Abstract
Objective
This study aimed to describe our experience of managing cesarean scar pregnancy (CSP) and outcomes depending on ultrasound imaging features.
Methods
A retrospective, cohort observational study was performed on 31 consecutive patients with CSP at 6 to 9 weeks of gestation from April 2015 to January 2021. All patients were evaluated for the residual myometrial thickness (RMT), growth direction of the gestational sac (GS), blood flow, and chorionic parenchyma using ultrasonography. Patients underwent curettage or methotrexate (MTX) combined with curettage in CSP depending on the age of the GS. Blood loss of >500 mL with curettage was considered major bleeding.
Results
Twenty-five (80.6%) patients had successful treatment, and six (19.4%) patients had major bleeding. The incidence of major bleeding was significantly higher in patients with >7 weeks of gestation, types II and III CSP, mixed and exogenous types of the growth direction of the GS, an RMT < 2 mm, and multiple lacunae formation in thickened chorionic parenchyma.
Conclusions
The exogenous and mixed types of the GS, an RMT < 2 mm, and multiple lacunae in thickened chorionic parenchyma may be high-risk factors for major hemorrhage by curettage in CSP.
Introduction
Cesarean scar pregnancy (CSP) is a long-term complication of cesarean section, and it is caused by the implantation and development of a fertilized egg in the scar site of a cesarean section. Improper diagnosis and treatment of CSP can lead to serious complications, such as major bleeding and uterine rupture.1–4 The previously excessive high rate of cesarean delivery due to social factors and the implementation of the national second-child policy in recent years has led to a rapid increase in the incidence of CSP in China.5–9 The main treatment of CSP is to terminate the pregnancy because of the serious consequences. There are many methods to treat CSP such as curettage. However, the principles of treatment are focused on saving lives, minimizing complications, and trying to preserve reproductive function.1–9 Ultrasound is the first choice for the diagnosis of CSP, and it also plays an important role in guiding the development of the treatment plan.
It should be noted that there is a lack of systematic research on the ultrasonic characteristics of CSP, and there is no recognized ultrasonic classification standard. Therefore, this study aimed to examine high-risk ultrasonic manifestations and ultrasonic classification of CSP to guide the clinical development of a reasonable treatment plan, and further reduce the complications of CSP.
Methods
Patients
A retrospective, cohort observational study was performed on 31 pregnant women with clinically diagnosed CSP at the Obstetrics and Gynecology Department who were continuously admitted to Beijing Tiantan Hospital. They were treated with curettage or curettage combined with methotrexate (MTX) from April 2015 to January 2021. The study was performed in Tiantan Hospital, Capital Medical University, Beijing, China, which is a tertiary academic hospital. The gestational age at the initial diagnosis was 6 to 9 weeks. The patients with CSP at ≤7 weeks-of gestational age underwent ultrasound-guided curettage. The patients with CSP at >7 to 9 weeks of gestational age underwent ultrasound-guided injections of MTX (1 mg/kg) twice by cervical and/or intragestational sac injection with a 1-week interval. The ultrasound-guided curettage was performed 1 week after the two MTX injections. Serum beta-human chorionic gonadotropin (β-HCG) concentrations were measured before and after MTX treatment. A blood loss of >500 mL associated with curettage was considered major bleeding. Complete clearance of conception products without major bleeding was considered successful curettage treatment. After the curettage, the patients’ serum β-HCG concentrations, pelvic ultrasonography, and menstruation were followed up until normal, and the follow-up time was 2 to 3 months after curettage. MTX was contraindicated for patients with severe liver and kidney diseases and blood diseases, and CSP of the exogenous type was contraindicated for curettage. All patients who were informed and counselled about the treatment options and potential risks signed an informed consent form. The reporting of this study conforms to the STROBE guidelines10 in the EQUATOR Network guidelines (https://www.equator-network.org/). Institutional Review Board approval was waived owing to the nature of the retrospective study. The privacy rights of human subjects were always observed. All patients’ details have been de-identified in the study.
Ultrasonographic detection
All patients underwent an ultrasound examination with a General Electric ultrasound system (LOGIQ E9 ultrasound system; GE Healthcare, Waukesha, WI, USA). The frequency of the convex array probe was 4.0 to 5.5 MHz, and the frequency of the vaginal probe was 5 to 8 MHz. All patients underwent transvaginal combined with transabdominal ultrasonography to observe the size and shape of the uterus, the residual myometrial thickness (RMT) in the cesarean scar, the implantation site and the direction of growth of the gestational sac (GS), the blood flow in the scar area, and the chorionic parenchyma. The gestational age was estimated comprehensively on the basis of the time of cessation of menstruation, the size of the GS, the development of the embryo, and serum β-HCG concentrations.
The diagnosis of CSP was based on the accepted criteria in the literature.1–9 These criteria were no GS in the uterine cavity, the GS was attached to the anterior wall of the uterine isthmus and protruded into the uterine cavity or expanded to the bladder, and abundant blood flow with a high-velocity and low-resistance blood flow frequency spectrum was detected in the chorionic tissue. Depending on the RMT and the growth direction of the GS, the Chinese experts’ consensus classification criteria of CSP, issued by the Division of Obstetrics and Gynecology of the Chinese Medical Association in 2016, are as follows: type I, when the RMT is >3 mm and the GS grows into the uterine cavity; type II, when the RMT is <3 mm and the GS grows into the uterine cavity; and type III, when the RMT is <3 mm and the GS grows outside the serosa. 11
In this study, according to the positional relationship between the GS and uterine cavity and serosal line, the growth direction of the GS was divided into the following three types (Figure 1a–c). 1) In the endogenous type, the GS was completely located in the uterine cavity and the lower segment of the uterus did not bulge out of the serosa. 2) In the mixed type, most of the GS was located in the uterine cavity and the lower segment of the uterus bulged out of the serosa. 3) In the exogenous type, the GS was almost completely located in the lower segment of the uterus and bulged out of the serosa. Two senior physicians who had worked for more than 15 years consulted the ultrasound images of 31 patients with CSP and classified them in accordance with the above-mentioned criteria.

Schematic diagram of trichotomy of the growth direction of the gestational sac in cesarean scar pregnancy in ultrasound images. (a) Endogenous type of gestational sac. The endogenous type is completely located in the uterine cavity, and the lower segment of the uterus does not bulge out of the serosa. (b) Exogenous type of gestational sac. The exogenous type is almost completely located in the lower segment of the uterus and bulges out of the serosa. (c) Mixed type of gestational sac. In the mixed type, most of the gestational sac is located in the uterine cavity, and the lower segment of the uterus bulges out of the serosa. The white dotted line indicates the contour of the uterine serosa, and the yellow dotted line indicates the contour of the gestational sac.
Statistical analysis
SPSS 20.0 statistical software was used to analyze the data (IBM SPSS for Windows, IBM Corp., Armonk, NY, USA). Measurement data with a continuous and normal distribution data are expressed as the mean ± standard deviation, and were compared using the t-test. With regard to nonparametric data, enumeration data are expressed as the rate (%) and were compared using the chi-square test. A P value <0.05 was considered statistically significant.
Results
Thirty-one patients with CSP were included in the study. A flowchart of patients in the study is shown in Figure 2. The patients had a mean age of 33.8 ± 5.6 years (26–47 years), a mean number of 1.2 ± 0.4 cesarean deliveries (1–3 times), and a mean interval of 6.2 ± 3.1 years (1–12 years) between the current pregnancy and the previous cesarean delivery. The mean initial serum β-HCG concentration was significantly higher in patients with CSP at >7 to 9 weeks of gestational age who underwent ultrasound-guided injections of MTX followed by curettage than in those with CSP at ≤7 weeks of gestational age who underwent ultrasound-guided cutterage (P < 0.001, Table 1). However, there was no significant difference in the mean serum β-HCG concentration before curettage between the two groups of patients.
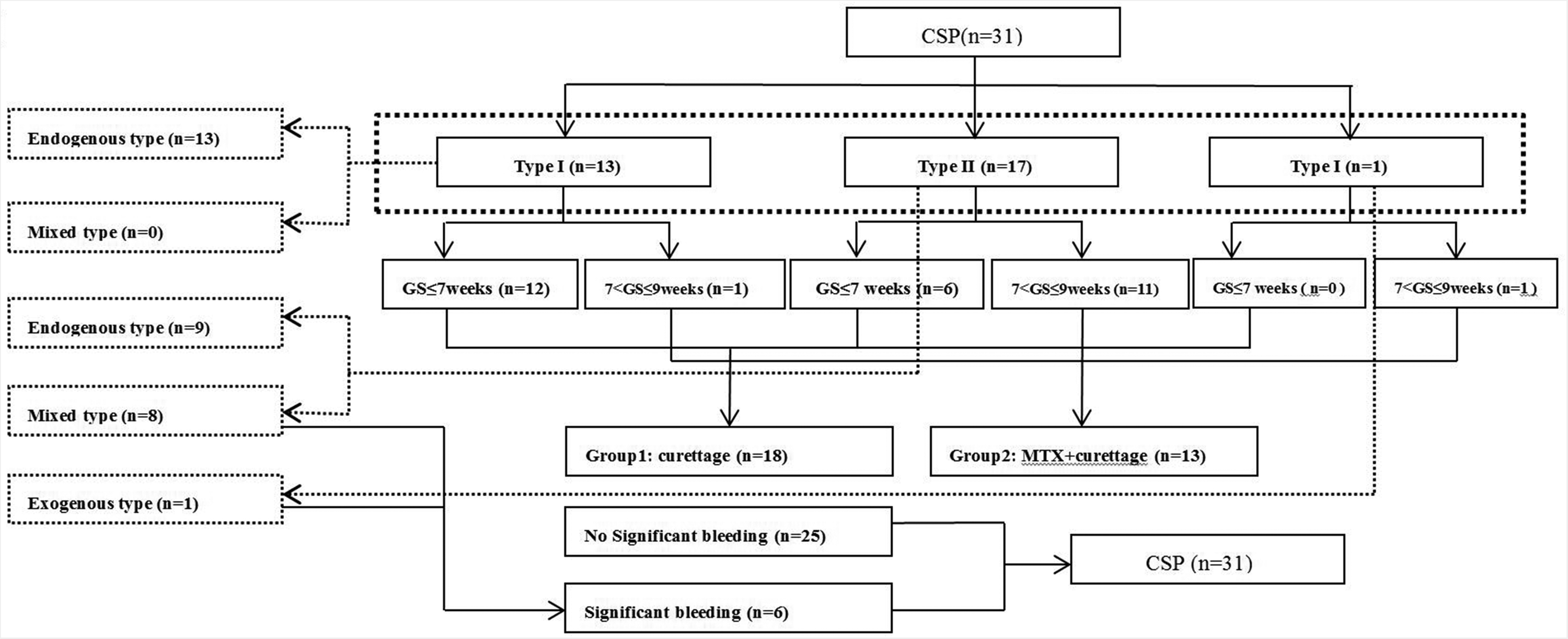
Flowchart of the study.
Serum β-HCG concentrations in 31 cases of cesarean scar pregnancy.

β-HCG, beta-human chorionic gonadotropin.
Among thirty-one patients, 25 (80.6%) had successful curettage, with 12 cases of type II and 13 cases of type I CSP (Figure 3a–c). Six (19.4%) patients had major hemorrhage after curettage, with five cases of type II and one case of type III. Among 17 cases of type II (8 cases of the mixed type and 9 cases of the endogenous type of GS), 5 of 8 cases with the mixed type of GS had major hemorrhage after curettage. Six cases of CSP with major hemorrhage were treated as follows. One case of type III was cured by an open surgery of wedge resection of the lesion (Figure 4a–c). One case of type II was cured by uterine artery embolization, and the other four cases of type II were cured conservatively by the administration of uterotonic drugs and/or catheter balloon compression (Figure 5a–c). In our study, the incidence of major bleeding in patients with the endogenous type, mixed type, and exogenous type was 0%, 62.5%, and 100%, respectively. The incidence of major bleeding after curettage was significantly higher in patients with a gestational age of >7 weeks, types II and III CSP, mixed and exogenous types in the growth direction of the GS, and an RMT < 2 mm (all P < 0.05, Table 2). Multiple lacunae in thickened chorionic tissue at the lower segment of the anterior wall of the uterus were observed in eight patients with type II CSP with the mixed type of GS at a gestational age of 7 to 9 weeks. Five of these patients had major hemorrhage after curettage.

(a) Longitudinal view of the uterus on transvaginal B-mode ultrasound: A deformed gestational sac attached to the scar has grown into the uterine cavity completely, and the lower segment of the uterus does not bulge out of the serosa. (b) Transvaginal color Doppler ultrasound shows abundant blood flow in the chorionic tissue at the scar of the anterior wall of the uterus. (c) A magnified image of the scar at the lower segment of the anterior wall of the uterus. The residual myometrial thickness is 1.9 mm, the margin is clear between the myometrium and chorionic tissue, and there is homogeneous hyperechoic chorionic tissue.

Type III cesarean scar pregnancy with the exogenous type of gestational sac at 60 days of gestation. There was 2000 mL of blood loss after curettage and it was cured by an open surgery of wedge resection of the lesion. (a) Longitudinal view of the uterus on transabdominal B-mode ultrasound. A round gestational sac similar to a bagel is located in the lower segment of the uterus. The lower segment of the uterus is thickened and extrudes outward (white arrow), and there are multiple lacunae (open arrow) in thickened chorionic tissue. (b) Longitudinal view of the uterus on transabdominal color Doppler ultrasound. Rich circular blood flow in thickened chorionic tissue can be seen. (c) Transvaginal B-mode ultrasound image of the patient's non-pregnant uterus. A fissure-like niche of the cesarean scar approximately 8.1-mm depth in the cesarean scar can be seen.
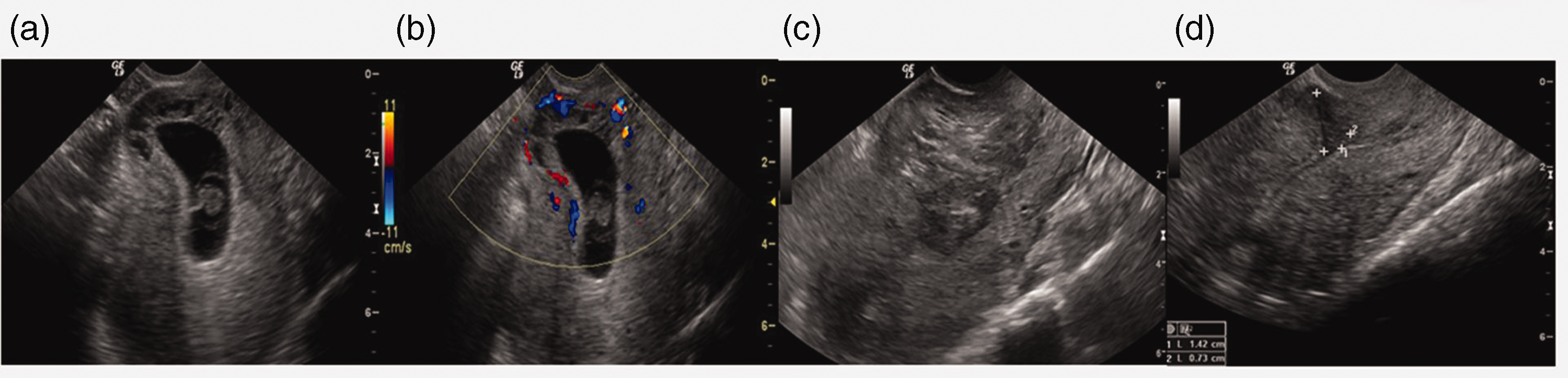
Type II cesarean scar pregnancy with the mixed type of gestational sac at the gestational age of 7 weeks and 4 days. There was blood loss of 500 mL after curettage, which was cured by conservative medical treatment (a) Transvaginal B-mode ultrasound shows that most of the gestational sac is located in the uterine cavity, and the lower segment of the uterus is slightly thickened and expanded to the serosa. There are multiple lacuna in thickened chorionic parenchyma at the scar. (b) Transvaginal color Doppler flow shows abundant blood flow in the chorionic tissue at the scar of the anterior wall of the uterus. (c) After curettage, a hematoma formed in the diverticulum of the fossa of the uterine scar. (d) After absorption of the hematoma, the size and morphology of the uterus has returned to normal, and a deep fissure defect connected with the uterine cavity in the lower segment of the anterior wall is clearly shown.
Analysis of parameters associated with major hemorrhage.

*P(I,II) = 0.035; #P(II,III) = 0.157; &P(I,III) < 0.001.
*P(1,2) = 0.023; #P(2,3) = 0.254; &P(1,3) = 0.001.
I and II indicate type I and type II, respectively, in the Chinese experts’ consensus classification criteria of CSP, issued by the Division of Obstetrics and Gynecology of the Chinese Medical Association in 2016.
Discussion
Ultrasound classification of CSP based on high-risk indicators is important for making an appropriate treatment plan, which not only helps to reduce inappropriate curettage, but also avoids overtreatment. Regarding the classification of CSP, Vial et al 12 first proposed two types of CSP in 2000, namely GS implantation in the scar and on the scar. GS implantation in the scar has deep implantation of the GS in a cesarean scar defect with progression towards uterine rupture and hemorrhage during the first trimester.12–14 This condition is the predecessor of endogenous and exogenous CSP. Ultrasound classification of CSP has become an important indicator for complications because the growth direction of the GS is related to its prognosis. The growth direction of the GS has become an important indicator for subsequent ultrasound classification of CSP. In 2016, based on the growth direction of the GS and the RMT at the scar site of the uterus, China issued an expert consensus on CSP, which divided CSP into three categories of types I, II, and III.
In our study, there were 31 cases of CSP, with 13 cases of type I, 17 cases of type II, and 1 case of type III. Six cases were complicated by major bleeding after curettage, with one case of type III and five cases of type II. The only case of type III at 60 days of gestation was misclassified as the endogenous type in the growth direction of the GS. This patient suffered from massive hemorrhage owing to improper curettage and was finally cured by an open surgery of resection of the lesion. In this case of type III, ultrasound imaging showed that the upper edge of the GS reached the lower uterine cavity, similar to images of endogenous type CSP. However some features of the exogenous type of CSP were ignored by the sonographer, such as a round gestational sac similar to a bagel. Additionally, there was thickening of the lower uterine segment protruding outside the serosa due to the fertilized egg completely implanted in the deep muscular layer of the scar and it developed in situ. The endogenous type of CSP is characterized by a sac that is elongated and deformed with an acute angle at the lower area of the sac because of a fertilized egg implanted at the superficial surface of the scar growing into the uterine cavity. Furthermore, we examined the patient's previous ultrasound images of the non-pregnant uterus and observed a fissure-like niche of a cesarean scar as deep as 8.1 mm in the muscular layer of the uterine scar. This scar laid the foundation for the fertilized egg to be implanted in the deep muscle layer of the uterine scar and further develop into exogenous CSP.
Among 17 cases of type II CSP in our study, there were 9 cases of the endogenous type and 8 cases of the mixed type, and 5 cases of the mixed type had major hemorrhage after curettage. The incidence of major hemorrhage in patients with the endogenous type, mixed type, and exogenous type of the GS was 0%, 62.5%, and 100%, respectively. These findings suggest that the mixed and exogenous types are high-risk CSPs, and only low-risk cases of the mixed type of CSP are suitable for curettage to terminate pregnancy. The traditional dichotomy of the endogenous and exogenous types of the GS in CSP might lead to inconsistency among different sonographers who could over-type or under-type the classification, followed by over-treatment or inappropriate curettage. Therefore, replacing the existing dichotomy of endogenous and exogenous types in the growth direction of the GS with the trichotomy of endogenous, exogenous and mixed types may be appropriate.
Uterine bleeding is related to the RMT in the lower part of the anterior uterine wall during curettage. In 2016, the Chinese experts’ consensus classification used an RMT of 3 mm as the threshold for a high and low risk of CSP, while some studies used 1.5 mm or 2 mm as the threshold.14–16 In our study, the RMT was subdivided into the three groups of <2, ≥2 and <3, and ≥3 mm, and the incidence of major hemorrhage in each group was compared. The incidence of major bleeding associated with curettage was significantly higher in the <2-mm group than in the ≥2- to <3-mm or ≥3-mm groups, but there was no significant difference in the incidence of major bleeding between the latter two groups. The 12 cases of type II with successful curettage had an RMT of >2 mm or close to 2 mm. On the basis of the above-mentioned findings, we consider that the high- and low-risk threshold of RMT could be relaxed to 2 mm for curettage treatment in CSP, which may allow for more patients to be treated with curettage and reduce overtreatment and its complications. Notably, the RMT needs to be accurately measured after enlarging the ultrasonic image.
CSP has the pathological basis for placental implantation. CSP may progress into typical placental implantation during the middle or late trimester if left untreated. The typical ultrasound features of placental implantation in CSP are a thinning or loss of the lower myometrium of the uterine scar where the placenta is attached and marked thickening of the placental tissue with an obscure margin from the myometrium. Multiple placental lacunae are due to high blood pressure from the radial and arcuate arteries entering the intervillous spaces of the myometrium.17,18 In this study, a case of type III with the exogenous type of GS at a gestational age of 8 weeks and 4 days showed the above-mentioned ultrasound manifestations of placental implantation, and massive hemorrhage by curettage was inevitable. Multiple lacunae in thickened chorionic tissue at the lower segment of the anterior wall of the uterus were observed in eight patients with type II CSP with the mixed type of GS at a gestational age of 7 to 9 weeks. However, there was a clear boundary between chorionic tissue and the muscular layer, and five of these patients had major hemorrhage after curettage and were cured by UAE, local compression, or medical treatment. Therefore, multiple lacunae in thickened chorionic tissue may be a sign for predicting major hemorrhage by curettage, and UAE combined with curettage or resection of lesions may be more suitable in these patients than curettage. Surgical resection of the lesion should be considered when the chorionic tissue and muscularis are not clearly demarcated.
The main treatment options for CSP include medicine, local injection of MTX, curettage, UAE, hysteroscopy, laparoscopic lesion removal, and hysterectomy.19,20 To date, there is no consensus on the optimal treatment of CSP. UAE, laparoscopic lesion removal, and hysterectomy are invasive treatments for CSP. UAE is considered as a minimally invasive method of reducing major bleeding. However, UAE may lead to premature ovarian failure because it temporarily blocks blood flow of the uterine artery. 21 MTX alone has a high risk of bleeding in the treatment of CSP, and it is often used in combination with other methods. 22 Curettage is still considered as the primary method for the treatment of CSP owing to its advantages of a simple method, low cost, little trauma, and no effect on reproductive function. As gestation advances in CSP, the risk of bleeding increases during curettage. Therefore, pregnancy should be terminated as soon as possible after a diagnosis. The risk of major bleeding associated with curettage is relatively low in patients with CSP at 7 to 8 weeks of gestation. 23 Some scholars believe that patients with CSP within 7 weeks of gestation can be treated by curettage directly.24–28 In this study, some of the patients were initially diagnosed late, and a few were referred to our hospital, which led to more than half of the patients with a gestational age >7 weeks. To reduce the risk of massive hemorrhage, MTX was used in patients at >7 weeks of gestation before curettage. There was no significant difference in serum β-HCG concentrations between patients with CSP at >7 weeks of gestation after MTX treatment and those with CSP at ≤7 weeks of gestation. However, the incidence of major hemorrhage in patients with CSP at >7 weeks of gestation was significantly higher than that in those with CSP at ≤7 weeks of gestation. We consider that the decrease in chorionic tissue activity caused by MTX may be helpful to reduce the incidence of major hemorrhage during curettage, but it cannot completely prevent it.
In this study, a patient with type I CSP with a gestational age of 9 weeks showed a well-healed scar, and curettage was successful. The most appropriate time for curettage to terminate a CSP pregnancy is within the gestational age of 7 weeks, but the specific treatment plan for the individual should be determined by a comprehensive evaluation of various high-risk factors. In addition, when patients with cesarean section have amenorrhea or abnormal vaginal hemorrhage, ultrasound should be performed as soon as possible to exclude CSP.
A new type of modified consensus for CSP was published. 29 According to the location of the GS, the three types of CSP are as follows: (1) CSP with the largest part of the GS crossing the uterine cavity line; (2) CSP with the largest part of the GS embedded in the myometrium and not crossing the uterine cavity line, and the GS not crossing the serosal line; and (3) CSP crossing the serosal line. A comparison of our classification depending on the growth direction of the GS with the classification recommended in the new consensus shows that both classifications are trichotomous and based on the relationship between the GS and the uterine cavity line and serosal line. The new consensus types 1 and 3 represent the endogenous and exogenous types in our study, respectively. Additionally, the type of CSP may change with the progression of pregnancy. The new consensus type 2 may progress to the mixed type or exogenous type in our classification as gestational age advances. Therefore, our classification on the growth direction of the GS is partly consistent with the new consensus. Our classification may be more suitable for CSP at >7 weeks of gestation, and the new consensus classification may be more suitable for a CSP at 6 to 7 weeks of gestation.
There are several limitations to our study. First, only 31 pregnant women with CSP from a single center were included in this retrospective study. A larger number of cases needs to be included in the future. Because of the small number of cases, this study did not quantitatively analyze the extent of the lower uterine segment of CSP bulging out of the serosa. In addition, the classification standard that we proposed may be more suitable for CSPs at >7 weeks of gestation. The rationality of the classification that we proposed needs to be verified by expanding cases in the future. Furthermore, selection bias is a limitation. To reduce the selection bias, patients should be continually selected in multiple centers in the future.
In conclusion, trichotomy of endogenous, exogenous, and mixed types depending on the growth direction of the GS could be appropriate in CSP. The exogenous and mixed types in the growth direction of the GS, types II and III CSP, an RMT < 2 mm, multiple lacunae in thickened chorionic tissue, and >7 weeks of gestation may be high-risk factors for major bleeding by curettage in CSP.
Footnotes
Author contributions
Hong-xia Zhang, Dong-fang Wu, and Wen He conceived and designed the experiments. Xin Liu and Hai-man Song collected the data. Hong-xia Zhang and Dong-fang Wu analyzed the data and wrote the manuscript. Teng-fei Yu assisted with the data analysis. All authors have read and approved the final manuscript.
Data availability statement
We ensure that the original research data to support the results/tables/figures presented in the manuscript are available if requested.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
