Abstract
Objectives
Early mobilisation and effective pain management after open nephrectomy for renal cell carcinoma often include epidural analgesia (EDA), requiring an infusion pump and a urinary catheter, thus impeding mobilisation. Spinal anaesthesia (SpA) may be an alternative. This randomised clinical trial evaluated whether SpA improves analgesia and facilitates mobilisation over EDA and which factors influence mobilisation and length of stay (LOS).
Methods
Between 2012 and 2015, 135 patients were randomised and stratified by surgical method to either SpA with clonidine or EDA. Mobility index score (MobIs), pain scale, patient satisfaction questionnaire, and LOS were the main outcome measures.
Results
SpA patients exhibited an increase in MobIs significantly earlier than EDA patients. Among SpA patients >50% reached MobIs ≥13 by postoperative day 3, while 29% of EDA patients never reached MobIs ≥13 before discharge. SpA patients had higher maximum pain scores on postoperative days 1 and 2, but both groups had similar patient satisfaction. One day before discharge, 36/64 SpA versus 22/67 EDA patients (56% and 33%, respectively) were opioid-free. SpA patients were discharged significantly earlier than EDA patients.
Conclusions
SpA facilitates postoperative pain management and is associated with faster mobilisation and shorter LOS.
The trial was registered at ClinicalTrials.org (ID-NCT02030717).
Keywords
Introduction
The mainstay of renal cell carcinoma (RCC) therapy consists of surgery comprising either radical nephrectomy or nephron-sparing surgery.1,2 A key determinant for the postoperative course is early mobilisation and effective pain management. Modern multimodal approaches for peri-operative care in the case of open kidney surgery often include epidural analgesia (EDA) via a catheter. With a neuraxial site of action, EDA provides good postoperative pain alleviation while avoiding parenteral opioids during use and is one of the cornerstones of modern peri-operative care.3–5 However, EDA requires the patient to be connected to an infusion pump and most often an indwelling urinary catheter, which both might impede postoperative mobilisation.
Spinal anaesthesia (SpA) is a well-established method for postoperative pain management after lower laparotomy. 6 Application of this technique for postoperative analgesia after open nephrectomy in the upper abdomen requires a level of spinal anaesthesia up to Th4 and produces a prolonged analgesic effect. The use of an adjunct, such as clonidine, for single-shot spinal anaesthesia has been shown to significantly prolong postoperative analgesia.6,7 SpA thus has the potential to effectively alleviate pain until pain management can be successfully continued with oral medication.
We previously showed that SpA patients were associated with a decreased length of stay (LOS) after open surgery for RCC. 8 In this study, we aimed to further evaluate the hypothesis that SpA improves analgesia and facilitates mobilisation compared with EDA. Because there is no instrument in the literature measuring the postoperative mobility, as opposed to that for postoperative recovery, of non-intensive care unit patients, a mobility index score (MobIs) was designed and used for the analyses (Table 1). Our designed score used variables describing mobility and its limiting factors, where mobility was defined as the ability to move freely or the ability to move physically. In general, we aimed to determine which peri-operative factors influenced mobilisation, pain, and LOS by comparing RCC patients randomised to either SPA with clonidine or traditional EDA.
Mobility index scores (example patient), translated from Swedish.

POD, postoperative day; NRS, numeric rating scale. Scores for question 1: NRS (8–10) = 0 points, NRS (6–7) = 1 point, NRS (4–5) = 2 points, NRS (2–3) = 3 points, NRS (0–1) = 4 points.
Materials and methods
The method used has previously been described. 8 A total of 191 patients with kidney tumours were scheduled for open surgery between 2012 and 2015 at the Department of Urology, Umeå University Hospital, Umeå, Sweden. After signed informed consent was collected by the urologist, 135 patients were eligible. These 135 patients were randomised by the anaesthesiologist using a web-based minimisation randomisation method to either SpA or EDA, stratified according to the method of surgery: nephron-sparing surgery via a flank incision or radical nephrectomy via a transverse incision in the upper abdomen (Figure 1).

Flow chart of patient enrolment.
The study was approved by the Ethical Committee of Northern Sweden (Dnr 08-096M) and the Swedish Medical Products Agency (Dnr 151:2011/22306, Eu-nr 2008-001491 • 77). The trial was registered as a randomised controlled trial (RCT) at ClinicalTrials.org (ID NCT02030717).
Patients in the SpA group received a single-shot spinal injection at the L2-3 or L3-4 interspace (block level Th4) with a mixture of 12 mg of hyperbaric bupivacaine, 160 µg of intrathecal morphine, and clonidine (45 µg, 60 µg, or 75 µg for patients aged >85 years, 60–85 years, or <60 years, respectively). After 10 minutes in the Trendelenburg position, the patient was positioned for surgery. In the EDA group, an epidural catheter was inserted at the Th8-9 or Th9-10 interspace, and a mixture of plain bupivacaine (1 mg/mL), fentanyl (2 µg/mL), and adrenaline (2 µg/mL) was continuously administered with an infusion pump. All patients were endotracheally intubated, on controlled ventilation, and received balanced general anaesthesia with sevoflurane and fentanyl. All other peri-operative routines including monitoring, blood pressure support, and fluid administration were similar in both groups and followed the local departmental standard.
Postoperatively, neuraxial pain management was supplemented with paracetamol in both groups. SpA patients also received oral oxycodone as needed, from postoperative day (POD) 0. For EDA patients, the EDA infusion rate was adjusted postoperatively as needed to optimise analgesia. Once pain control allowed, the epidural infusion was replaced by oral oxycodone as needed. Patients in both groups received intravenous morphine as rescue medication.
All patients were offered nutritious fluids postoperatively on the day of surgery (POD 0) and solids on POD 1. Mobilisation was encouraged and assisted according to a goal-specified plan. Patients were discharged when oral pain medication was considered sufficient and when they were mobilised, i.e., walked in the corridor, tolerated meals, and felt capable of taking care of their physical needs at home by themselves.
Our primary outcome, degree of postoperative mobilisation, was measured by the patients using the self-registered MobIs, based on 10 closed questions and three Numeric Rating Scale (NRS) questions. A diary was completed by all patients once a day. On the basis of a pilot study and an iterative process, a weighted index with scores ranging from 0 to 16 points was composed.
For further quality evaluation of the studied anaesthesia methods, we devised a questionnaire for assessment of patient satisfaction regarding different aspects of their hospital stay. A similar, iterative process to that described for development of the MobIs was used. A literature review aided in selecting the type of phrasing and construction of answer possibilities. One factor of relevance and analysed for the scope of this study was patient satisfaction with pain management (Table 2). The questionnaire was answered at discharge.
Patients’ satisfaction with pain management at the time of discharge from the hospital.
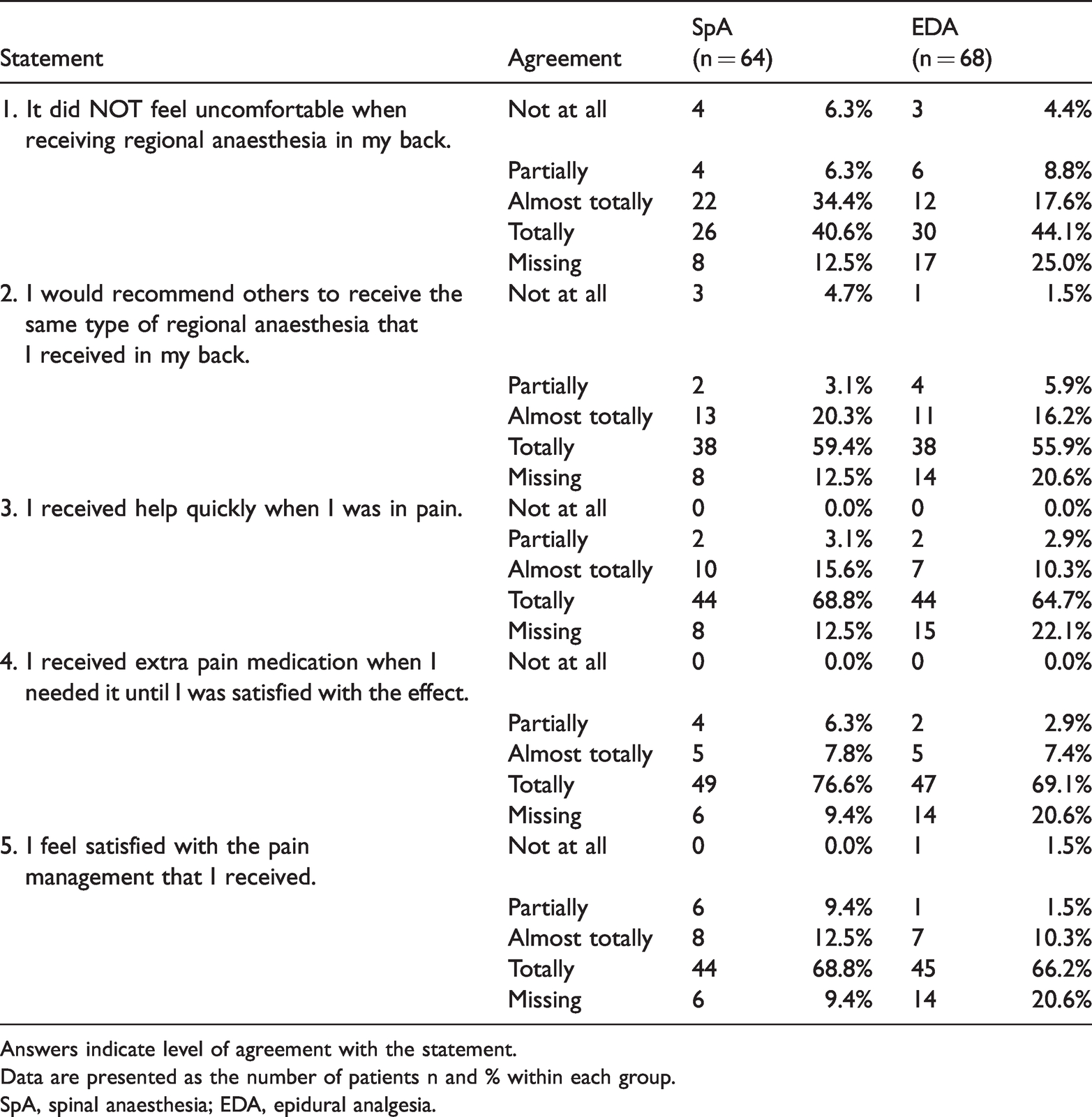
Answers indicate level of agreement with the statement.
Data are presented as the number of patients n and % within each group.
SpA, spinal anaesthesia; EDA, epidural analgesia.
The reporting of this study conforms to the CONSORT statements 9
Statistical analysis
The power analysis was based on a pilot study analysing the MobIs on POD 3. To achieve adequate statistical power (0.8) and significant results (alpha 0.05) and to confirm or reject the primary hypothesis that the SpA group would achieve a MobIs 2 points higher than that in the EDA group, a minimum of 38 patients in each group was needed.
IBM SPSS, Version 24.0 (IBM Corp., Armonk, NY, USA) was used for the statistical analysis. The χ2 test and Mann–Whitney U-test were used for comparisons between groups, and logistic regression modelling was used to explore relationships between variables. Variables selected for multiple regression were chosen on the basis of clinical significance and preselected by univariate analyses before inclusion in the multivariate analysis model.
For pain analysis in the multiple regression, two compound variables were created reflecting different aspects of pain. “Worst pain by POD 3” captured the single worst pain experience on POD 1, 2, and 3. “Cumulative pain by POD 3” was calculated by adding the NRS scores for “worst pain during movement over the last 24 hours” for POD 1, 2, and 3.
The distribution of the MobIs of all patients on the day before discharge was used as the grouping criterion. A MobIs ≥13 was considered as a valid cut-off level for analysis.
Results
The patient characteristics (Table 3) show an even distribution between the anaesthesia groups. As there were only minor differences between the intention-to-treat analysis and analysis according to the given treatment, we chose to report the results according to the given treatment only (Figure 1). We observed no harmful effects with regard to either method of anaesthesia employed.
Patient characteristics of the spinal anaesthesia group (SpA) and epidural anaesthesia group (EDA).
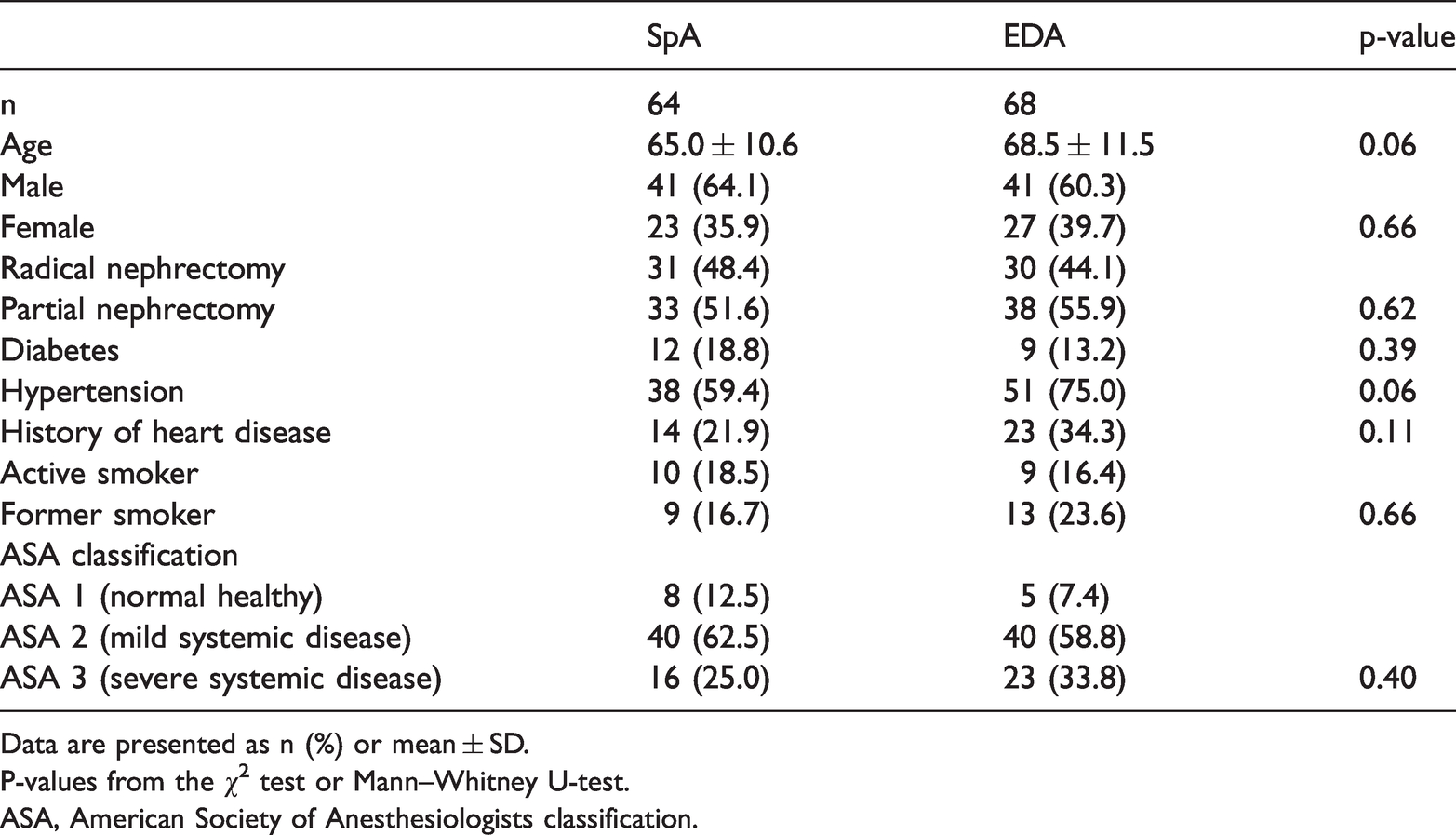
Data are presented as n (%) or mean ± SD.
P-values from the χ2 test or Mann–Whitney U-test.
ASA, American Society of Anesthesiologists classification.
Differences in the MobIs between groups are depicted in Figure 2. Patients in the SpA group reached significantly higher MobIs than patients in the EDA group on POD 3 to 5 (Figure 2). More than 50% of SpA patients had reached MobIs ≥13 on POD 3 (POD mean 2.98, 95% confidence interval (CI) 2.67–3.30), while half of the EDA patients first reached MobIs ≥13 on POD 5 (POD mean 4.50, 95% CI 3.87–5.13, p < 0.001). In the EDA group, a significantly greater number of patients never reached MobIs ≥13 before discharge compared with those in the SpA group (29.2% vs. 14.3%, respectively, p = 0.002) (Figure 3).

Mobility index on POD 1–6 and on the day of discharge.
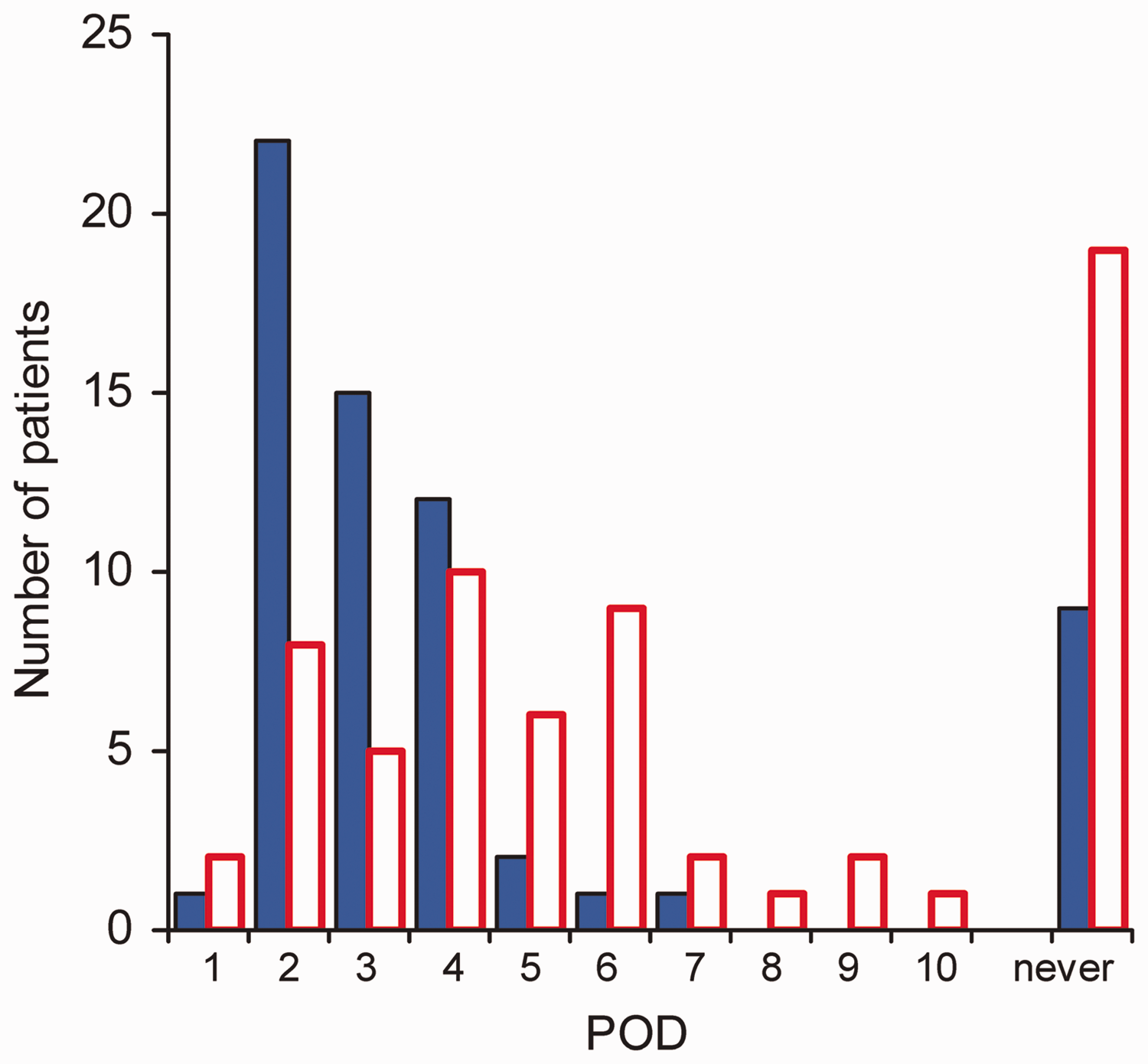
Number of patients with mobility index ≥13 on the respective postoperative day (POD).
Patients with SpA had higher maximum pain scores for movement on POD 1 and 2 than those of EDA patients (mean 4.86 (95% CI 4.13–5.59) and 2.76 (95% CI 2.17–3.36), respectively, p < 0.001). There was no difference in the NRS between the groups for the remaining days of the hospital stay as shown in Figure 4.

Numeric rating score on POD 1–6 and on the day of discharge.
In contrast to pain scores, most patients in both groups were almost or totally satisfied (>90%) with their pain management from POD 1 to discharge. Both SpA and EDA patients would recommend the type of neuraxial block they were allotted to others. There were no differences between groups with regard to the different aspects of satisfaction with pain management or other aspects of patient satisfaction (Table 2).
As per the study design, patients in the SpA group were given oral analgesics from POD 0, while most patients in the EDA group were first given oral analgesics after discontinuation of EDA (mean EDA use 4.32 days, 95% CI 3.85–4.79). There were no differences in overall total oxycodone consumption during the hospital stay (SpA mean 75.8 mg (95% CI 52.8–98.9) vs. EDA mean 52.9 mg (95% CI 39.2–66.5)). Additionally, paracetamol was used in similar amounts by the patient groups during the hospital stay. Pain management problems requiring involvement of anaesthesiologic pain service were more frequent in the EDA group (odds ratio (OR) 3.45, SpA 9.4% vs. EDA 32.4%, p = 0.001). Logistic regression analysis showed a five-fold higher likelihood for patients with pain management problems to be discharged on POD 6 or later (OR 5.16, 95% CI 1.82–14.62, p = 0.002).
Among the 14 patients discharged by POD 3, 12 had received SpA (85.7%). Furthermore, 7 (58.3%) of those 12 SpA patients had not required oxycodone on the day before discharge. One day before discharge, 36 of 64 SpA patients (56.3%) were oxycodone-free compared with 22 of 67 EDA patients (32.8%, p = 0.007). Concurrently, SpA patients had a significantly shorter LOS than that of EDA patients (p = 0.001).
According to the multiple logistic regression, the anaesthesia method only predicted achieving a MobIs ≥13 on POD 3. Patients treated with SpA had an OR of 4.87 (95% CI (2.27–10.45), p < 0.001) of reaching a MobIs ≥13 on POD 3. Patients who reached a MobIs ≥13 on or before POD 3 had an almost five-fold greater likelihood to be discharged no later than POD 5 compared with EDA patients. Pain scores had no influence on the LOS. Postoperative complications of any sort, which were evenly distributed across both methods of anaesthesia and types of surgery, increased the likelihood of being discharged on POD 6 or later by more than 10-fold (Figure 5). Nephron-sparing surgery was also associated with a shorter LOS (Table 4).

Multivariate logistic regression with LOS as the dependent variable, final model. Predictive value of this model, 74.6% and Nagelkerke R-squared, 0.325.
Univariate logistic regression for length of stay.

POD, postoperative day; NRS, numeric rating scale; MobIs, mobility index score; NSS, nephron-sparing nephrectomy; RN, radical nephrectomy; SpA, spinal anaesthesia; EDA, epidural analgesia; F/M, female/male; ASA, American Society of Anesthesiologists.
Discussion
In this RCT, patients treated with SpA with clonidine during open surgery for RCC had significantly faster mobilisation than patients receiving EDA. We found that faster mobilisation was one of the main factors that accounted for the difference in LOS between the anaesthesia groups. 8 The positive influence of SpA on mobilisation, as shown by the significantly higher MobIs achieved earlier during the hospital stay, supports our hypothesis that faster mobilisation is an underlying cause of earlier discharge.
Because there was no instrument available to measure postoperative mobility in non-intensive care unit patients after abdominal surgery in the literature, the MobIs was constructed for this study. 10 We found an association between the MobIs and LOS. Because of a lack of instruments to measure mobility, no comparison with other studies was possible. In the present study, factors associated with a shorter LOS included the presence of an indwelling urinary catheter and whether the patient was able to eat in the dining room of the care unit. Validation of our instrument (MobIs) for evaluating postoperative mobility after abdominal surgery is warranted.
In our study, we noted a discrepancy between the groups regarding pain scores and patient satisfaction. Patients in the SpA group had higher NRS pain scores than patients in the EDA group on the first two PODs, while patient satisfaction with pain management at the same time was equally high in both groups. It should be noted that we studied the incidence of maximal pain during movement on the respective POD. However, the mean maximal pain score for SpA patients in the present RCT was 4.86 on both POD 1 and 2, and this score was only one point above an accepted average pain threshold and quite in line with pain in movement scores previously reported after nephrectomy.11,12 Gerbershagen et al described a higher pain tolerance for the worst pain both with regard to the desire for additional analgesics as well as patient satisfaction with pain therapy, which aides in explaining the discrepancy that we observed. 11
In an RCT with a study design similar to ours that compared SpA with morphine and clonidine as additives and EDA in women after laparotomy for gynaecological malignancy, the authors found a similar phenomenon on POD 0 and 1, with a higher NRS score during movement, as observed in the SpA patients in the present study. 13 In their study, they did not observe an overall difference in NRS scores between the analgesia groups. In line with our results, they also reported a significantly shorter LOS in the SpA group. 13
In a cross-sectional study including more than 9000 patients, van Boekel et al found that the relationship between pain scores during movement and patient opinion of acceptability of pain or nurse assessment of movement appropriate for recovery is not as straight forward as might be assumed. 14 They concluded that pain management should not be guided by pain scores alone. In our study, patients in the SpA group were equally as satisfied with their pain management and mobilised faster despite higher pain scores. Furthermore, pain variables did not influence the LOS in the logistic regression analysis, except for pain management problems in the ward. Such management problems did lengthen the LOS and were more frequent in the EDA group. Management problems may also play a significant role in how patients experience pain management. This aspect warrants further exploration.
Mimic et al explored psychological factors related to postoperative pain after nephrectomy. 15 Their findings corroborate our findings that EDA achieves superior NRS scores compared with other modes of pain management in the early recovery phase. After POD 3, they found that psychological factors, specifically anxiety, pain expectancy, and pain catastrophising, superseded the type of pain management as predictors of pain intensity as measured by the NRS. However, they also found that a sizable variability remained among the variables that was not captured by their model and concluded, in accordance with our opinion, that pain scores fail to capture a patient’s pain experience in its entirety.
A study examining gynaecological laparotomies also reported less total opioid consumption in the SpA group than that in the EDA group. 13 This result corroborates our findings. The inclusion of patient satisfaction regarding pain management, in contrast to measuring NRS scores only, is an important step toward more fully capturing a patient’s pain experience. One further important result of the present RCT is that most SpA patients did not need oxycodone on the day before discharge, even though SpA patients on average had a shorter LOS. This analgetic improvement might possibly be a long-standing effect of clonidine added to SpA.
We chose, mostly for logistical reasons, to include all anaesthesiologists on urology rotation as patient responsible anaesthesiologists in our study. Their experience ranged from resident to experienced attending physician. While this general participation of different anaesthesiologists resulted in one patient not following protocol and thus created a crossover between groups, it also strengthened the generalisability of the results as it reflects the typical clinical situation in many hospitals. We did, for example, note a variation in technique for spinal anaesthesia that mainly included not always adhering to the protocol for achieving a block up to the T4 level, which, in some cases, might have been responsible for less optimal SpA. This makes our results more clinically robust and applicable to any routine clinical setting and thus improves the clinical value of our study. Among the limitations, we used a new mobility index (MobIs) designed for the study. However, the MobIs also contributed an important strength to our study and was useful for patient outcome measures.
Conclusion
Peri-operative pain management using SpA with clonidine is an important alternative in the pain management of patients treated with open surgery for RCC. While employing this strategy using SpA with clonidine, patients benefited from easier pain management in the ward, faster mobilisation, a shorter LOS, and less need for opioids at discharge.
Footnotes
Acknowledgements
The authors thank Kerstin Almroth for skilful technical assistance with the study.
Author contributions
Study concept and design: Thurm and Ljungberg.
Acquisition of data: Thurm, Kröger Dahlin, Johansson, and Ljungberg.
Analysis and interpretation of data: Thurm, Hultin, Johansson, Kröger Dahlin, Winsö, and Ljungberg.
Drafting of the manuscript: Thurm and Ljungberg.
Critical revision of the manuscript for important intellectual content: Thurm, Hultin, Kröger Dahlin, Johansson, Winsö, and Ljungberg.
Statistical analysis: Thurm, Johansson, and Ljungberg.
Obtaining funding: Hultin and Ljungberg.
Administrative, technical, or material support: None.
Supervision: Hultin, Winsö, and Ljungberg.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from Umeå University, Västerbotten County, and through a regional agreement between Umeå University and Västerbotten County Council on cooperation in the field of Medicine, Odontology and Health.
