Abstract
This case report describes a 69-year-old male patient with a renal artery aneurysm that was followed up with contrast-enhanced magnetic resonance angiography at 8 months after coil embolization treatment. Due to the disappearance of residual lumen with few metal artifacts, the therapeutic effect was satisfactory. At present, the indications for the treatment of renal artery aneurysms are still controversial and there are very few reports of postembolization images of renal artery aneurysms, with no criteria for reintervention and few reports for monitoring the embolized aneurysms. Further reports and research are still needed for the treatment of this rare disease.
Introduction
Renal artery aneurysm (RAA), which is mainly caused by fibromuscular dysplasia, atherosclerosis and dilatation after stenosis, is a rare type of visceral aneurysm; accounting for 14–37% of all visceral aneurysms, with an incidence of 0.1% in the population.1,2 Few patients have symptoms such as abdominal pain, hypertension and haematuria, 3 but most patients show no obvious clinical symptoms and are often diagnosed during imaging examinations. 4 RAAs have been categorized into three types: (i) type 1 includes saccular aneurysms arising from the main renal artery or a large segmental branch; (ii) type 2 includes fusiform aneurysms; and (iii) type 3 includes intralobar artery aneurysms. 5 Endovascular repair techniques, including artery stenting and aneurysm embolization, are highly recommended because of the advances in interventional materials and techniques, accompanied by avoidance of general anaesthesia, shorter hospital stays and lower complication rates. 6 However, as a rare and asymptomatic disease, there is still a lot of work to do on postoperative RAAs follow-up approaches. In order to evaluate the treatment of RAAs, appropriate imaging modalities should be used to conduct follow-up studies. When patients are examined by magnetic resonance (MR) or computed tomography (CT) imaging, the metal artifacts produced by indwelling coils or metal stents make it difficult to evaluate the condition of the aneurysm.7,8 Contrast-enhanced MR angiography (CE-MRA), which can effectively attenuate the phenomenon of metal artifacts, provides a clear observation of visceral aneurysms. This current case report describes a patient with RAA that was treated with renal artery stenting and coil embolization. Eight months after the operation, CE-MRA was used for follow-up of the patient. Few metal artifacts and disappearance of residual lumen confirmed the satisfactory therapeutic effect.
Case report
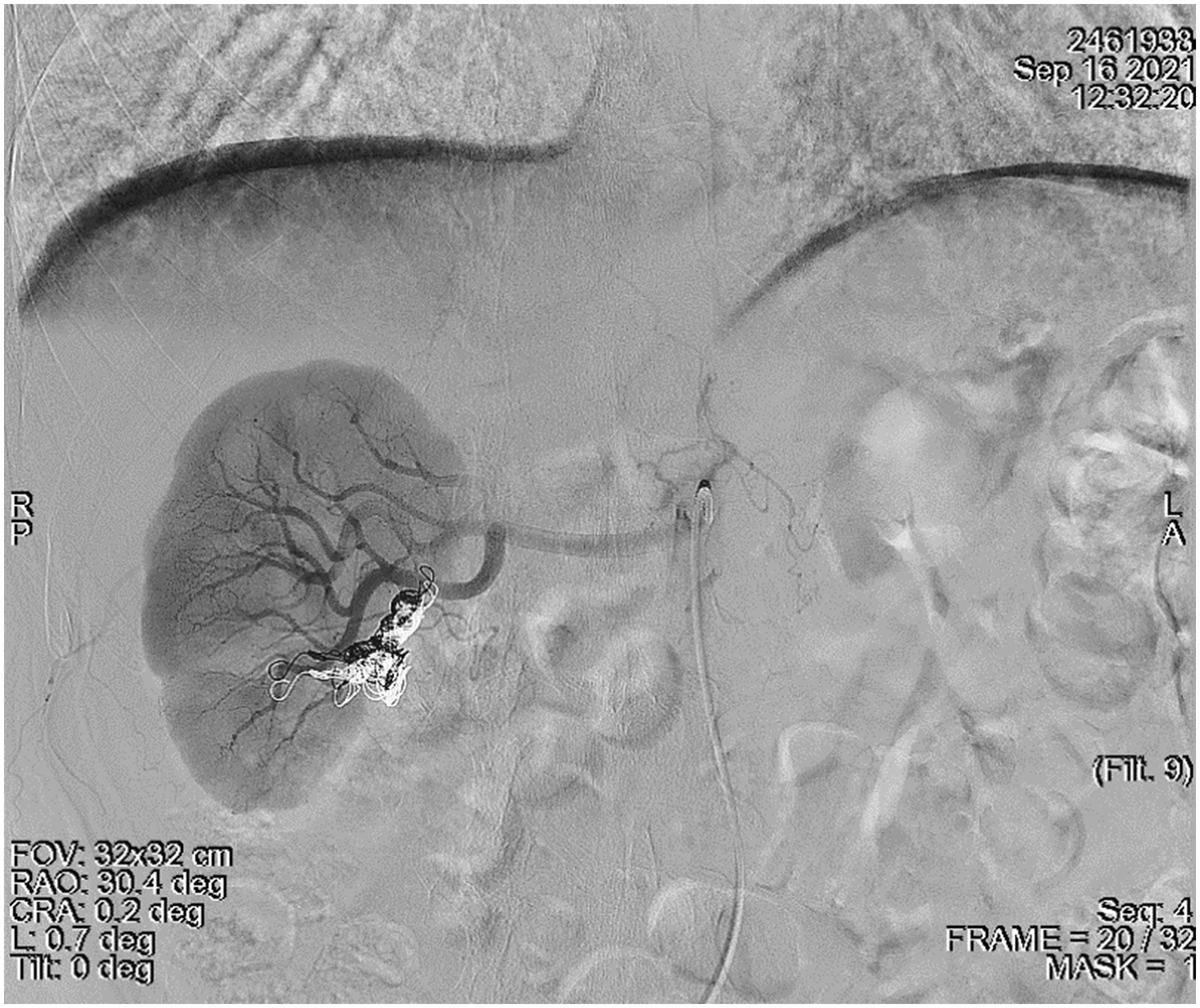
In January 2021, a 69-year-old male patient with no other symptoms except haematuria was referred to the emergency department of Peking University First Hospital, Beijing, China. CT imaging demonstrated a 1.8 cm × 2.3 cm × 2.0 cm well-circumscribed aneurysm that was enhanced homogenously with few calcifications arising from a branch of the main renal artery. The patient did not have any recent surgeries or traumas. Renal scintigraphy evaluating the split renal function showed that there were no abnormalities in either kidney function. Renal arteriography showed there was no stenosis in the trunk or branches of the right renal artery, and an aneurysm was observed in the lower pole branch of the right renal artery, with the parenchyma of the right kidney well visualized (Figure 1). This patient underwent endovascular embolization after angiography. A 7F guiding catheter was introduced to the trunk of the right renal artery, followed by continuous heparinized saline infusion, and the micro guidewire and microcatheter were inserted through the aneurysm lumen into the distal outflow branch of the aneurysm. A 6 mm × 20 cm and a 10 mm × 30 cm detachable microcoil (Interlock-35; Boston Scientific, Marlborough, MA, USA) were deployed successively under fluoroscopy in the distal outflow tract, and then an 8 mm ×20 cm Interlock-35 coil (Boston Scientific) was deployed in the aneurysm sac. A 6 mm × 20 cm Interlock-35 coil (Boston Scientific) and an MWCE-18S-3/2 Tornado® Embolization Coil (Cook Medical, Bloomington, IN, USA) were deployed in the proximal inflow passage of the aneurysm lumen. Postoperative angiography showed that most of the aneurysm sac was opacified and the rest of the renal parenchyma was well perfused (Figure 2). Haematuria disappeared after the surgery.
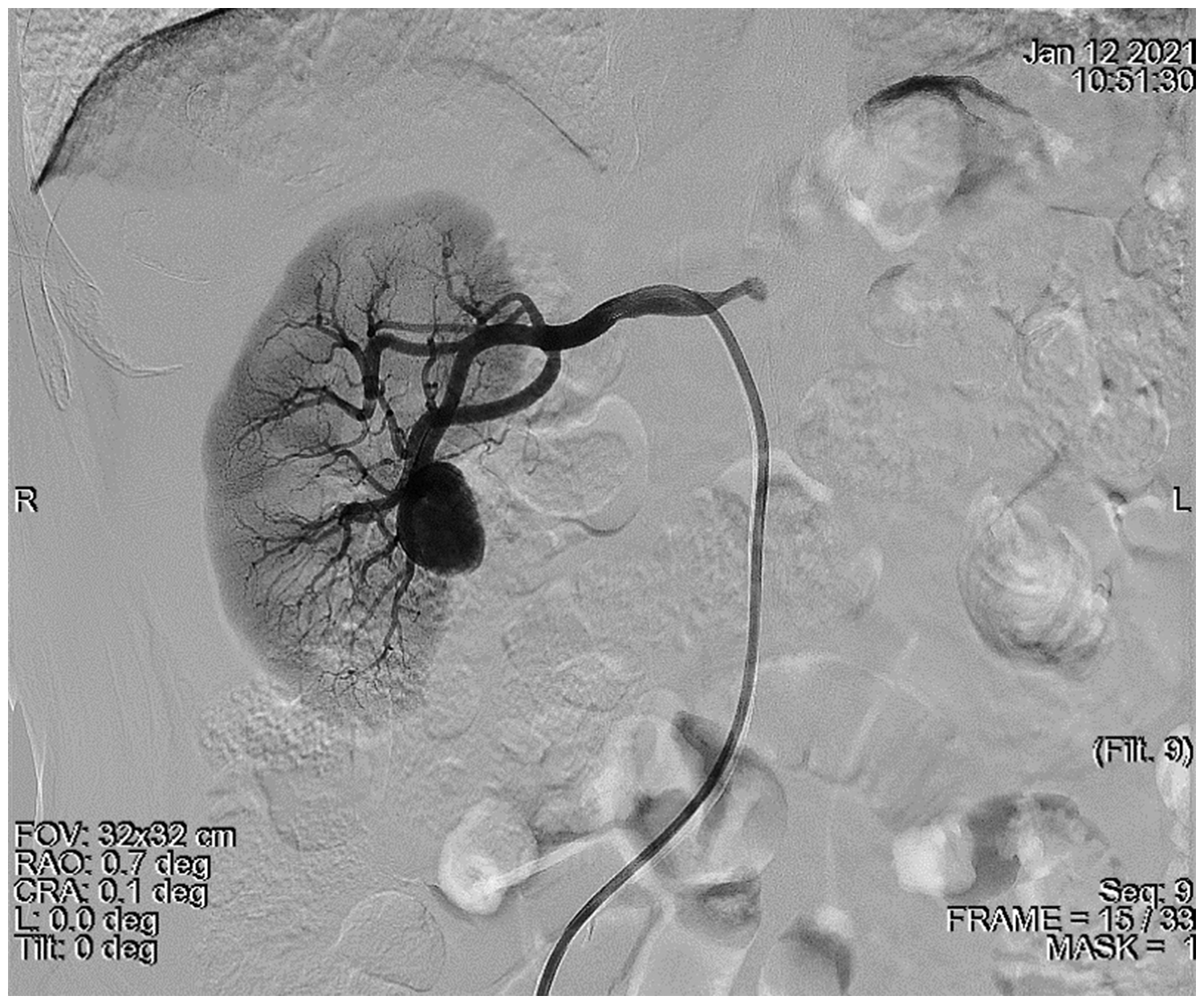
Pre-embolization angiogram of the renal artery aneurysm in a 69-year-old male patient with no other symptoms except haematuria.

Postembolization angiogram in a 69-year-old male patient with a renal artery aneurysm but no other symptoms except haematuria.
The patient was scheduled to return for additional follow-up 8 months after the procedure. Under CE-MRA examination (3.0 Tesla, echo time 22, echo train length 27, repetition time 700, flip angle 120), there were few metal artifacts and the embolized aneurysm had clear and intact margins without significant shrinkage (Figure 3). However, there appeared to be a slightly higher signal at the aneurysm edge by digital subtraction processing (Figure 4). To identify if this was a small amount of blood flow into the residual lumen, the patient underwent angiography. Three-dimensional angiography showed that the wall of the main trunk of the right renal artery was patent, without stenosis or filling defects. The coil was located in the lower pole branch of the right renal artery and no contrast agent filling was found in the previous aneurysm lumen (Figure 5), which meant that there was no residual lumen in this patient. The embolization of the renal artery aneurysm was satisfactory.

At 8 months after the embolization procedure, contrast-enhanced magnetic resonance angiography examination of a 69-year-old male patient demonstrated that the embolized aneurysm had clear and intact margins, with few metal artifacts observed (arrow).

At 8 months after the embolization procedure, digital subtraction processing of a contrast-enhanced magnetic resonance angiography image of a 69-year-old male patient appeared to show a slightly higher signal at the aneurysm edge (arrow).
At 8 months after the embolization procedure, contrast-enhanced magnetic resonance angiography examination of a 69-year-old male patient demonstrated a satisfactory embolization effect with no contrast agent filling found in the aneurysm lumen.
Written informed consent for treatment and publication of the case report and accompanying images was obtained from the patient. A formal ethical review by an ethics committee was not required because of the nature of this study (case report). The reporting of this study conforms to CARE guidelines. 9
Discussion
Currently, well-accepted surgical indications for RAAs include the following: (i) aneurysm diameter ≥ 2 cm; (ii) women of childbearing age; (iii) lumbago, backache, haematuria and other clinical symptoms; (iv) renal artery stenosis, thromboembolism and dissection associated with refractory hypertension. 10 Due to the lack of randomized controlled studies, the first-line treatment options of RAAs are still controversial. Previously, RAAs were often diagnosed definitively when they ruptured spontaneously, which occurs in 14–30% of patients and has a fatality rate of 80%. 4 In recent years, with the development of imaging technology, the detection rate has been distinctly improved. For example, research has shown that the incidence of ruptured RAAs has been reduced to 3–5% and nonpregnancy-related mortality has been reduced to less than 10%. 2 Therefore, the threshold for intervention may raise due to the benignity of RAAs. 11 The 2020 guidelines of the American Society of Vascular Surgery for the diagnosis and treatment of visceral aneurysms recommend that a maximum aneurysm diameter of 3 cm should be the invasive threshold for patients with asymptomatic RAAs, however, variation in treatment is acceptable on the basis of individualized clinical decision-making. 4 Complications of endovascular treatment are uncommon, but include renal vessel damage, infarction, hypertension, untargeted embolism (coil migrates to the renal main trunk or systemic circulation), stent thrombosis, continued enlargement of the aneurysm, abdominal pain and infection. 8
For patients with distinct indications for surgery, endovascular therapy, including coil embolization and stenting, is often preferred. 6 It can effectively block blood flow into the aneurysm lumen, but perhaps not 100%, because of the gaps between the coils and the vessel wall. If the aneurysms cannot be packed densely, blood flow can enter the lumen through the gaps, and turbulence of blood flow in the residual aneurysm may lead to the expansion or movement of the coils, increasing the risk of rupture, which has been reported in the follow-up of intracranial aneurysms. 12 Therefore, aneurysm volume and residual lumen are issues that clinicians should focus on during follow-up, so as to accumulate experience for the effective and reasonable treatment of this rare disease.
When follow-up for aneurysms is needed, the metal artifacts produced by the coils or metal stents remain a large obstacle to the quality of the images. Despite the orthopaedic metal artifact reduction function and dual-spectrum CT technology being able to reduce metal artifacts,13–15 the observation of organs and tissues close to metal objects is still not satisfactory in computed tomography. In MR imaging, the presence of metal objects makes the original magnetic field strength uneven and leads to spatial positioning and expression error so that the signal around the metal is inconsistent with the anatomical structure, resulting in artifacts, which seriously affect the quality of images and interfere with clinical diagnosis. 16 Metal materials can be classified as paramagnetic, diamagnetic or ferromagnetic according to different magnetic susceptibilities.17,18 The contrast-enhanced MR angiography technology used in this current case is able to shorten the T1 value of blood significantly after injecting contrast agent and make a clear contrast with the surrounding tissues, which means it can be used to observe the blood flow in the aneurysm lumen without being affected by blood turbulence. In several studies, CE-MRA has been used in the follow-up of postoperative visceral aneurysms, providing high-quality images equivalent to digital subtraction angiography for the evaluation.19,20 The Society for Vascular Surgery clinical practice guidelines on the management of visceral aneurysms has included MRA as a recommended modality for RAAs follow-up.4,21 The safety problems caused by metal implants in magnetic fields need to be addressed by clinicians. It is notable that the evaluation of heating and induced currents for implants of various shapes and sizes shows these implants do not appear to have any safety issues. 22
With no literature or guidelines for the classification of unruptured RAAs, how to grade embolized RAAs is also a problem remaining to be solved. In contrast, the Raymond-Roy grading is used for embolized intracranial aneurysms, which are, namely, total occlusion (grade 1), residual neck (grade 2) and residual aneurysm (grade 3). 23 It should be stressed, however, that this grading method does not apply to embolized RAAs. In general, renal aneurysms and intracranial aneurysms have vastly different sizes, while unruptured intracranial aneurysms are much smaller.24,25 The larger lumen renal aneurysms remain, the more complex thrombus organization and residual blood flow they have. Some research has suggested that favourable results for coil packing of visceral aneurysms seem to depend on the coil packing density and recanalization more often occurs in patients with large aneurysms.26,27 In this regard, we suggest that the ratio of the maximum flow signal area to the total lumen area can be used to evaluate the condition of the residual lumen. Further research and exploration are still needed to establish unruptured RAA grading standards.
In the follow-up evaluation, it is crucial to distinguish which renal aneurysms are more likely to rupture. A multicentre study showed that only aneurysms larger than 3 cm ruptured, accounting for 0.55% of 547 patients with untreated RAAs. 11 One of the ruptured aneurysms was reported, which occurred in a patient with suspected connective tissue disease. 11 Pregnancy is associated with a higher rate of aneurysm rupture, possibly due to changes in blood vessel flow and hormone levels, which may weaken the elastic tissue of the artery wall. However, the true incidence of RAAs rupture in pregnant women remains unknown. 2 Since it was found that in the assessment of other aneurysms, such as aortic aneurysms, rupture is caused by internal leakage or residual lumen enlargement, it is believed that a large residual lumen can also lead to rupture of postoperative RAAs.28,29 Therefore, for RAAs in patients with pregnancy, connective tissue disease and other factors that may increase the risk of cardiovascular events, clinicians should focus on the aneurysm size and residual lumen in the follow-up evaluation. Nonetheless, the lack of sufficient clinical studies makes clear that more research is needed to determine which aneurysms are more likely to rupture.
At present, there are still few reports on the postoperative follow-up of RAAs, with no classifications of unruptured RAAs performed. With the aid of the popularization of ultrasound and CT scans, an increasing number of asymptomatic renal artery aneurysms will be found accidentally. By drawing on the theory and practical experience of other aneurysms, it should be possible to establish grading and evaluation criteria for unruptured RAAs. In addition, due to the few metal artifacts produced, CE-MRA is a good method for the follow-up of postoperative RAAs.
