Abstract
Objective
The current study aimed to investigate the application of contrast-enhanced ultrasound (CEUS) in diagnosis and treatment of cesarean scar pregnancy (CSP).
Methods
A retrospective study was performed in 35 patients with clinically suspected CSP who requested termination of pregnancy and underwent contrast-enhanced ultrasound (CEUS). The patients were classified into two groups on the basis of whether they received uterine artery embolization (UAE). The CEUS characteristics of the two groups were reviewed.
Results
CEUS features of CSP were early enhancement of the cesarean scar and continuous infusion of contrast agent between the gestational sac and cesarean scar. Myometrial thickness in the cesarean scar was thinner in the UAE group than in the non-UAE group by CEUS and transvaginal ultrasound. Myometrial thickness measured by CEUS was thinner than that measured by transvaginal ultrasound in both groups. The parameters of the time-intensity curve in the UAE group were characterized by a faster arrival time, shorter time to peak, higher peak intensity, and greater enhancement rate compared with the non-UAE group.
Conclusions
CEUS may be a novel supplementary method to diagnose and assess CSP, and to help evaluate whether UAE is required.
Keywords
Introduction
Caesarean scar pregnancy (CSP) is a rare type of ectopic pregnancy 1 in which the embryo is implanted at a cesarean scar. Trophoblasts invade the scar, leading to abnormal placental implantation, life-threatening hemorrhage, and even uterine rupture. 2 Over the past 20 years, the cesarean section rate has gradually increased worldwide, and is even as high as 46.2% in China. 3 With implementation of the two-child policy in China since 2015, the number of pregnant women with a history of cesarean section has rapidly increased, leading to a marked increase in the morbidity rate of CSP. 4
Transvaginal ultrasound (TVS) is the most effective and widely used method in clinical diagnosis of CSP. The sensitivity of CSP diagnosed by TVS is approximately 84.6%. 5 Nonetheless, differentiating CSP from intrauterine pregnancy and cervical pregnancy in some cases remains challenging.6,7 A false diagnosis may directly affect the clinical treatment. 8
There is no consensus for treatment of CSP. The main therapeutic management for CSP includes medicine, local injection of methotrexate, curettage, uterine artery embolization (UAE), hysteroscopy, laparoscopic lesion removal, and hysterectomy.9,10 UAE is considered as a minimally invasive method of controlling and preventing massive bleeding, and UAE followed by curettage is widely used in China as an effective method for CSP. However, UAE is an invasive procedure with some potential risk, and it is associated with complications, such as pain, fever, nausea, and vomiting, and it affects the patient’s reproductive future. Recent studies have suggested that not every CSP requires UAE and some patients are actually over-treated by UAE. 11 Therefore, there is concern about how to avoid excessive application of UAE.
Contrast-enhanced ultrasound (CEUS) allows for real-time and dynamic observation of perfusion of the microcirculation in tissue, which enhances blood flow of micro-vessels in lesions. CEUS has good application value in diagnosing gynecological diseases, such as uterine fibroids, adenomyoma, endometrial lesions, and adnexal tumors.12–14 Ordén et al. 15 reported application of CEUS in ectopic pregnancy and found that CEUS showed characteristic findings in early ectopic pregnancy. The current study aimed to evaluate the application of CEUS in diagnosing CSP and to compare features of CEUS in patients who underwent UAE. The findings from this study might reduce unnecessary excessive treatment for patients with CSP.
Methods
Patients
The protocol of this study followed the requirements of the Declaration of Helsinki and was approved by the Ethics Committee of Beijing Shijitan Hospital (2017, No. 34). This study was conducted in compliance with the EQUATOR Network guidelines (https://www.equator-network.org). Written informed consent forms were obtained from all patients.
This retrospective, observational study was performed in 81 patients with clinically suspected CSP at the Obstetrics and Gynecology Department, Beijing Shijitan Hospital, Capital Medical University from December 2017 to December 2018. All patients had a history of cesarean section (30 with 1 cesarean section, and 5 with 2 or more cesarean sections). The patients’ human chorionic gonadotropin (HCG) value was high (1219–149281 mIU/mL). Sixteen patients received UAE and 19 did not. All of these patients were classified into two groups on the basis of whether they received UAE. A flowchart of the study is shown in Figure 1.

Flowchart of the study participants. CSP, cesarean scar pregnancy; CEUS, contrast-enhanced ultrasound; UAE, uterine artery embolization; HCG, human chorionic gonadotropin.
The inclusion criteria were as follows: (1) patients with a history of cesarean section; (2) patients who terminated pregnancy; (3) patients without contraindications for TVS, such as agenesis of the vagina and concern of introducing infection; (4) the gestational sac (GS) was located in the lower uterine segment, as shown by TVS; (5) patients with elevated serum HCG levels; and (6) all patients signed an informed consent form after a detailed explanation.
The exclusion criteria were as follows: (1) patients with severe cardiovascular disease or lung disease; (2) patients with a contrast allergic history or other severe allergic history; (3) patients who refused to sign the informed consent; (4) patients who did not receive a CEUS examination; and (5) patients who were lost to follow-up.
The diagnosis of CSP was based on the following criteria: (1) an empty uterine cavity and cervical canal; (2) detection of a GS embedded in the cesarean scar; (3) a thin or absent myometrium between the GS and the bladder; and (4) the presence of abundant blood flow signals in the area of the cesarean scar.6–8 We identified two types of CSP as follows. 16 In endogenous CSP, the fertilized egg was implanted on the scar site, it grew into the uterine cavity, and it was partially located in the lower segment of uterine cavity. In exogenous CSP, the fertilized egg was implanted deeply in the cesarean scar, it grew in the direction of bladder and abdominal cavity, and it was completely located at the cesarean scar.
UAE was performed by interventional radiologists. The uterine artery was embolized with gelatin sponge particles (0.5–1.0 mm in diameter) through femoral artery intubation. 17 All patients were informed and counselled about the treatment options and potential risks. Assessment of the myometrial thickness as measured by TVS was an important factor in determining UAE treatment. Weilin and Liet al. 11 suggested that endogenous CSP with a myometrial thickness >3 mm might be treated via suction curettage. Therefore, indications for the treatment choice of UAE in this retrospectively study were as follows: (1) an endogenous CSP (myometrial thickness ≤3 mm); (2) an exogenous CSP; (3) massive bleeding, (4) a large GS, (5) high HCG levels, and (6) the patients’ own preference.18,19
Ultrasonographic detection
The LOGIQ E9 color Doppler ultrasound machine (GE Healthcare, Milwaukee, WI, USA) equipped with a transvaginal probe (frequency, 4–9 MHz; mechanical index, 0.06) was used. The contrast agent used was SonoVue (Bracco Imaging Company, Milan, Italy), which was diluted with 5 mL of 0.9% normal saline and mixed by vigorous shaking as a microvesicle suspension. A total of 2.4 mL of suspension was injected through the antecubital vein in a bolus (within 1–2 s), followed by flushing of 5 mL of 0.9% normal saline. The entire dynamic process of contrast agent perfusion was saved for later analysis. The dynamic images were diagnosed by two senior physicians with more than 5 years’ experience of gynecological ultrasound diagnosis. All cases were followed up for 2 months.
The TVS examination method of a CSP was as follows. The patient was asked to empty her bladder, the bladder lithotomy position was adopted, and then the uterus and bilateral adnexa were carefully examined through TVS. The position, size, and morphology of the GS were observed on the median sagittal plane of the uterus. The relationships of the GS with the cesarean scar and bladder were observed. The presence of the embryo and fetal heartbeat was observed and recorded. Additionally, local zoom ultrasound imaging was used to measure the myometrial thickness in the cesarean scar, and the average was calculated on the basis of three measurements. Measurement for myometrial thickness is shown in Figure 2.

Myometrial thickness as measured by transvaginal ultrasound (a) and contrast-enhanced ultrasound (b) in an endogenous cesarean scar pregnancy. The myometrial thickness measured by TVS (red line in a) is visibly thicker than that measured by contrast-enhanced ultrasound (red line in b).
Observational indices of CEUS were as follows: the site of initial enhancement, morphology of the GS, the mode of enhancement, and the presence or absence of continuous perfusion between the GS and the cesarean scar. The enhancement range of the lesion was evaluated. Regions of interest were drawn on the cesarean scar area and normal myometrium (1 cm above the caesarean scar) on an image that visually appeared to have obvious enhancement on CEUS. Time-intensity curves were generated by the ultrasonic instruments automatically, while the arrival time, time-to-peak intensity, and peak intensity of enhancement were calculated automatically by the software.
Statistical analysis
IBM SPSS version 19.0 software (IBM Corp., Armonk, NY, USA) was adopted for statistical analysis. Data with a continuous and normal distribution data are expressed as mean ± standard deviation, and were compared using the t-test. Data with a non-normal distribution are expressed by median and quartile. The rank sum test was used to compare parameters that did not conform to a normal distribution. Additionally, a diagnosis consistency test was performed. The area under the receiver operating characteristic curve was calculated. A difference of P < 0.05 was deemed as statistically significant.
Results
Pathological results
Thirty-five patients who underwent CEUS were finally enrolled and their mean age was 33.23 ± 4.07 years, with a mean gestational age of 6.62 ± 1.26 weeks. The demographic details of the patients are shown in Table 1. There were no significant differences in maternal age, gestational age, the number of cesarean sections, HCG levels, and the mean diameter of the GS between the two groups.
Demographic details of the 35 patients included in the study.

Data are mean ± standard deviation.
UAE, uterine artery embolization; HCG, human chorionic gonadotropin.
The pathological results and treatments for the patients are shown in Table 2. Among them, the volume of blood loss in the UAE group (25.3 ± 17.2 mL) appeared to be less than that in the non-UAE group (105.2 ± 282.1 mL), but this was not significant. One patient in the non-UAE group experienced active bleeding (1600 mL) during curettage, and this patient received intraoperative balloon compression hemostasis. One patient in the UAE group suffered from intermittent vaginal bleeding after curettage in another hospital. She had massive bleeding during curettage before the UAE procedure, the amount of bleeding was approximately 600 mL, and she underwent balloon compression hemostasis. None of the patients received blood transfusion. Upon surgical and (or) pathological verification, 24 patients were diagnosed with CSP (including 22 endogenous CSP and 2 exogenous CSP cases), and 11 did not have CSP (including 3 cervical pregnancies, 7 lower uterine pregnancies and 1 scar diverticulum). TVS misdiagnosed four lower intrauterine pregnancies and three cervical pregnancies as CSP. The sensitivity, specificity, positive predictive value, negative predictive value, Youden index, accurate diagnosis rate, Kappa value, and area under the curve of CEUS in diagnosing CSP were 95.8%, 81.8%, 92.0%, 90.0%, 77.6%, 91.4%, 0.796, and 0.888, respectively.
Pathological results and treatments of the patients in this study.
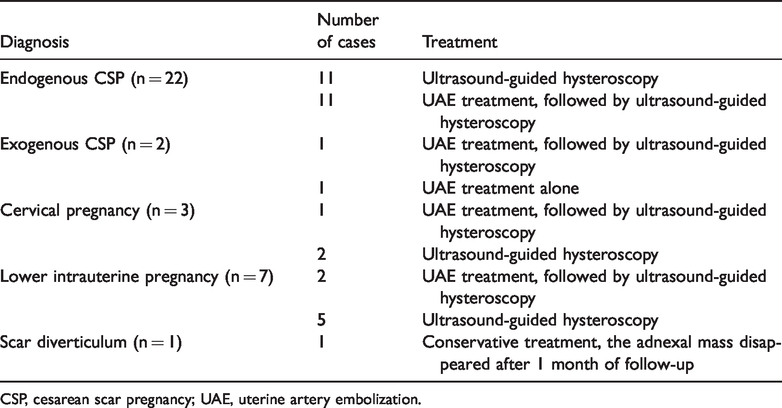
CSP, cesarean scar pregnancy; UAE, uterine artery embolization.
Features of CEUS in CSP
Comparison of CEUS time-intensity curve parameters between CSP and non-CSP cases is shown in Table 3. In CSP cases, the mean arrival time and time-to-peak intensity were significantly shorter than those in non-CSP cases (both P<0.001).The mean peak intensity and enhancement rate in CSP cases were significantly greater than those in non-CSP cases (both P<0.001) (Table 3).
Comparison of contrast-enhanced ultrasound time-intensity curve parameters between the CSP and non-CSP groups.

Data are mean ± standard deviation. CSP, cesarean scar pregnancy.
The features of CEUS in CSP were early enhancement of the cesarean scar (earlier than the myometrium) and continuous perfusion of the contrast agent was observed between the GS and the cesarean scar (Figure 3). Furthermore, myometrial thickness in the cesarean scar as measured by CEUS appeared to be thinner than that measured by TVS in the UAE and non-UAE groups (both P<0.001). Myometrial thickness was significantly greater in the non-UAE group than in the UAE group by TVS and CEUS (both P<0.01) (Table 4).

Contrast-enhanced ultrasound features of an endogenous cesarean scar pregnancy and exogenous cesarean scar pregnancy. (a) Early rapid enhancement of the scar (yellow arrows) in an endogenous cesarean scar pregnancy. (b) Continuous contrast agent perfusion between the gestational sac and the scar (yellow arrows) in an endogenous cesarean scar pregnancy. (c) Contrast-enhanced ultrasound demonstrating a ring-enhanced gestational sac in an exogenous cesarean scar pregnancy (yellow arrows).
Comparison of myometrial thickness in the cesarean scar between the UAE and non-UAE groups.

Data are mean ± standard deviation.
aComparison between the UAE and non-UAE groups; bP < 0.001 compared with TVS.
UAE, uterine artery embolization; TVS, transvaginal ultrasound; CEUS, contrast-enhanced ultrasound.
CEUS time-intensity curve parameters between the UAE and non-UAE groups
The CEUS time-intensity curve parameters in both groups are shown in Table 5, The mean arrival time and time-to-peak intensity were significantly shorter than those in non-UAE group (both P<0.01). Additionally, the mean peak intensity and enhancement rate in the UAE group were significantly greater than those in the non-UAE group (both P<0.05).
Comparison of contrast-enhanced ultrasound time-intensity curve parameters between the UAE and non-UAE groups.
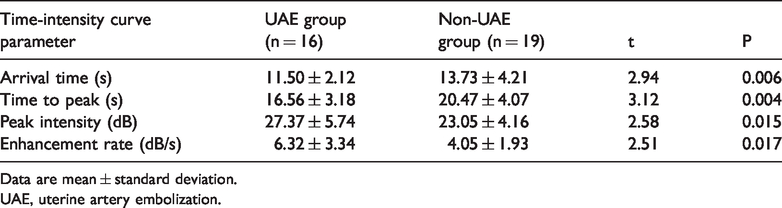
Data are mean ± standard deviation.
UAE, uterine artery embolization.
Discussion
CSP is mainly diagnosed using TVS combined with a previous history of cesarean section. With improvement in the understanding and diagnostic levels of clinicians, the misdiagnosis rate of CSP has been reduced. However, the resolution ability of TVS is still inconclusive in some CSP cases. Timor-Tritsch and Monteagudo 20 reviewed 751 cases of CSP, and found that 13.6% (107/751) of these cases had been misdiagnosed as cervical pregnancies, spontaneous abortions, or low intrauterine pregnancies. In some intrauterine pregnancies, the GS fills the cesarean scar diverticulum and the space between the GS and the scar disappears, which results in a false diagnosis of CSP. In our study, TVS misdiagnosed four lower intrauterine pregnancies and three cervical pregnancies as CSP. In all of these cases, the fertilized eggs were implanted in the lower uterine segment or cervix, and the enlarged GS partially filled the scar diverticulum and was tightly attached to the cesarean scar. Additionally, color Doppler ultrasound showed a blood flow signal between the GS and the scar, while TVS was not able to determine the relationship between the GS and cesarean scar well, leading to misdiagnosis.
CEUS can dynamically display perfusion in the microcirculation in the uterus and lesion. An arteriovenous shunt is formed between the CSP villus and the myometrium and the contrast agent first reaches the implantation site of the embryo along with the blood flow. Therefore, the implantation site is the first area to be enhanced. 15 Such a feature of CEUS can precisely and directly show the implantation site of the fertilized egg. CEUS can also dynamically display the continuous perfusion process from the scar to the CSP lesion, and clearly displays blood flow around the GS by enhancement of micro-bubbles in blood flow. This can compensate for the limitations of color Doppler ultrasound on low-velocity blood flow and accurately shows the blood supply of CSP. Moreover, CEUS is a pure blood pool technique in which hematocele or effusion with no blood supply manifests as a non-enhanced region. For intrauterine pregnancy or cervical pregnancy in which a partial GS fills the cesarean scar diverticulum, CEUS can be used to observe the narrow band-like non-enhanced space between the GS and the scar. This is distinctly different from continuous perfusion of CSP. For a cesarean scar diverticulum, CEUS manifests as a triangle or round-like non-enhanced region, which is connected with the uterine cavity. Based on the above-mentioned enhancement features, CEUS allows for the differential diagnosis of CSP from lower intrauterine segment pregnancy, cervical pregnancy, and cesarean scar diverticulum. Therefore, CEUS can compensate for the disadvantages of TVS, improve the accurate diagnostic rate of CSP, and reduce the misdiagnosis rate. In this study, the accuracy rate of CEUS in diagnosing CSP was as high as 91.4%.
At present, there is no consensus on the optimal treatment of CSP. For CSP, pregnancy should be terminated as early as possible to select the most appropriate individualized treatment to avoid unnecessary excessive treatment.17,21 UAE effectively reduces the risks of intraoperative massive hemorrhage. UAE followed by dilation and curettage (D and C procedure) is recommended as the most beneficial treatment for lower complication rates, a shorter duration of hospitalization, and a more rapid resolution of CSP. Nonetheless, prior UAE is a significant risk factor for the placenta accreta spectrum in a subsequent pregnancy.22,23 Furthermore, UAE is an invasive treatment, which can induce post-embolization complications, including pain, fever, infection, and deep vein thrombosis. Additionally, because UAE temporarily blocks blood flow of the uterine artery, it may affect ovarian perfusion. This situation results in premature ovarian failure, a reduction in menstruation, and premature menopause, and affects the future fertility of patients.24,25
Consequently, UAE cannot be excessively carried out before uterine curettage for CSP, and the therapeutic scheme should be selected carefully. Assessment of myometrial thickness in a cesarean scar is important for selection of treatment. Ultrasound-guided suction curettage is a feasible method for a lower risk of endogenous CSP in patients with a myometrial thickness between the GS and the bladder of >3 mm. 11 Agten et al. 26 reported that all of their patients with a morbidly adherent placenta had a myometrial thickness of ≤2 mm in their first scan, and proposed that measuring myometrial thickness might be helpful for individualizing counseling of patients.
The decision for UAE may be affected by measurement of myometrial thickness in a cesarean scar, as well as the degree of hyperperfusion of a trophoblastic reaction, which may be the major benefit of CEUS versus TVS alone. CSP shares common pathological features with placental implantation, and there is abundant low-resistance blood flow in the myometrium invaded by villi. 27 The myometrium of cesarean scar is thin and weak, and the contraction ability of scar tissue is poor. Curettage used alone to terminate CSP may result in serious hemorrhage, even leading to hysterectomy. 28 Infiltration of villi into the myometrium could manifest as a continuous high enhancement region on CEUS, and is distinctly different from the normal uterine myometrium. In the current study, myometrial thickness as measured by CEUS appeared to be thinner than that measured by TVS. This finding indicates that the invasion range of CSP villi to the myometrium is actually greater than that displayed on TVS, and that CEUS is better able to judge the depth of villi invading than myometrium than TVS. We also found that the arrival time/time-to-peak intensity was faster, the peak intensity was higher, and the enhancement rate was greater in patients who received UAE treatment than in those who did not. Time-intensity curve parameters may serve as a semi-quantitative reference indicator of whether UAE should be selected.
Certain limitations should be noted in this study. This was a retrospective study, observations were made without prospective criteria for clinical management, and the sample size in this study was small. Therefore, the diagnostic criteria should be further investigated using a large sample size. Additionally, CEUS is not the major criterion for management of UAE. However, in our study, four patients who were diagnosed with CSP with a myometrial thickness of <3 mm by TVS were finally diagnosed as having a lower intrauterine pregnancy filling the cesarean scar diverticulum by CEUS. CEUS showed that the implantation position was not at the cesarean section site in these patients, and the perfusion features and time-intensity curve parameters of CEUS were not consistent with CSP. Clinicians sufficiently communicated with the patients, prepared for emergency intraoperative massive hemorrhage, and successfully carried out ultrasound-guided uterine curettage. Fortunately, intraoperative blood loss was <100 mL in all patients, and unnecessary UAE before uterine curettage was avoided. No abnormalities were observed in postoperative CEUS, and HCG levels progressively decreased to normal levels.
In conclusion, CEUS can dynamically display characteristics of the microcirculation in CSP. CEUS can clearly show the relationship between a cesarean scar and the GS, and enable judgement of the depth of villus infiltration more accurately compared with conventional TVS. CEUS can be used as a supplementary examination method of TVS, and CEUS can provide more precise diagnostic information for clinical selection of appropriate treatment and avoid excessive UAE.
