Abstract
The immune-related adverse events associated with immunotherapy may affect endocrine glands and other tissues. Two Chinese patients with malignancies were treated with programmed cell death-1 (PD-1) inhibitors (nivolumab and pembrolizumab) and followed up with biochemical tests over 1 year. After PD-1 treatment for 6 to 10 months, the patients developed symptoms of diabetes, ketoacidosis, and insulin secretion failure. Type 1 diabetes mellitus was confirmed by the characteristic fluctuation of blood glucose that was controlled with multiple daily insulin injections. Neither patient’s insulin depletion status was reversed in subsequent years. To decrease the life-threatening complications of diabetic hyperosmolar syndrome and ketoacidosis caused by type 1 diabetes mellitus, it is necessary to monitor the blood glucose and hemoglobin A1c levels. Islet β-cell autoantibodies and human leukocyte antigen genes can provide additional information in select cases.
Keywords
Introduction
Programmed cell death-1 (PD-1) inhibitors are a type of immune checkpoint inhibitor (ICI) that have served as effective anti-cancer drugs in recent years. They bind PD-1 receptors and block their interactions with programmed cell death ligand-1 (PD-L1). 1 Notably, PD-1 inhibitors are also associated with immune-related adverse events, which may influence endocrine glands and other tissues. Two monoclonal antibodies of PD-1 inhibitors, nivolumab and pembrolizumab, are widely used to treat malignancies. However, they cause irreversible impairment in multiple endocrine glands, especially islet β cells of the pancreas. We herein describe two Chinese patients who developed type 1 diabetes mellitus (T1DM) induced by PD-1 inhibitor therapy for malignancies.
Case report
Case 1
An 81-year-old woman was diagnosed with locally advanced nasopharyngeal carcinoma (T1N0M0) in 2017. She was treated with nivolumab at 2 mg/kg every 2 weeks. After six doses, a computed tomography scan showed no focal tumor. However, 6 months later (August 2018), she was admitted to our hospital with complaints of polyuria, polydipsia, weight loss, and vomiting. No evidence of pancreatitis was found by computed tomography examination and amylase and lipase measurement. Laboratory tests showed ketonuria, hyperglycemia (1124 mg/dL [62.45 mmol/L]), metabolic acidosis (pH, 7.23; bicarbonate ion, 14.5 mmol/L), a slightly high hemoglobin A1c level (8.8%), positive glutamic acid decarboxylase antibody (GADA), and absence of insulin-secreting capacity (C-peptide concentration of 0.02 ng/mL on fasting and 2 hours after a meal); additionally, whole-exome sequencing revealed the genotype HLA-DQA1/DQB1 (Table 1). These findings and the patient’s clinical course were compatible with the diagnostic criteria for fulminant T1DM. 2 She was prescribed multiple daily insulin injections. Weak insulin-secreting capacity or endogenous insulin secretion was found during subsequent years of clinical follow-up. Interestingly, GADA became negative after 3 years. The patient also developed autoimmune thyroiditis after developing T1DM; she exhibited subclinical hypothyroidism (elevated thyroid-stimulating hormone concentration with normal free triiodothyronine and free thyroxine concentrations) and positive thyroperoxidase and thyroglobulin antibodies (Table 1).
Laboratory data of Case 1.
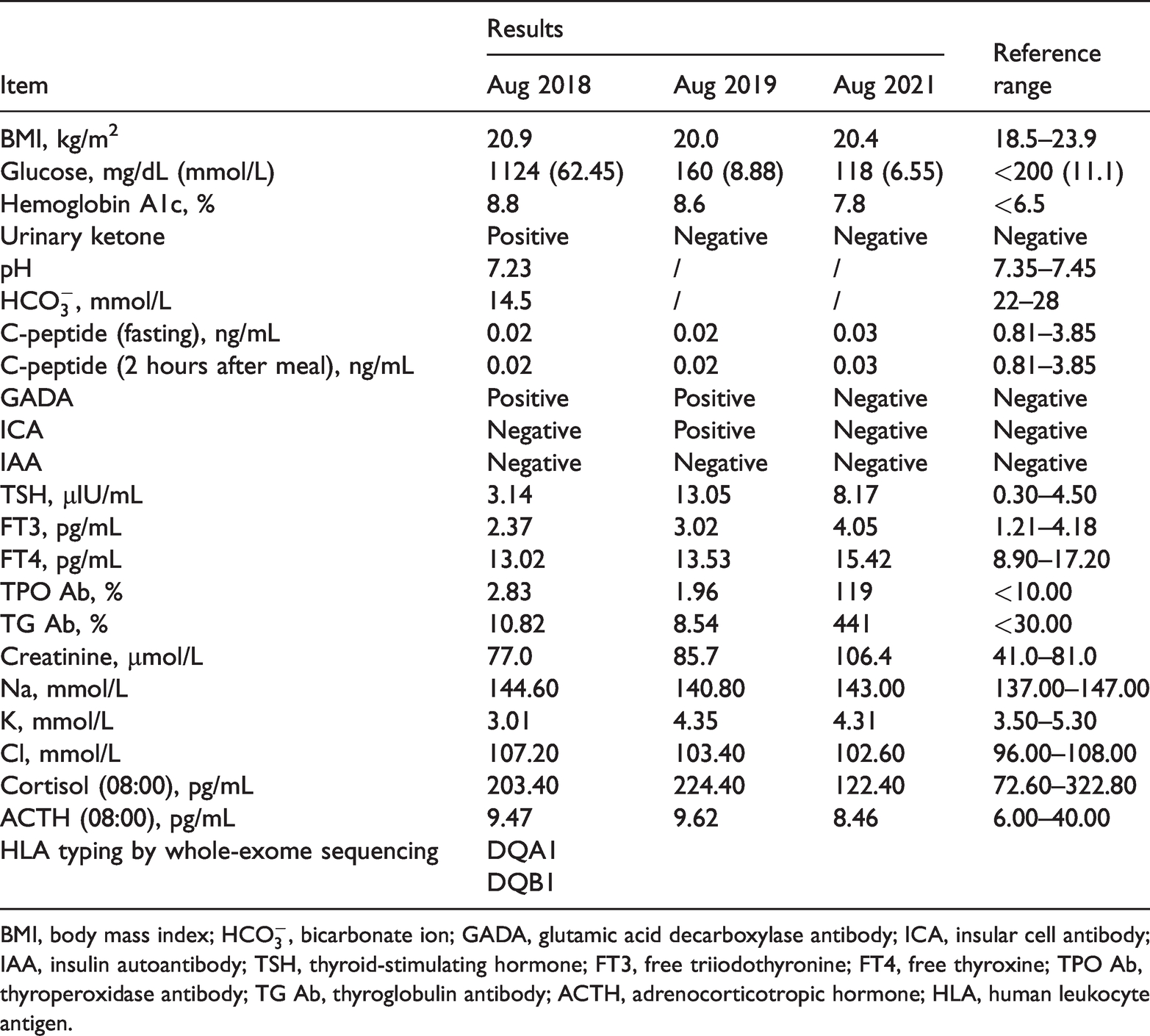
BMI, body mass index;
Case 2
A 76-year-old man with a 2-year history of type 2 diabetes mellitus was diagnosed with non-small cell lung cancer (T2aN1M0) in 2017. Because of his advanced age and asthenia, he was treated with 1.7 mg/kg of pembrolizumab every 17 days (instead of 200 mg every 3 weeks). After five doses, the tumor size was obviously reduced, but hypothyroidism appeared (low thyroxine and high thyroid-stimulating hormone concentrations). He was treated with L-thyroxine at 50 μg/day. After the 11th (final) dose in the 10th month of treatment, the patient was admitted to the emergency department with vomiting, diarrhea, weight loss, and lethargy but without polyuria, polydipsia or polyphagia. No evidence of pancreatitis was found by ultrasonography examination and amylase and lipase measurement. Laboratory tests suggested ketonuria, hyperglycemia (590.40 mg/dL [32.80 mmol/L]), negative insulin autoimmune antibodies, and some basal secreting capacity of β cells (fasting C-peptide concentration of 1.42 ng/mL), although the reserved capacity was poor (C-peptide concentration of 1.67 ng/mL at 2 hours after a meal). Whole-exome sequencing showed the genotype HLA-DQA1/DQB1 (Table 2). The patient’s glucose concentration was controlled by multiple daily insulin injections. In the 4th and 12th months of treatment, laboratory tests showed absence of C-peptide and insulin-secreting capacity (C-peptide, 0.01–0.04 ng/mL on fasting and 2 hours after a meal), low serum adrenocorticotropic hormone (ACTH) (3–5 pg/mL), low serum cortisol (49–70 pg/mL), and hyponatremia (126–132 mmol/L) (Table 2). Therefore, we corrected the diagnosis to ICI-induced brittle T1DM requiring multiple daily doses of insulin by the characteristic brittle glucose fluctuations. The patient developed evidence of hypothyroidism, decreased serum ACTH and cortisol concentrations, and intractable hyponatremia, indicating that the functions of the thyroid gland and pituitary gland were also impaired after treatment with pembrolizumab. The patient was treated with hydrocortisone; however, he died in December 2019.
Laboratory data of Case 2.

BMI, body mass index;
Written informed consent was obtained from the patients and their families for publication of these case reports. The study protocol was approved by the Ethics Committee of The First Affiliated Hospital of Kunming Medical University. The reporting of this study conforms to the CARE guidelines. 3
Discussion
Although neither patient described in this report had advanced diabetes, chronic diseases, or autoimmune diseases, they abruptly developed manifestations of T1DM after several months of treatment with PD-1 inhibitors. The cause of the diabetes was presumed to be an immune-related mechanism that was provoked by the immunotherapy. The first patient presented with fulminant T1DM, whereas the second patient presented with a clinical condition similar to latent autoimmune diabetes in adults.
The precise pathogenesis of T1DM induced by PD-1 inhibitors remains unclear. PD-1 is considered an immune checkpoint receptor that serves to restrict immune responses against self-tissues and tumor cells. 4 It mainly inhibits T-cell receptor–dependent T-cell activation to suppress T-cell proliferation, cytokine production, and cytotoxicity. 5 Moreover, research has shown that PD-1 can induce anergy and exhaustion of T cells and guide the differentiation of regulatory T cells.6,7 Therefore, PD-1 inhibitors are assumed to activate T cells to attack tumor cells but to also destroy self-tissues. One study showed that CD8+ T cells play a pathogenic role in the rapid onset of T1DM. About 30% of islet-infiltrating CD8+ T cells in pre-diabetic nonobese diabetic mice expressed PD-1, indicating that these PD-1+ CD8+ T cells were under the control of PD-1. After using PD-1/PD-L1 inhibitors, islet-infiltrating CD8+ T cells were rapidly activated to start proliferation. These CD8+ T cells were beyond the control of PD-1 and likely destroyed islet β cells. 5 Another study showed that pancreatic islets expressed PD-L1 at low levels in mice and humans, whereas PD-L1 was dramatically upregulated in inflamed islets. 8 Therefore, an inability to upregulate PD-1 and efficiently use the PD-1/PD-L1 pathway may be related to the development of T1DM induced by PD-1 inhibitors. 9 Affected patients usually follow a fulminant course and develop diabetic ketoacidosis. 10 Another possible mechanism of fulminant T1DM after using anti-PD-1 antibodies involves induction of PD-1 signal blocking, as proposed by Hosokawa et al. 11 The negative immune regulation is thought to consequently activate T cells to attack not only malignant cells but also islet β cells (Figure 1).

Possible mechanism underlying how PD-1 inhibitors lead to type 1 diabetes mellitus. PD-1 inhibitors target the PD-1 on the surface of T cells and block their interactions with PD-L1. The negative immune regulation of T cells is broken. Large numbers of proliferating T cells exert antitumor effects by attacking tumor cells. Similarly, in the presence of susceptibility factors such as human leukocyte antigen, islet β cells are attacked by T cells, and type 1 diabetes mellitus develops.
In clinical practice, we should be alert to clinical signs and symptoms of hyperglycemia. Specific screening strategies are needed to evaluate the risk of ICI-induced diabetes. We advocate for screening of the baseline blood glucose and hemoglobin A1c levels, and islet β-cell autoantibodies and human leukocyte antigen (HLA) genes may provide additional information in select cases. In patients with pre-existing type 2 diabetes, the worsening of glycemic control after immunotherapy indicates that pancreatic islets have been further destroyed by ICI treatment. In patients with normal baseline glycemia, ICI-induced diabetes should be diagnosed and treated if the glucose concentration increases. However, the screening of islet β-cell autoantibodies and HLA genes should be studied in more detail before definitive conclusions are drawn. Anti-GADA positivity before the beginning of immunotherapy may be a risk factor for diabetes. Conversely, in patients with anti-GADA negativity, the emergence of anti-GADA after immunotherapy may be the consequence of β-cell necrosis. Massive β-cell destruction could result in rapid release of intracellular GAD with the consequent development of anti-GADA. 12
In Case 1 of the present report, the patient was found to be negative for GADA after 3 years. We speculate that this may have been a result of the cessation of β-cell necrosis and stabilization of the T1DM. With respect to HLA genes, a recent literature review indicated that 14 of 21 patients with ICI-induced diabetes had high T1DM-risk HLA genotypes. 13 Another study showed that PD-1 inhibitors induced diabetes that was homozygous for the high-risk HLA-DR3/DQ2 (DRB*0301, DQA*0501, DQB*0201) haplotype. 14 Other researchers have found differences between populations and HLA genotypes: the DR3-DQ2 and DR4-DQ8 haplotypes are the main risk factors for T1DM, whereas the DR4-DQ4 and DR9-DQ9 haplotypes are associated with fulminant diabetes in Asians. 8 However, both of our patients had the haplotype HLA-DQA1/DQB1. Although such analyses could provide additional information for clinicians, they are not likely to be cost-effective given the relative rarity (1%–2% of PD-1/PD-L1 blockade-treated patients) and negativity of anti-GADA in about 50% of patients.8,15 Therefore, further research is essential to identify relevant risk factors in the development of PD-1 inhibitor-induced autoimmune diabetes.
In the present study, we also observed that the first patient developed autoimmune thyroiditis and subclinical hypothyroidism after developing T1DM. This may have been a type of destructive thyroiditis with release of thyroid antigen and consequent secondary antibody production and hypothyroidism. 16 We suspect that the second patient had pituitary dysfunction because of the low ACTH value. A hypoglycemia test was not performed because of this patient’s asthenia. Research has shown that insulitis, thyroid disorders, hypophysitis, and adrenalitis may occur after treatment with high doses of ICIs, eventually resulting in T1DM, hyperthyroidism or hypothyroidism, hypopituitarism, and adrenocortical hypofunction. 17 We advise physicians to pay more attention to this situation.
In conclusion, it is important for clinicians to be aware of the endocrine-related adverse events of ICIs and screen the functions of related endocrine glands in patients who are using ICIs. This will help to identify hypoendocrinism early, allowing the patient to be supplemented with necessary hormones in a timely manner and avoiding some life-threatening complications. At the same time, strengthening cooperation between endocrinologists and oncologists can bring benefits to patients. However, it is important to appraise the advantages and disadvantages of ICIs before initiating therapy with them.
Note: The patient in Case 1 said that although nivolumab cured her nasopharyngeal carcinoma, it caused her to develop brittle T1DM and required multiple daily insulin injections. However, her daily life was not affected. She thanked nivolumab for saving her life. She hoped that clinicians will pay more attention to the endocrine side effects of PD-1 inhibitors.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221121940 - Supplemental material for Type 1 diabetes mellitus induced by PD-1 inhibitors in China: a report of two cases
Supplemental material, sj-pdf-1-imr-10.1177_03000605221121940 for Type 1 diabetes mellitus induced by PD-1 inhibitors in China: a report of two cases by Jingmei Luo, Jiagang Feng, Chunyan Liu, Zhongce Yang, Dong Zhan, Yanan Wu, Li Pan and Lihua Zhang in Journal of International Medical Research
Footnotes
Acknowledgements
The authors appreciate all the participants for their continuous participation in and contributions to this research project.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Data assessment method
The data related to this manuscript can be accessed through the medical record management system of the First Affiliated Hospital of Kunming Medical University, Kunming, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81960156), High Level Health Technology Personnel Training Project of Yunnan Province (D-2018044), Health Science and Technology Program of Yunnan Province [2019FE001(-040)], and the Joint Project of Kunming Medical University and Science and Technology Agency (202101AY070001-049). The funders played no role in the study design, data analysis, or interpretation or writing of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
