Abstract
Objective
This study was performed to evaluate the correlation between parameters measured by bedside ultrasonography and detection of intracranial organic lesions in patients with impaired consciousness in an intensive care unit (ICU) setting.
Methods
We retrospectively reviewed the medical records of patients who were admitted to our ICU from April 2017 to July 2019. Patients who underwent computed tomography or magnetic resonance imaging examination and measurement of the flow velocity of the carotid and intracranial arteries and the optic nerve sheath diameter by ultrasonography were selected for analysis.
Results
In total, 64 patients were analyzed in this study. Of these, intracranial lesions were detected by computed tomography or magnetic resonance imaging in 17 (27%) patients. The left:right ratio of the end-diastolic velocity of the bilateral common carotid artery (CCA-ED ratio) and the pulsatility index of the middle cerebral artery (MCA-PI) were significantly higher in patients with than in those without intracranial lesions. The cut-off value of the CCA-ED ratio was 1.55 (sensitivity, 66.7%; specificity, 81.6%), and that of the MCA-PI was 1.21 (sensitivity, 57.1%; specificity, 76.7%).
Conclusion
Bedside ultrasonography is useful for predicting intracranial lesions requiring therapeutic intervention in ICU patients with impaired consciousness.
Keywords
Introduction
Disturbance of consciousness is not rare in critically ill patients, and approximately 12% of intensive care unit (ICU) patients reportedly present with neurological complications. In addition, several studies have demonstrated that impaired consciousness and neurological complications are associated with morbidity and mortality in the ICU setting.1–3 Neurological examination is often difficult to perform in critically ill patients admitted to the ICU because of the effects of sedatives. Brain computed tomography (CT) and magnetic resonance imaging (MRI) are widely used for detailed examination of intracranial diseases, but the need to transfer the patient and the long examination time increase the risk in critically ill patients with unstable respiratory and circulatory states.
Measurement of carotid and intracranial blood flow by carotid and transcranial Doppler ultrasonography has been used to evaluate stenosis of major arteries and occlusive lesions in patients with ischemic stroke.4–6 More recently, the combination of carotid duplex ultrasonography and the head-up test were reported to detect stenosis of the intracranial vertebral basilar arteries. 7 In the field of neurosurgery and neurocritical care, transcranial Doppler ultrasonography has been used to monitor vasospasm after subarachnoid hemorrhage, patency after cerebrovascular bypass, and microemboli during endovascular treatment. 8 In recent years, measurement of the pulsatility index (PI) of intracranial arteries and the optic nerve sheath diameter (ONSD) by ultrasound has been reported to be useful for noninvasive identification of elevated intracranial pressure (ICP).9–11 In clinical practice, it is more important to determine whether intracranial lesions that require therapeutic intervention are present than to identify ICP elevation or vascular lesions. If simple bedside examinations can predict the presence of intracranial lesions, it will be possible to avoid unnecessary transfer of unstable critically ill patients.
Therefore, the present study was performed to investigate whether ultrasonography is useful for determining the presence of intracranial organic disease in critically ill patients with impaired consciousness in the ICU setting.
Methods
Ethics
This study was initiated after obtaining approval from the Kyushu University Hospital Institutional Review Board (#21010–00). All procedures for the acquisition of clinical data, analyses, and preparation of this manuscript were conducted in strict compliance with the institutional guideline. Informed consent was waived because of the retrospective nature of this study. The opt-out method on our official website was used, which was approved by the Kyushu University Hospital Institutional Review Board.
Patient selection
Among the patients who were admitted to our ICU from April 2017 to July 2019, we selected patients who met the following three criteria: (1) presentation with disturbance of consciousness under interruption of sedation, (2) examination by brain CT or MRI, and (3) examination by carotid ultrasonography, examination by transcranial color-coded duplex sonography (TCCS), and measurement of the ONSD.
Ultrasonography protocol
Carotid ultrasonography, TCCS, and measurement of the ONSD were performed using a Philips CX50 ultrasound system (Philips Healthcare, Tokyo, Japan) while examiners were blinded to the CT and MRI findings. Carotid ultrasonography was performed with a 7.5-MHz linear transducer. The patients laid in the supine position with their heads turned away from the side being examined. TCCS was performed with a 2-MHz transducer via the patient’s temporal bone window. We measured the peak flow velocity and end-diastolic velocity of the bilateral common carotid artery (CCA), internal carotid artery (ICA), vertebral artery, M1 segment of the middle cerebral artery (MCA), and P1 segment of the posterior cerebral artery. The PI was automatically calculated using the following formula: PI = [(systolic velocity−diastolic velocity)/mean velocity]. In addition, the left:right ratio of the end-diastolic velocity (ED ratio) was calculated by dividing the velocity on the larger side by that on the smaller side. The ONSD was measured using a 7.5-MHz linear probe at 3 mm behind each eye globe.
Statistical analysis
All data are expressed as the mean ± standard deviation. The patients were dichotomized according to the presence of intracranial lesions detected by CT or MRI, and their clinical characteristics and measurement values determined by ultrasound examination were compared using an unpaired t-test, the Mann–Whitney U test, or the χ2 test as appropriate. The PI was compared on either the left or right side, whichever was the greater side. The larger ONSD was used for analysis. For the measured values with significant differences between the two groups, receiver operating characteristic (ROC) curve analysis and the estimated area under the ROC curve were used to quantify how accurately the measured values could predict intracranial lesions. ROC analysis was also used to visually assess the optimal cutoff. Furthermore, the sensitivity and specificity were evaluated.
All statistical analyses were performed using JMP® Software version 15.0.0 for Macintosh (SAS Institute Inc., Cary, NC, USA) and EZR version 1.36 (Saitama Medical Center, Jichi Medical Univ., Saitama, Japan, The R Foundation, Vienna, Austria). 12 A P-value of <0.05 was considered statistically significant.
Results
In total, 64 patients (mean age 59.6 ± 16.8 years; females, n = 25) met the inclusion criteria and were investigated in this study. The median Glasgow coma scale score was 6 (interquartile range, 3–8). The main reasons for ICU admission were trauma (n = 14), sepsis (n = 12), consciousness disturbance (n = 11), postoperative management (n = 9; cardiovascular, n = 4; brain, n = 3; gastrointestinal, n = 1; peripheral vascular, n = 1), management of heart disease (n = 5; acute coronary syndrome, n = 3; idiopathic ventricular fibrillation, n = 1; dilated cardiomyopathy, n = 1), acute respiratory failure (n = 4), acute liver failure (n = 3), acute kidney injury (n = 3), thyroid storm (n = 2), and multiorgan failure because of hematologic malignancy (n = 1). Among the 64 patients, an intracranial lesion was detected by CT or MRI in 17 (27%). Cerebral hemorrhage was the most frequent finding (n = 8). Cerebral infarction was the second most frequent finding (n = 5), followed by posterior reversible encephalopathy syndrome (n = 2), diffuse cerebral edema (n = 1), and subarachnoid hemorrhage (n = 1).
There were no significant differences in age, sex, Glasgow coma scale score, or laboratory data between the patients with and without an intracranial lesion (Table 1). Among the parameters measured by ultrasonography, the ED-ratio of the CCA (CCA-ED ratio) and the PI of the MCA (MCA-PI) were significantly higher in patients with than in those without intracranial lesions (P = 0.007 and P = 0.017, respectively, Table 2). The ROC curves of the CCA-ED ratio and MCA-PI for predicting intracranial lesions are shown in Figure 1. The areas under the curves were 0.69 (0.44–0.94) and 0.66 (0.48–0.85), respectively. The optimal threshold of the CCA-ED ratio for predicting intracranial lesions was 1.55 (sensitivity, 66.7%; specificity 81.6%), and that of the MCA-PI was 1.21 (sensitivity, 57.1%; specificity 76.7%).
General characteristics and laboratory data of patients with and without intracranial lesions.
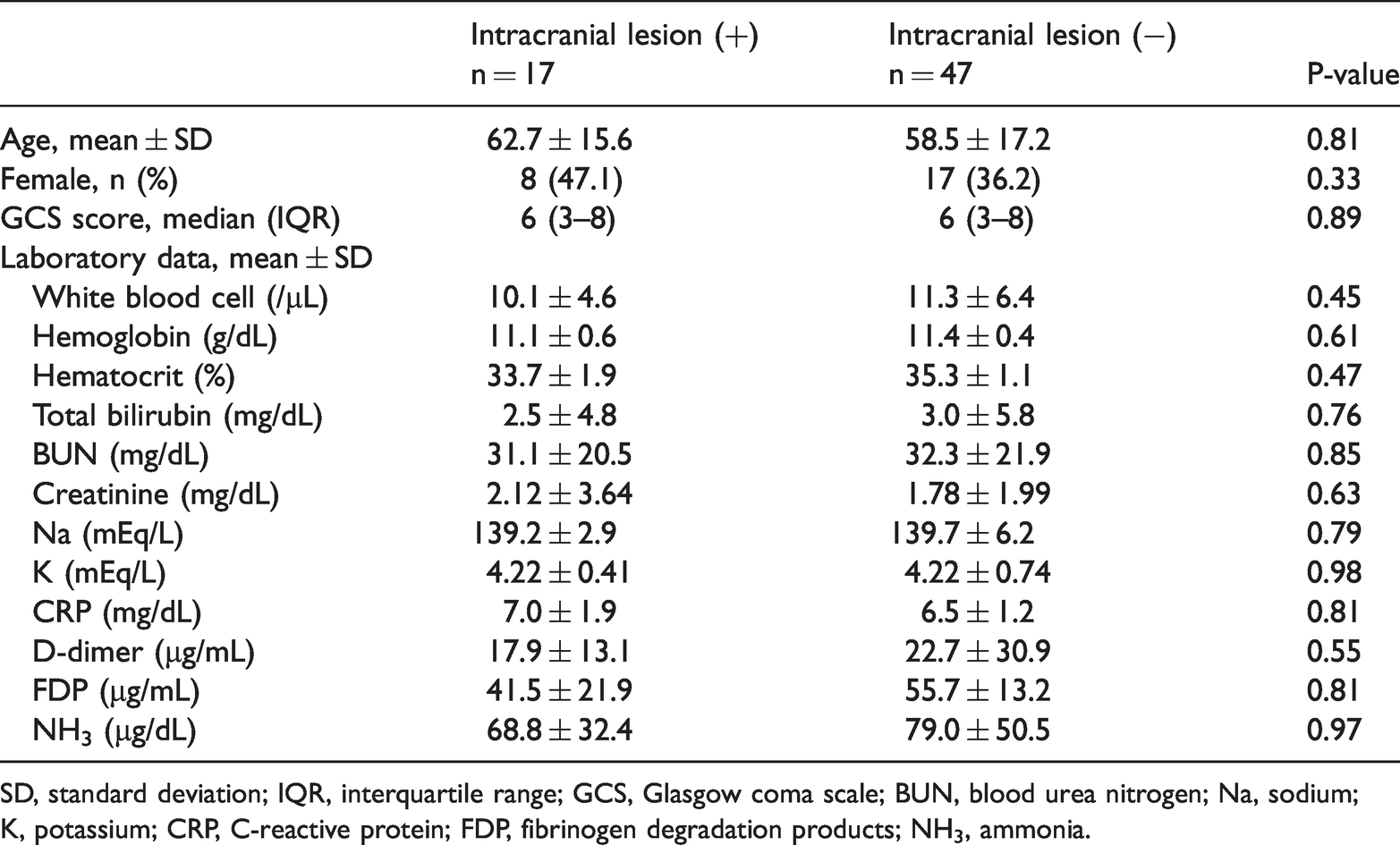
SD, standard deviation; IQR, interquartile range; GCS, Glasgow coma scale; BUN, blood urea nitrogen; Na, sodium; K, potassium; CRP, C-reactive protein; FDP, fibrinogen degradation products; NH3, ammonia.
Comparison of ultrasonography parameters between patients with and without intracranial lesions.

Data are presented as the mean ± standard deviation. CCA, common carotid artery; ED ratio, left:right ratio of the end-diastolic velocity; PI, pulsatility index; ICA, internal carotid artery; VA, vertebral artery; MCA, middle cerebral artery; PCA, posterior cerebral artery; ONSD, optic nerve sheath diameter.
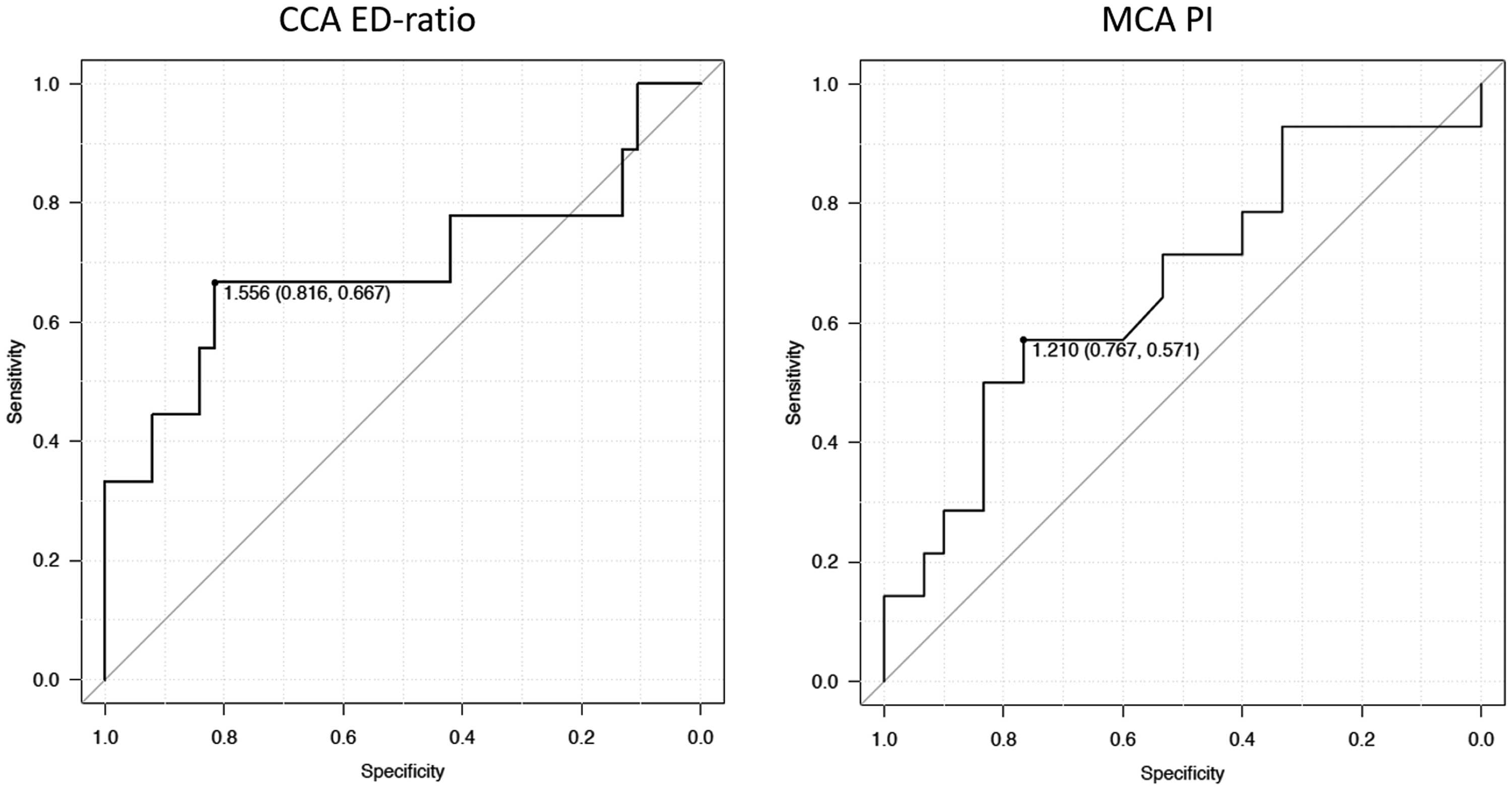
Receiver operating characteristic curves of the left:right ratio of the end-diastolic velocity of the bilateral common carotid artery (CCA-ED ratio) and pulsatility index of the middle cerebral artery (MCA-PI). The area under the curve of the CCA-ED ratio was 0.69 (0.44–0.94) and that of the MCA-PI was 0.66 (0.48–0.85).
Discussion
In the present study, the CCA-ED ratio and MCA-PI were associated with the presence of intracranial lesions in critically ill patients with impaired consciousness. The correlation was moderate to weak; however, to the best of our knowledge, this is the first study to assess the utility of neuro-sonographic techniques for diagnosis of intracranial lesions in the ICU setting.
Past studies have examined the association between noninvasive ultrasound methods and increased ICP but not the association with intracranial lesions by imaging.9–11,13,14 Many pathological conditions present with disturbance of consciousness, and the treatment intervention differs for each condition. Therefore, in this study, we examined whether noninvasive ultrasound examination could predict the presence of intracranial lesions requiring therapeutic intervention identified by CT and MRI.
Past studies have shown that the CCA-ED ratio indicates major arterial occlusion of the distal portion of the ICA or MCA in patients with ischemic stroke.4,6 According to those studies, a CCA-ED ratio of >1.3 distinguishes patients with occlusion of the ICA or proximal MCA. In the present study, the cut-off value of the CCA-ED ratio was 1.55. However, half of the patients with intracranial lesions in our study had intracranial hemorrhage, and only two patients had major arterial occlusion. A unilateral increase in the ICP by a hematoma might lead to a decrease in the end-diastolic flow velocity of the ipsilateral CCA.
The PI is considered to be useful for evaluating peripheral vascular resistance. The MCA-PI derived by transcranial Doppler ultrasonography is reportedly correlated with the ICP and cerebral perfusion pressure in various intracranial disorders, such as traumatic brain injury, intracranial hemorrhage, subarachnoid hemorrhage, and hypoxic brain injury after cardiac arrest.9,10,14–17 In the present study, the increased MCA-PI in patients with intracranial lesions might reflect an increased ICP secondary to a hematoma or cerebral edema. In addition, increased peripheral vascular resistance because of ischemic stroke might have also contributed.
Several studies have examined the association between the ONSD and increased ICP in patients with traumatic and non-traumatic causes of high ICP. In these studies, the ONSD demonstrated better sensitivity and specificity than other noninvasive techniques. 10 A recent meta-analysis of 18 prospective studies showed that the ONSD had a pooled sensitivity of 0.90, specificity of 0.85, and summarized ROC of 0.93 for ICP elevation. 18 In the present study, the ONSD was not significantly correlated with the presence of intracranial lesions. The reason for the different results between the present study and preceding studies is presumed to be that most of the patients with intracranial lesions in our study had focal lesions without remarkable ICP elevation. The cut-off values of the ONSD for ICP elevation in past studies ranged from 4.8 to 6.4 mm. 19 The ONSD in the patient with diffuse cerebral edema in this study was >6 mm.
Noninvasive methods of ICP measurement are based on different pathophysiological mechanisms underlying ICP elevation. For example, the ONSD reflects the cerebrospinal fluid pressure of the intra-orbital subarachnoid space, and TCCS reveals alterations of cerebral blood flow. Several recent studies have demonstrated that the multimodal combination of noninvasive methods can increase the accuracy of intracranial hypertension detection.10,11 Similarly, in our study, the combination of the CCA-ED ratio with the MCA-PI resulted in a greater area under the ROC curve (0.781).
The findings from this study have several implications for future clinical practice. Our method could be useful in other situations in which brain imaging is unavailable or cannot be performed immediately. For example, it can be applied as a screening tool in the emergency department or pre-hospital setting. Although the results of only a single examination were analyzed in this study, repeated examination or continuous monitoring could yield a higher prediction rate for intracranial lesions.
Limitations
This study has several limitations. First, because this study was retrospective and involved a single center, the number of patients was relatively small. Institutional bias may have also been present, and not all of the results of this study can be generalized. Second, parameters that affect cerebral blood flow and ICP, such as the arterial blood pressure and partial pressure of CO2, were not considered. In some preceding studies investigating noninvasive measurement of ICP, patients with traumatic brain injury or stroke were managed in a state of normocapnia (partial pressure of CO2 >35 mmHg) and adequate cerebral perfusion pressure (>60 mmHg).10,20 The backgrounds of the patients in our study were diverse, and it was difficult to carry out unified management. Third, the MCA-PI is increased by several conditions, such as age, hypertension, and cerebral white matter hyperintensities. 21 In addition, the MCA-PI is decreased in the presence of severe stenosis or occlusion of the ICA. It should be taken into account when interpreting the results of this study that these factors were not adjusted. Finally, ultrasonography was performed by only one investigator, which limits the generalizability of the results.
Conclusion
In this study, the CCA-ED ratio and MCA-PI based on bedside sonographic examination showed a significant correlation with the presence of intracranial lesions in critically ill patients presenting to the ICU with consciousness disturbance. However, the correlation was moderate to weak. Further studies are needed to confirm and validate our findings.
Footnotes
Author contributions
Conceptualization: YS, MS, TA, MN, KN, JM, KT.
Data curation: YS, SM.
Formal analysis: YS, SM.
Writing original draft: YS.
Writing–review & editing: YS.
Supervision: AT, MN, NK, JM, KT, TK.
Declaration of conflicting interest
The authors have no conflicts of interest to disclose.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
