI on SPI; and second, the levels of indicators of stress, such as catecholamines, were not measured during surgery.
Conclusion
The administration of S-ketamine reduces the SPI, the NRS score, and sufentanil use. Thus, by using the SPI, opioid use can be administered on an individual and rational basis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231198386 - Supplemental material for Effect of S-ketamine on the intraoperative Surgical Pleth Index in patients undergoing video-assisted thoracoscopic surgery: a single-center randomized controlled clinical trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605231198386 for Effect of S-ketamine on the intraoperative Surgical Pleth Index in patients undergoing video-assisted thoracoscopic surgery: a single-center randomized controlled clinical trial by Xian Chen, Yumin Zhu, Ke Peng, Qinyun Wang and Changdong Feng in Journal of International Medical Research
Footnotes
Author Contributions
KP conceived and designed the study. XC prepared materials, collected data, and wrote the manuscript. YZ analyzed and interpreted the data. CF conceived and designed the study, collected data, and revised the manuscript. QW designed the study and collected data.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The study was supported by the Beijing Medical Award Fund (no.: YXJL-2021-0170-0256).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
