Abstract
Objective
To conduct a network meta-analysis of randomised controlled trials to determine the optimal clinical choice of first-line therapy for patients with ALK receptor tyrosine kinase (ALK) gene rearrangement non-small cell lung cancer (NSCLC).
Methods
Clinical trials in patients with histologically confirmed ALK gene rearrangement NSCLC, that included ALK inhibitors as first-line therapy, were identified using database searches. A Bayesian network meta-analysis was conducted to calculate the efficacy and safety of the included first-line treatments.
Results
Nine trials with 2,407 patients were included for analyses. Lorlatinib was better than brigatinib for progression-free survival (PFS) (hazard ratio 0.79, 95% confidence interval 0.63, 0.98). In subgroup analyses, lorlatinib exhibited the highest probability of best PFS ranking in patients with or without baseline brain metastases (38% and 80%, respectively); brigatinib had the highest probability of best PFS ranking among Asian patients (47%). Alectinib offered the highest survival advantage (57% probability), while lorlatinib was likely to be the best treatment for an objective response (41% probability). Alectinib displayed the highest probability of being ranked lowest for grade ≥3 adverse events (86%).
Conclusions
Lorlatinib was associated with the best PFS overall, and was suitable for patients with or without brain metastases. Brigatinib was associated with the best PFS in Asian patients.
Keywords
Introduction
Lung cancer is a common but complex malignancy, with a high rate of morbidity and mortality. Non-small cell lung cancer (NSCLC) accounts for 80–85% of all reported lung cancer cases, 1 and rearrangement of the ALK receptor tyrosine kinase (ALK) gene (also known as anaplastic lymphoma kinase) has been identified as one of the driving factors of NSCLC, 2 with ALK gene rearrangement occurring in approximately 3–5% of NSCLC cases. Patients with ALK gene rearrangement tend to be young (approximately 50 years old at the time of diagnosis) and never smokers (approximately 70–75%), but also include a small number of smokers. Additionally, most of the cancers are adenocarcinomas and have no sex preference. 3 To date, first-line treatment options for patients with ALK gene rearrangement in NSCLC include various generations of ALK inhibitors, such as first-generation crizotinib, second-generation alectinib, brigatinib, ceritinib, and ensartinib, and third-generation lorlatinib, 4 however, which of these options provides optimal outcomes remains unclear.
Despite the results of several randomized controlled trials, and paired meta-analyses using only direct comparison models, there is inconclusive evidence regarding the comparative efficacy and safety of first-line treatment, and thus, optimum treatment, in patients with advanced ALK gene rearrangement NSCLC. A network meta-analysis, in which evidence from direct and indirect comparisons is synthesized, enables the determination of best available treatments. 5 Previous network meta-analyses have only partially compared treatment options for patients with advanced ALK gene rearrangement NSCLC, as they have not included more recent alternative therapies or available trials, or were not sufficiently specific in terms of accurate treatment options for different ethnic populations or tumours with different subtypes of mutations.6–9 Through a carefully designed comparative synthesis, the aim of the present study was to conduct a network meta-analysis of randomized controlled trials to investigate the relative efficacy and safety of all first-line treatments in patients with advanced ALK gene rearrangement NSCLC, and to analyse subgroups of patients with ALK gene rearrangement, to determine the best clinical choice.
Materials and methods
Search strategy
The PubMed, Embase, and ClinicalTrials.gov databases were searched for articles published in English, up to 1 March 2021, using the keywords ‘crizotinib’, ‘alectinib’, ‘ceritinib’, ‘brigatinib’, ‘lorlatinib’, ‘ensartinib’, and ‘non-small cell lung cancer’, and the category was limited to ‘clinical trial’. The specific search terms are presented in supplementary Table 1. To improve the credibility of this research, the outcomes of included studies were updated using data from the abstracts of key international conferences up to 1 March 2021 (i.e., the World Conference on Lung Cancer, the American Society of Clinical Oncology, the European Society for Medical Oncology, and the Chinese Society of Clinical Oncology). This network meta-analysis was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension statement 2020 for network meta-analysis (supplementary Table 2).10,11 The protocol was registered in the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY202170079; https://inplasy.com/inplasy-2021-7-0079/).
Study selection
Studies that met the following criteria were included: phase II/III randomized controlled trials; included patients with histologically confirmed NSCLC with ALK gene rearrangement; comprised two or more comparison arms; and reported at least one of the following clinical outcomes: objective response rate (ORR), progression-free survival (PFS), overall survival (OS), and grade ≥3 adverse events (AEs).
Following removal of duplicates, the titles and abstracts were screened by two researchers (CFZ and LZ). The full texts of potentially eligible articles were then assessed by two researchers (JHT and CJZ) for final inclusion. The risk of bias of individual studies was analysed using the Cochrane risk-of-bias tool. 12 Items were scored as having a low, high, or unclear risk of bias.
Data extraction
Two independent researchers (YZ and XH) analysed the included trials according to a predefined protocol, and any discrepancies were resolved by discussion with a third researcher (ZL). The following information was extracted: study ID, intervention arm, control arm, median PFS, and median OS. ORR data based on Response Evaluation Criteria in Solid Tumours (RECIST) criteria were extracted directly from the published trials. For trials in which both per-protocol and intention-to-treat (ITT) analysis data were disclosed, data from ITT populations were preferentially extracted and evaluated by blinded independent central review to include more objective data. Data from the original published studies were replaced with the most recent data from conference abstracts, according to availability. Data recorded as treatment-related grade ≥3 AEs were preferentially extracted; however, if AEs were not specified in a study as being treatment-related, all grade ≥3 AEs were extracted.
Data synthesis and statistical analysis
All direct and indirect data were synthesized to compare different treatments in terms of efficacy and safety. Survival outcomes (PFS and OS) are presented as hazard ratio (HR) and binary outcomes (ORR and grade ≥3 AEs) are presented as odds ratio (OR), along with corresponding 95% confidence intervals (CIs). The principal evaluation criterion was PFS, and the secondary criteria were ORR, OS, and safety. Subgroup analyses were performed in patients with or without baseline brain metastases, and in Asian patients.
A Bayesian network meta-analysis protocol was applied for its advantages in offering a relatively straightforward method for conducting probabilistic statements and predictions on the treatment effects. 13
Using Stata software, version 16.0 (Stata Corp, College Station, TX, USA), network plots were generated for different treatments to clarify mutual relationships in the included treatments. 14 Heterogeneity was assessed by conducting a pairwise meta-analysis based on two or more trials. Network meta-analyses were performed in OpenBUGS software, version 3.2.3 (https://www.mrc-bsu.cam.ac.uk/software/bugs/openbugs/) under the Bayesian framework using a Markov Chain Monte Carlo simulation technique.
Transitivity was ensured by including literature and study groups with strict certification. The fit of consistency and inconsistency models was compared using the deviance information criterion (DIC), 15 a Bayesian model evaluation criterion that measures model fit adjusted with the complexity of the model, where smaller DIC values correspond to more preferable models. For PFS and OS effects, 150 000 sample iterations were generated with 80 000 burn-ins and a thinning interval of 1. For ORR and grade ≥3 AEs, 70 000 sample iterations and 50 000 burn-ins were added, and the thinning interval was increased to 10 to minimize autocorrelation. Non-informative uniform and normal prior distributions and three different initial values were used to fit the model. The convergence of iterations was evaluated by visual inspection of the three chains to establish homogenous parameter estimates and the Brooks-Gelman-Rubin diagnostic. Bayesian ranking profiles of comparable treatments in different populations were also listed, as the Bayesian ranking results were almost in line with the pooled analyses using hazard and odds ratios. 13
To assess the robustness and reliability of the results, three sensitivity analyses were planned. The dosage of alectinib was 300 mg in the J-ALEX study, but was 600 mg in the other clinical studies; therefore, the J-ALEX study was excluded from the pooled PFS analysis and further subgroup analysis in PFS, to make the research more persuasive. Publication bias was measured by Funnel plot.
Results
Study selection and study characteristics
A total of 6524 records were identified. After removal of duplicate records, 134 records were identified from the initial title and abstract screen, and underwent full-text review. Finally, nine randomized controlled trials were deemed eligible for inclusion, with a total of 2,407 patients enrolled to receive seven different treatments (Figure 1).16–28 A network diagram of treatments is presented in Figure 2, in which seven studies are represented. The main characteristics of all of the studies are reported in supplementary Table 3. Assessment of bias risk showed that studies mainly exhibited low risk of bias with few concerns (supplementary Figure 1 summarizes the detailed risk of bias).
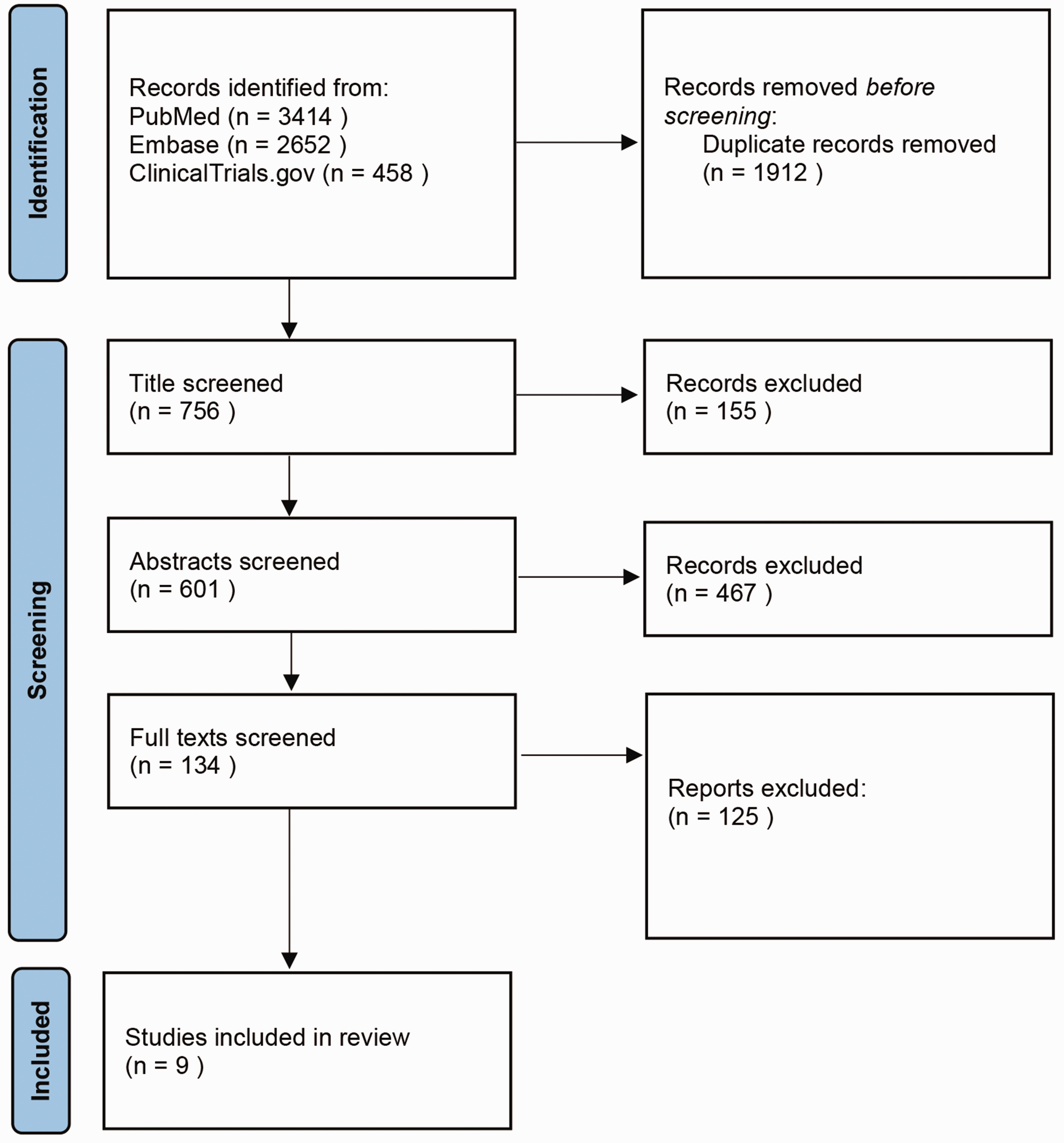
PRISMA flowchart showing selection of studies investigating ALK receptor tyrosine kinase inhibitors as first-line therapy in patients with ALK gene rearrangement non-small cell lung cancer.
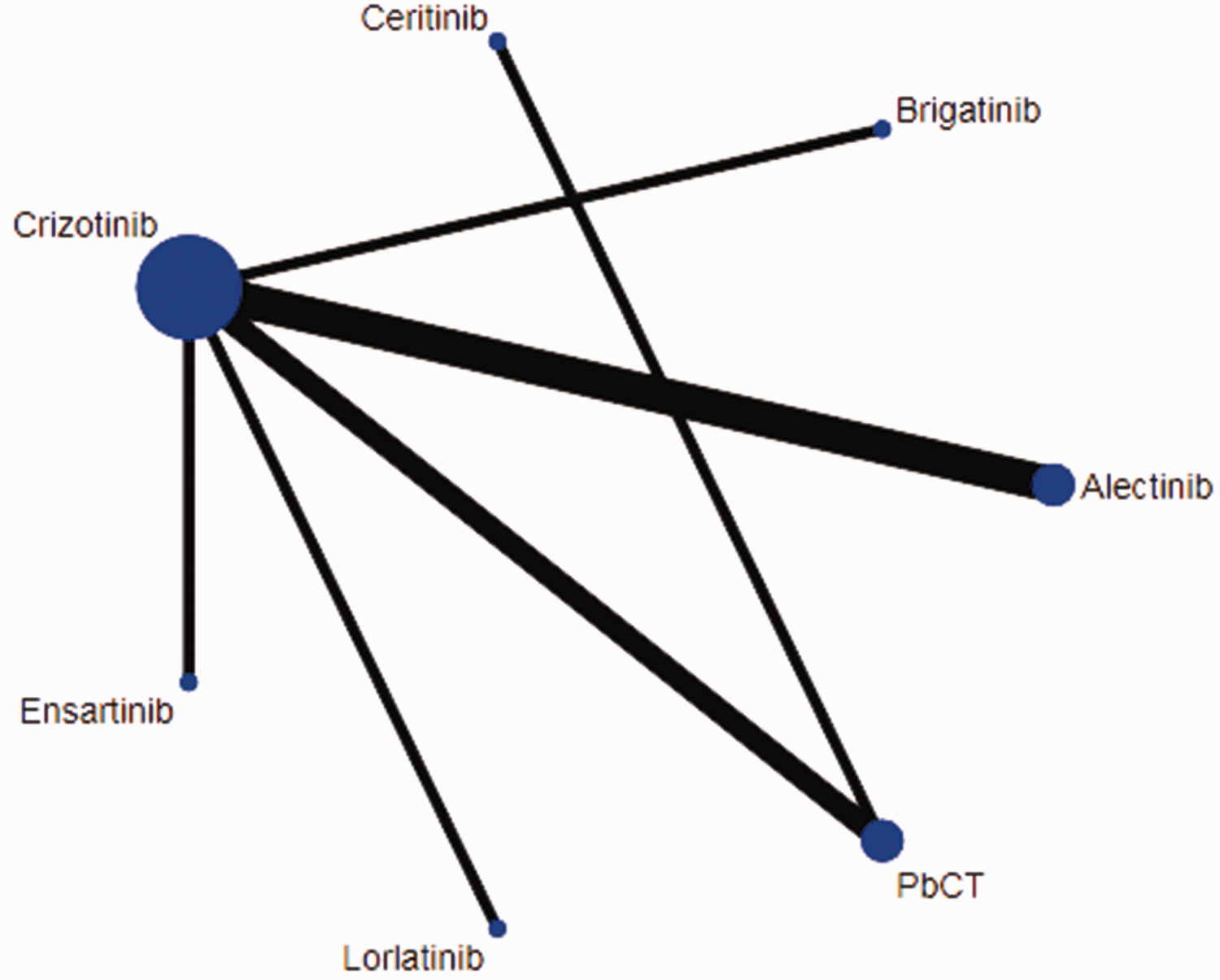
Network diagram of different treatments in patients with ALK gene rearrangement non-small cell lung cancer. PbCT, pemetrexed-based chemotherapy. Node size represents number of participants randomized to each drug; edge thickness represents the number of clinical trials in which head-to-head comparisons between drugs have been conducted.
The network meta-analysis included seven treatments for PFS and OS, seven treatments for ORR, and six treatments for grade ≥3 AEs (supplementary Figure 2). The applicable model for analyses was selected according to the results of DIC testing (supplementary Table 4).
Network meta-analysis of clinical efficacy
In terms of PFS, targeted drugs showed significant advantages over pemetrexed-based chemotherapy for ALK gene rearrangement advanced NSCLC in the pooled analysis. In all patients, alectinib, brigatinib, lorlatinib, and ensartinib significantly prolonged the PFS and performed better than crizotinib and ceritinib. When comparing these drugs one by one, lorlatinib was associated with better PFS than brigatinib (Table 1a). In Bayesian rank analysis, lorlatinib was placed at the highest position (89%; supplementary Table 5). Through a dual evaluation of surface under the cumulative ranking (SUCRA) curve rating, lorlatinib was found to have the highest probability of reaching the best PFS benefits (84.36%) with relatively low probability of grade ≥3 adverse effects (Figure 3).
Pooled estimates of the network meta-analysis, showing: (a) progression-free survival; (b) overall survival; (c) objective response rate and (d) grade ≥3 adverse events associated with first-line therapy in patients with ALK gene rearrangement non-small cell lung cancer.

Data presented as hazard ratio (95% confidence interval [CI]) for progression-free survival and overall survival, and odds ratio (95% CI) for overall response rate and grade ≥3 adverse events.
PbCT, pemetrexed-based chemotherapy.

Surface under the cumulative ranking (SUCRA) curve values of both progression-free survival (efficacy) and grade ≥3 adverse events (tolerability) in pooled analysis of first-line treatments in patients with ALK gene rearrangement non-small cell lung cancer. Ensartinib was excluded from this ranking due to the lack of adverse event descriptions). PbCT, pemetrexed-based chemotherapy.
In terms of OS, alectinib was associated with providing the best OS (57% probability of being ranked first; supplementary Table 5), and also significantly prolonged the OS compared to pemetrexed-based chemotherapy (HR 0.73, 95% CI 0.61, 0.88) or crizotinib (HR 0.81, 95% CI 0.70, 0.94) (Table 1b).
In terms of the ORR, pemetrexed-based chemotherapy was associated with the worse ORR compared with other treatments (probability of being ranked last, 93%; supplementary Table 5). Compared with pemetrexed-based chemotherapy, alectinib (OR 11.54, 95% CI 2.40, 68.88), brigatinib (OR 8.79, 95% CI 1.03, 88.24), ceritinib (OR 7.18, 95% CI 1.22, 44.53), crizotinib (OR 5.16, 95% CI 1.55, 19.30), and lorlatinib (OR 13.39, 95% CI 1.70, 127.71) all yielded a greater ORR benefit (Table 1c). The Bayesian ranking showed that lorlatinib was likely to be the best treatment in achieving an objective response (41%; supplementary Table 5).
Subgroup analysis for PFS
Six treatments were included in the analysis of patients with baseline brain metastases, among which, lorlatinib had the highest rank (38%; Figure 4A; supplementary Table 5) and therefore provided the best PFS benefit. Six treatments were also included in the analysis of patients without baseline brain metastases, and lorlatinib again yielded the best PFS benefits among all treatments (80%; Figure 4B). Furthermore, of the six treatments included in the subgroup analysis of Asian patients, brigatinib displayed the highest probability of being ranked best for PFS (47%) (Figure 4C, supplementary Table 5; supplementary Figure 3).

Bayesian ranking profiles for progression-free survival between first-line treatments in subgroup analyses of patients with ALK gene rearrangement non-small cell lung cancer, showing: (a) patients with brain metastases; (b) patients without brain metastases and (c) Asian patients. PbCT, pemetrexed-based chemotherapy.
Results of toxicity analysis
In terms of safety, little difference was found in grade ≥3 toxicity between alectinib, brigatinib, crizotinib, and lorlatinib (Table 1d). Ceritinib (750 mg, fasted) was associated with the highest probability (82%) of being ranked first for grade ≥3 AEs versus other treatments, while alectinib had the highest probability (86%) of having the lowest level of grade ≥3 AEs among all treatments (supplementary Table 5).
Convergence testing and heterogeneity analysis
Results of analyses of the convergence of iterations are shown in supplementary Figure 4. Additionally, forest plots of four feasible pairwise comparisons with heterogeneity estimates are shown in supplementary Figure 5. Heterogeneity was detected in comparisons of crizotinib versus pemetrexed-based chemotherapy for ORR, PFS, and grade ≥3 AE analysis. Heterogeneity was also observed among alectinib versus crizotinib in the safety analysis. According to the direct comparison results in supplementary Figure 5, consistency was observed between direct and indirect evidence after comparing results from pairwise meta-analyses and network meta-analyses. The funnel plot demonstrates a low publication bias for this study and the high confidence level of the findings (Figure 5).
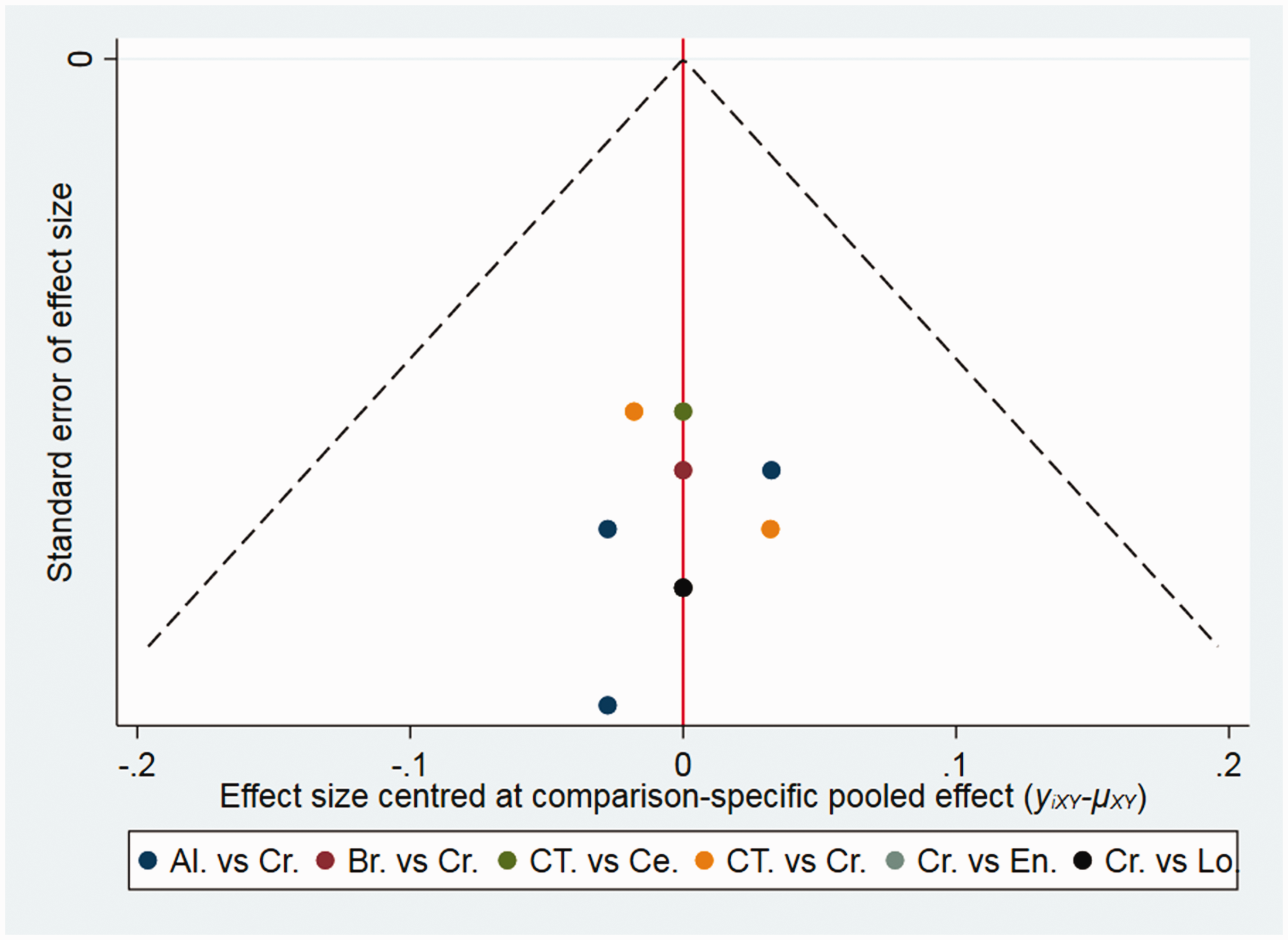
Funnel plot of publication bias analysis among studies investigating ALK receptor tyrosine kinase inhibitors as first-line therapy in patients with ALK gene rearrangement non-small cell lung cancer. Al, alectinib; Br, brigatinib; Ce, ceritinib; Cr, crizotinib; CT, pemetrexed-based chemotherapy; En, ensartinib; Lo, lorlatinib.
Sensitivity analysis
The J-ALEX study was excluded from the sensitivity analysis. The results of excluding the J-ALEX study did not show relevant deviations compared with the original network meta-analysis that included the J-ALEX study. The results showed the same probability of ranking best for PFS in pooled analysis (89%), but higher probabilities in subgroup analysis (patients with brain metastases, 40%; patients without brain metastases, 81%; and Asian patients, 51%; supplementary Table 6).
Discussion
As the range of novel ALK inhibitors for treating ALK gene rearrangement NSCLC continues to increase, the optimal selection sequence for these inhibitors in such treatment requires further investigation. However, systematic comparisons among treatment strategies remain scarce. The present study provides evidence to fill this knowledge gap, by accurately evaluating the application of ALK inhibitors to treat ALK gene rearrangement NSCLC.
In this network meta-analysis, the efficacy and safety of first-, second-, and third-generation targeted therapy for ALK gene rearrangement NSCLC was comprehensively compared. The results suggest that targeted therapy is superior to chemotherapy, which concurs with previous analyses.17,23 Furthermore, the third-generation ALK inhibitor lorlatinib was shown to be consistent in providing the best PFS and ORR in patients with ALK gene rearrangement NSCLC, through direct and indirect comparison of the evidence. Peled et al. 29 reported that lorlatinib showed outstanding extracranial and intracranial efficacy in ALK/ROS proto-oncogene 1, receptor tyrosine kinase (ROS1) (+) NSCLC. The CROWN trial investigators also found that those who received lorlatinib had significantly longer PFS than those who received crizotinib. 27 This may be because, in previously untreated patients, lorlatinib could eliminate rare subclones containing ALK-resistant mutations, or prevent the emergence of such resistant subclones. Additionally, lorlatinib continued to display significant efficacy in patients with tumour progression receiving first- or second-generation inhibitors, and achieved better efficacy in patients with secondary ALK-resistant mutations.30–32 Furthermore, clinical studies have shown that lorlatinib is effective in the treatment of central nervous system (CNS) metastases.31,33 In a phase 2 study of lorlatinib, 53–56% of intracranial reactions were confirmed. 34 Additionally, in the CROWN trial, lorlatinib significantly reduced the cumulative incidence of CNS progression, suggesting that the prolonged PFS associated with lorlatinib might be partly due to the prevention of CNS metastasis.27,35
In the present meta-analysis, ceritinib (750 mg fasted) wash shown to cause more toxicity in general, while alectinib was associated with the least side effects. In the ASCEND-8 study, ceritinib (450 mg with food) had comparable exposure and better gastrointestinal tolerability than 750 mg fasted. Both arms showed a similar robust efficacy in terms of ORR, disease control rate, and time to response. In terms of PFS, the median PFS in the ceritinib (450 mg with food) group was still increasing after 38 months of follow-up, while PFS in patients who received 750 mg fasted was 12.2 months. These results suggested a preferred ceritinib dosing regimen of 450 mg in patients with ALK gene rearrangement NSCLC. 36 However, the drug attenuation trial was a self-control study that did not meet the inclusion criteria for the present meta-analysis, and was therefore excluded. Regarding alectinib, although treatment cycles were longer than for other drugs in several studies, the incidence of grade ≥3 treatment-related AEs, and AEs causing treatment interruption, was lower in patients who received alectinib than in the control group.37,38 These results fully demonstrate the safety of alectinib and reveal that alectinib is associated with the least side effects in an indirect and direct comparison between all available ALK inhibitors.
Sensitivity analyses showed that the overall results remained relatively robust. In a previously published meta-analysis of first-line treatments for patients with ALK gene rearrangement NSCLC, 8 the subgroup of Asian populations, among which the rate of lung cancer is the highest in the world, was not investigated. In the present subgroup analysis of Asian patients, brigatinib, but not lorlatinib, was associated with better PFS. Possible reasons for this result include the following: first, there may be differences in drug metabolism between different ethnic groups; 39 secondly, the efficacy of brigatinib was better in the Asian population than in the whole population; and thirdly, the clinical characteristics of eligible patients differed between each randomized controlled trial, in terms of smoking history, ethnicity, region, medical compliance, the proportions of different tumour histologies. The present study found a superior efficacy for brigatinib in the treatment of Asian patients, and although this result requires further confirmation, the impact of these distinctions cannot be ignored.
The present study ensured homogeneity of the study population by including only patients with ALK gene rearrangement NSCLC. All monotherapies (including first-, second-, and third-line treatments) were compared and all of the major efficacy and toxicity outcomes were comprehensively analysed using the broadest available data, including previously published or recently updated results. In particular, the effectiveness of treatment was compared between subgroups. Additionally, the transitivity, heterogeneity, and inconsistency of the network were fully investigated, which increased the reliability and credibility of this study and provided a certain reference value for treatment. Based on the evidence of all randomized controlled trials, the present results provide a reference source for clinicians to evaluate the advantages and disadvantages of practical choice among several good treatment choices. In patients with or without brain metastases, lorlatinib seems to be the best treatment for ALK gene rearrangement NSCLC; however, its toxicity still requires close attention. As a supplement to recent guidelines, the present findings may help to clarify the role of targeted therapy in the standard care of patients with ALK gene rearrangement NSCLC, and highlight which treatment may be most suitable in such patients. Future trials should emphasize face-to-face comparisons between second- and third-line strategies, which may be more effective, especially under the continuous challenge of drug resistance.
We acknowledge several limitations of the present study. First, the inevitable confounding factors remain in this network meta-analysis based on internal observation of clinical trial data. Most treatments were indirect comparisons, and most direct evidence came from trials in the current network. Therefore, attention should be paid to their dependence on the previous distribution and transitivity and consistency assumptions when interpreting estimates, even if only randomized controlled trials were included. Secondly, this study only evaluated the advantages and disadvantages of first-line treatment and did not explore whether there is appropriate follow-up treatment after first-line treatment, which often greatly impacts OS. Thirdly, data on OS may lead to heterogeneity when used as the evaluation endpoint to assess the actual effect of each treatment. None of the tyrosine kinase inhibitor head-to-head studies included in this meta-analysis achieved a median OS. Therefore, clinicians still need to wait for the latest results to improve the OS rate. Additionally, patients with ALK gene rearrangement NSCLC might receive other treatment options, which hinder the interpretation of the OS; thus, PFS was reported as the primary outcome event in this study. Finally, as some trials remain in progress, complete data could not be included in the full analysis. More clinical trial results are needed to support further research.
Conclusion
In ALK gene rearrangement advanced NSCLC, lorlatinib was associated with the best PFS overall and was suitable for patients with or without brain metastases. Brigatinib was associated with the best PFS in Asian patients. Moreover, alectinib was the least toxic and ceritinib (750 mg, fasted) was associated with the most severe AEs. These findings provide further evidence for selecting appropriate ALK inhibitors in ALK gene rearrangement advanced NSCLC, but require validation in large randomized controlled studies.
Research Data
Research Data for First-line treatments for patients with advanced ALK gene rearrangements in NSCLC: a systematic review and network meta-analysis
Research Data for First-line treatments for patients with advanced ALK gene rearrangements in NSCLC: a systematic review and network meta-analysis by Jiahao Tao, Chuangjie Zheng, Cuifen Zhang, Ling Zhou, Zeyu Liu, Yanqun Zhou, Xuewu Huang, Lizhu Lin and Linzhu Zhai in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221132703 - Supplemental material for First-line treatments for patients with advanced ALK gene rearrangements in NSCLC: a systematic review and network meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605221132703 for First-line treatments for patients with advanced ALK gene rearrangements in NSCLC: a systematic review and network meta-analysis by Jiahao Tao, Chuangjie Zheng, Cuifen Zhang, Ling Zhou, Zeyu Liu, Yanqun Zhou, Xuewu Huang, Lizhu Lin and Linzhu Zhai in Journal of International Medical Research
Footnotes
Acknowledgments
Author contributions
Conception and design: Jiahao Tao and Cuifen Zhang; Administrative support: Linzhu Zhai; Provision of study materials: Ling Zhou, Jiahao Tao and Cuifen Zhang; Collection and assembly of data: Chuangjie Zheng and Yanqun Zhou; Data analysis and interpretation: Zeyu Liu, Jiahao Tao and Cuifen Zhang; Manuscript writing: all authors; Final approval of manuscript: all authors.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt (pending publication) of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the National Natural Science Foundation of China (grant No. 81873147), the Beijing Xisike Clinical Oncology Research Foundation (grant No. Y-2020Sciclone/zb/qn-0005), and ‘Double first-class’ and high-level university construction project of Guangzhou University of Chinese Medicine (grant No. A1-2601-22-415-014).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
