Abstract
Multifocal motor neuropathy (MMN) is an immune-mediated and acquired demyelinating motor polyneuropathy. Several cases of polyneuropathy associated with severe acquired respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccination have been reported. However, MMN has not been reported as a complication of SARS-CoV-2 vaccination. In this study, we report a case of MMN with progressive muscle weakness following the second dose of the Pfizer-BioNTech mRNA vaccine. It was diagnosed by clinical evaluation and electroneuromyography. SARS-CoV-2 vaccination is increasing rapidly all over the world. Some cases of polyneuropathy, especially Guillain–Barré syndrome, have been reported after vaccination. This is the first case report of MMN after SARS-CoV-2 vaccination.
Introduction
Multifocal motor neuropathy (MMN) is an immune-mediated and acquired demyelinating motor polyneuropathy. The main clinical features of the disease are asymmetric, progressive weakness in two or more peripheral nerves without sensory symptoms. Weakness is more prominent in the distal extremities. Fasciculation and cramps are symptoms in muscles with weakness. MMN is a rare disease with a frequency of 1–2/100,000. The disease has an autoimmune etiology, but specific autoantibodies are not often detected in patients. 1 Therefore, the disease is diagnosed according to clinical and electrophysiological evaluation. The presence of multifocal motor conduction blocks with electroneuromyography is a major neurophysiological feature. 1, 2
The global pandemic caused by severe acquired respiratory syndrome coronavirus 2 (SARS-CoV-2) infection includes many obligations such as vaccine production, vaccination itself, and management of this process. Several vaccines have been approved by the US Food and Drug Administration (FDA) for emergency vaccination. Potential side effects associated with vaccination include pain, fatigue, fever, and allergic reactions. 3 A few cases of Guillain–Barré syndrome (GBS) have been reported after SARS-CoV-2 vaccination. 4 However, MMN associated with SARS-CoV-2 vaccination has not been reported. In this study, we report a case of MMN as a potentially complication after the second dose of the SARS-CoV-2 vaccine.
Case report
The reporting of this case report conforms to the Equator network guideline (The CARE Guidelines: Consensus-based Clinical Case Reporting Guideline Development: https://www.equator-network.org/reporting-guidelines/care/). 5 Informed consent form was obtained from patient for publication of this case report. The characteristics of the case were reported to the Scientific Research Evaluation Commission of the Ministry of Health, Turkey. The case report was approved by the commission. Meanwhile, ethics committee approval is not required for case reports.
The patient was a 52-year-old man referred to the neurology clinic with weakness, especially in the lower extremities, and gait disturbance for 15 days. There was no disease or functional disability in his previous medical history. There was no history of trauma, fever, night sweats, bowel and bladder dysfunction, or infection. The second dose of SARS-CoV-2 vaccine (Pfizer-BioNTech) was received 25 days before presentation.
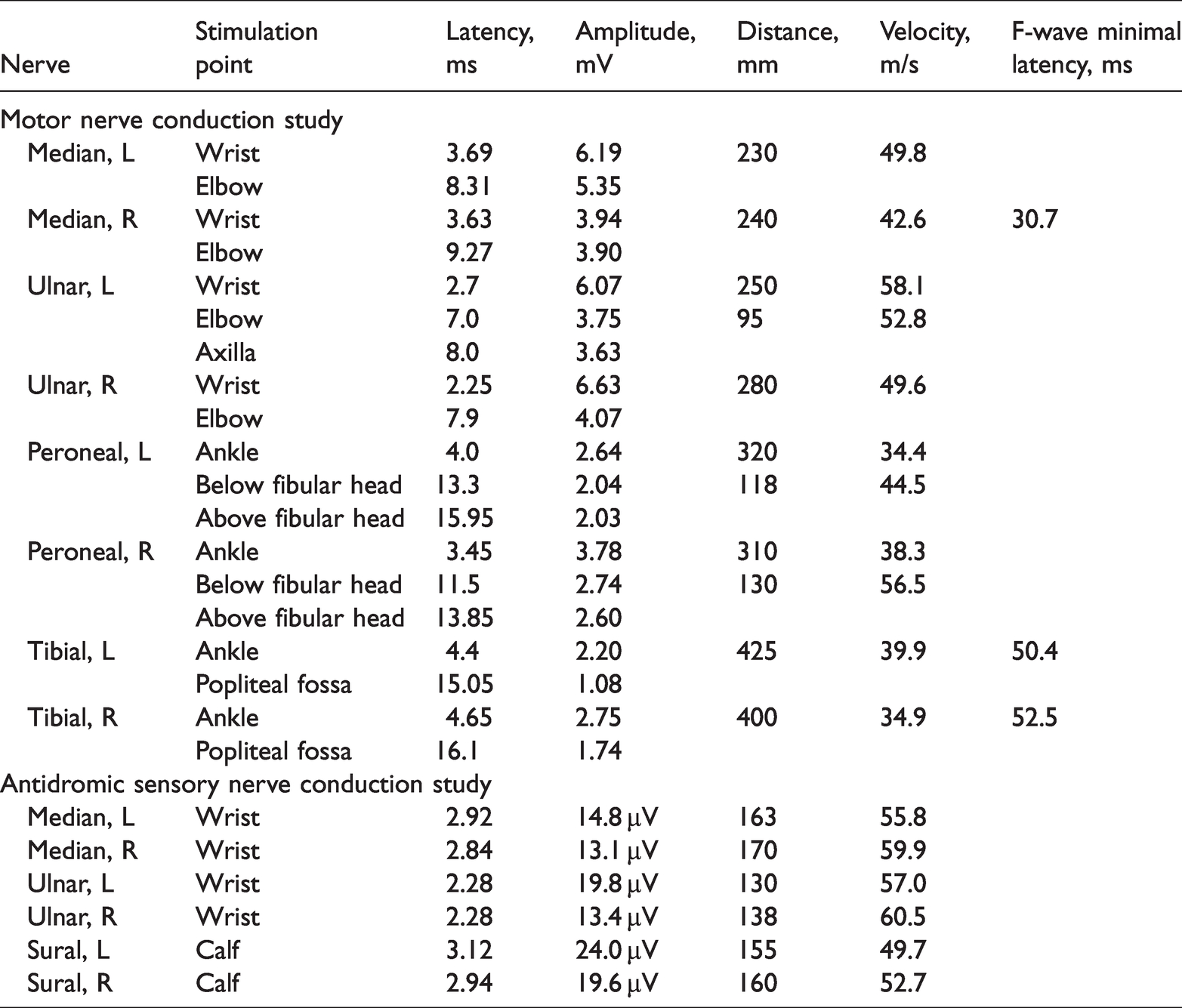
Pupillary reflexes and eye movements were normal in the neurologic examination. Facial asymmetry was not observed. The uvula was in the midline position, and the gag reflex was normal bilaterally. There was no atrophy of the tongue, and the tongue movements were normal. Mild muscle weakness was detected in both the upper and lower limbs in the neurological examination. We performed manual muscle strength tests. The upper extremity muscle strength on the right was 4–5/5 at C5–C6, 4/5 at C7, and 3/5 at C8–T1. On the left, the muscle strength from C5 to C7 was 4/5, and that at C8–T1 was 3–4/5. The lower extremity muscle strength from L2 to S1 on the right was 4/5. On the left, the muscle strength was 3/5 at L2–L3 and 3–4/5 at L4–S1. The patient was able to walk, albeit with difficulty. The strength of specific muscles was evaluated. Strength deficits were detected in the triceps brachii, abductor pollicis longus, and interosseous muscles in the right upper extremity and the biceps brachii, abductor pollicis, and interosseous muscles in the left upper extremity. In addition, strength deficits were detected in the bilateral tibialis anterior, vastus medialis, biceps femoris, and right gastrocnemius in the lower extremity. Deep tendon reflexes, flexor plantar responses, and sensory data were normal. Muscle fasciculations were noted, especially in the tibialis anterior and vastus medialis. Pathologic muscle atrophy was not observed. In the nerve conduction study, conduction blocks were detected in the left tibial, right tibial, left ulnar, and right peroneal motor nerves (Figure 1 and Table 1). The conduction velocities, amplitudes, and latencies of the sensory nerves were normal (Table 1). In needle electromyography, denervation potentials and polyphasia-extended motor unit potentials (MUPs) were observed in L5 innervated muscles. MUPs were normal in other muscles, but fasciculation potentials were observed (Table 2). The diagnosis of MMN was confirmed on the basis of clinical and electrophysiological tests. 6 – 8
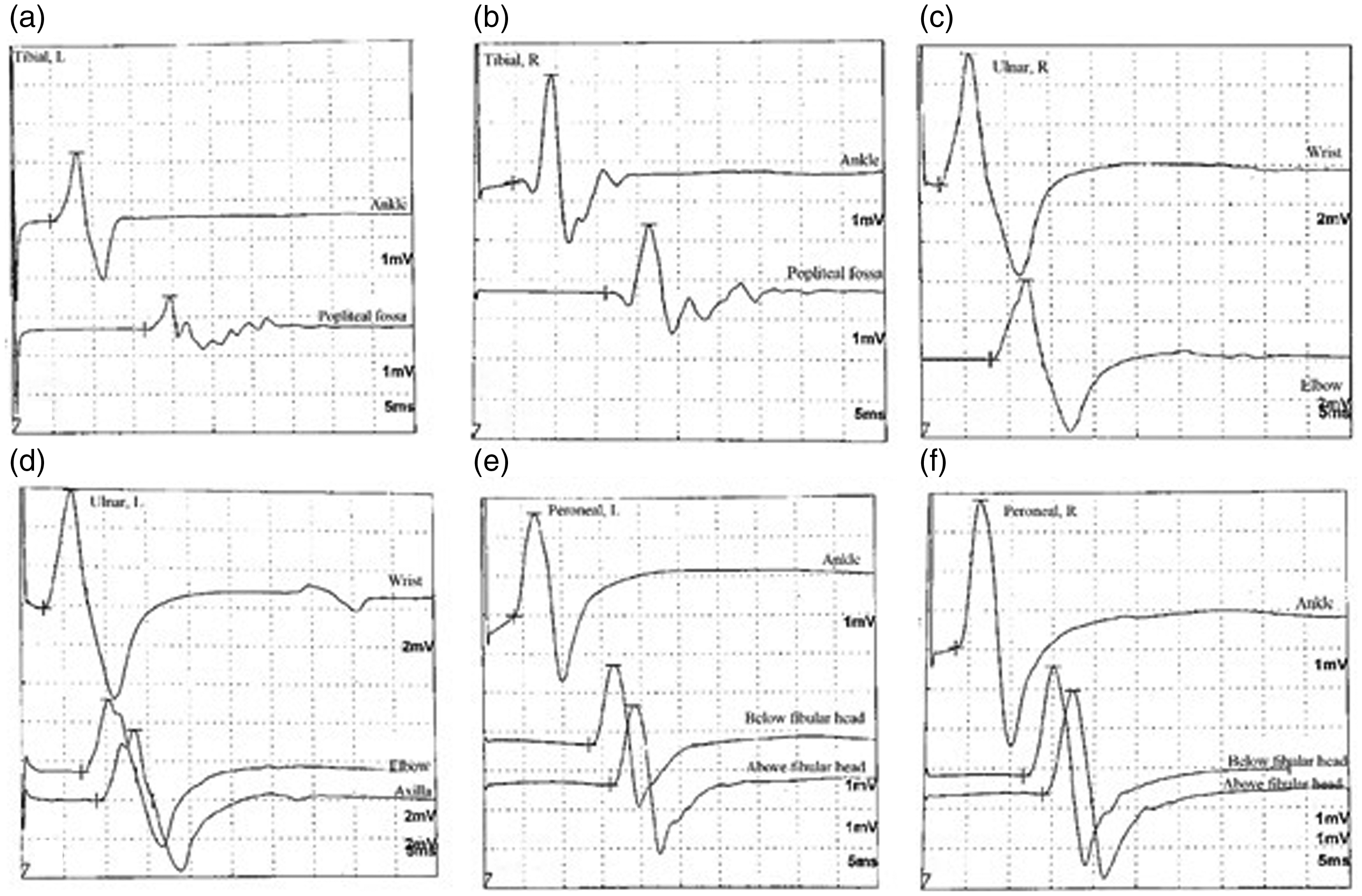
Motor nerve conduction study. (a) Left tibial, (b) right tibial, (c) right ulnar, (d) left ulnar, (e) left peroneal and (f) right peroneal.
Nerve conduction study.
Needle electromyography parameters in the patient.

Contrast-enhanced brain and thoracic magnetic resonance imaging (MRI) data were normal. Cervical MRI revealed diffuse annular bulging. It was chronic and incidental images (Figure 2). Chest X-ray was normal, and the nasopharyngeal SARS-CoV-2 swab test was negative. Standard laboratory tests and special blood tests (HbA1c, evaluation for vasculitis [ANA, ENA, anti-DNA, c-ANCA, p-ANCA], HIV, HBV, HCV, serum vitamin B12 and folate levels, serum protein electrophoresis, and paraneoplastic evaluation) were normal. Anti-ganglioside antibodies were negative. The cerebrospinal fluid (CSF) opening pressure was 180 cm H2O. Glucose, microprotein, and electrolyte levels were normal in CSF biochemical analysis. Real-time PCR for herpes simplex virus, Epstein–Barr virus, enterovirus, varicella zoster, cytomegalovirus, and adenovirus and CSF microscopy and culture were negative. Intravenous immunoglobulin (0.4 g/kg/day for 5 days) was promptly started according to the European Federation of Neurological Societies/Peripheral Nerve Society guideline for the management of MMN. 7 No treatment-related complications were observed. Gait impairment was reduced, but muscle strength was the same as that on admission. At 2 weeks after admission, the patient was treated in the neurology clinic and discharged for rehabilitation. His neuropathic symptoms had regressed in the follow-up visit (in the second month) after rehabilitation. Manual muscle strength tests were performed. The upper extremity muscle strength from C5 to C7 was 5/5 and that at C8–T1 was 4–5/5 on the right and left. The lower extremity muscle strength from L2 to S1 was 5/5 on the right. On the left, the muscle strength was 4–5/5 at L2–L3 and 5/5 at L4–S1. Because the muscle symptoms had improved, radiological or electrophysiological tests were not repeated, and immunosuppressive treatment was not started. It was planned to continue the neuro-rehabilitation program.

Magnetic resonance imaging. (a) T2/axial, (b) T2-FLAIR/coronal, (c) T1/axial-contrast enhanced, (d) T1/coronal-contrast enhanced, (e, g) T2/sagittal and (f, h) T1/sagittal-contrast enhanced.
Discussion
Global vaccination in response to the SARS-CoV-2 pandemic has rapidly proceeded. An international effort has been made to shorten the duration of vaccine development to protect the community. Considering the high efficacy, it was not possible to compare the long-term efficacy and safety of the vaccines with a control group. 3
The potential association between vaccines and autoimmune diseases has been a matter of concern. However, reports of vaccine-associated inflammatory neuropathies have raised some concerns. The possible relationship between vaccination and inflammatory neuropathies has been a concern since the swine influenza vaccination program in 1976. At that time, the vaccination process was interrupted by increased cases of GBS. 9 Subsequently, studies have been conducted to evaluate the relationship between vaccination and GBS. However, the relationship between GBS and vaccination has not been conclusively demonstrated. 10 Some studies reported a small but statistically significant increased risk of GBS after influenza vaccination. 11 Only a few studies have evaluated the relationship between vaccines and chronic inflammatory neuropathies. Eleven percent of patients with chronic inflammatory demyelinating polyneuropathy have a history of vaccination within 8 weeks from the beginning of neuropathy symptoms. 12 However, no studies detected a causal association between vaccination and MMN.
Several types of vaccines against SARS-CoV-2 infection have been developed, including mRNA vaccines developed by Pfizer-BioNTech, Moderna, and CureVac. These vaccines introduce mRNA into cells, usually via a lipid nanoparticle. Intracellular mRNA mediates recognition of spike proteins on the surface of SARS-CoV-2. Antibodies against the spike protein are produced. If the antibody encounters the actual virus, it mediates the recognition and destruction of the virus without causing disease. However, in some people, the production of antibodies against myelin can trigger autoimmune processes. Inflammatory polyneuropathies may arise through this mechanism. 3,13 A case of GBS associated with these mechanisms was reported after second dose of the Pfizer-BioNTech vaccine. 14 Considering the growing number of individuals vaccinated for coronavirus disease 2019 (COVID-19), the incidence of GBS will increase in the near future. The causal relationship between COVID-19 vaccines and GBS development should be considered with caution. 15 However, in the literature, MMN has not reported during the development of COVID-19 or the vaccination process. The current case involved the development of MMN after SARS-CoV-2 vaccination. No factors other than vaccination were detected in the etiology of MMN. There was a probable causal relationship between vaccination and the onset of symptoms. Therefore, it was considered that the disease might be associated with vaccination.
In conclusion, the current case report is the first evidence of a probable causal association between SARS-CoV-2 vaccination and MMN. Physicians should report potentially complications of the SARS-CoV-2 vaccine without forgetting the benefits.
Footnotes
Authors’ contributions
FE, ST and RA conceptualized the study, had full access to all data and take responsibility for the integrity of the data. FE, ST, RA and SO contributed to the writing of the manuscript. All the authors contributed to the data interpretation, reviewed, and approved the final version.
Declaration of competing interest
The authors declares that there is no conflict of interest.
Ethics statement
Informed consent was obtained from the patient.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
