Abstract
Although high-grade serous cancer (HGSC) accounts for >70% of ovarian epithelial cancers, it is rarely associated with endometriosis. No previous study has reported an association between the malignant transformation of uterine ligament endometriosis and HGSC. Here, we reported two cases of Chinese female patients with HGSC arising from endometriosis in the uterosacral ligament. They had a long-term history of endometriosis and dysmenorrhea. Both were diagnosed with HGSC at stage IIB. They underwent operations and six cycles of chemotherapy with paclitaxel and carboplatin and have remained disease-free to date. Genomic analysis showed no known/suspected pathogenic variations or somatic homologous recombination deficiency in the two cases. In conclusion, these rare cases of HGSC from endometriosis might indicate a new origin of ovarian type II carcinoma. Patients with a long-term history of endometriosis and sudden aggravation of dysmenorrhea or vaginal bleeding should be aware of the possibility of endometriotic malignant transformation.
Keywords
Introduction
Endometriosis, defined as the presence and growth of endometrial-like glandular epithelium and stroma outside the uterine cavity, is a chronic, estrogen-dependent, benign gynecologic disease. 1 The incidence of malignant transformation ranges between 0.7% to 2.5%. 2 Eighty percent of endometriotic malignant transformation occurs in the ovary, with the remaining 20% occurring in extraovarian sites. 3 Women diagnosed with malignant extraovarian endometriosis are 10 to 20 years younger than those diagnosed with ovarian or endometrial cancer. 4 The histopathology of malignant extraovarian endometriosis is primarily represented by endometrioid carcinoma (69.1%), sarcoma (25%) and clear cell carcinoma (4.5%). 5 Endometrioid and clear cell carcinoma are considered endometriosis-associated ovarian cancers.
High-grade serous cancer (HGSC) is the most aggressive and common ovarian carcinoma and is believed to be closely associated with a TP53 mutation (type II carcinoma). Although HGSC accounts for >70% of ovarian epithelial cancers, it is rarely associated with endometriosis. In addition, direct malignant transformation from endometriosis to HGSC is extremely rare. There are only two previous reports of primary HGSC in rectal endometriosis. 6, 7 No previous study has reported an association between the malignant transformation of ligament endometriosis and HGSC. Here, we report two unusual cases of HGSC arising from uterosacral ligament endometriosis. The two cases were misdiagnosed as endometriosis before the operation. We present the two cases in accordance with the CARE reporting checklist. 8 The recognition of HGSC from endometriosis may further expand the understanding of the origin of ovarian type II carcinoma.
Case presentation
Case 1
Clinical history
The first case is a 39-year-old premenopausal Chinese woman, gravida 1, para 1, with a history of dysmenorrhea for 20 years. She had aggravated sacrococcygeal pain for more than 4 months. A solid mass between the rectum and the vagina and congenital uterus duplex was found on magnetic resonance imaging (MRI) at a nearby hospital. Her past medical history indicated that she underwent an open myomectomy because of abdominal pain at the local hospital in 1996. During the operation, she was found to have a congenital deformity of a double uterus and double cervix. She underwent a cesarean section in 2016.
The patient came to our hospital for deep infiltrating endometriosis (DIE) in August 2019. Her cancer antigen 125 (CA125) and human epididymis protein 4 (HE4) levels were 151.7 U/mL and 104.00 pmol/L, respectively. Her pelvic MRI study showed the following: 1) a 26-mm × 35-mm left posterior pelvic mass, indicating neoplastic lesions or endometriosis; 2) a double horn uterus and double cervix deformity; and 3) vaginal and rectal lesions, suggesting endometriosis (Figure 1). Ultrasound showed the absence of the left kidney.
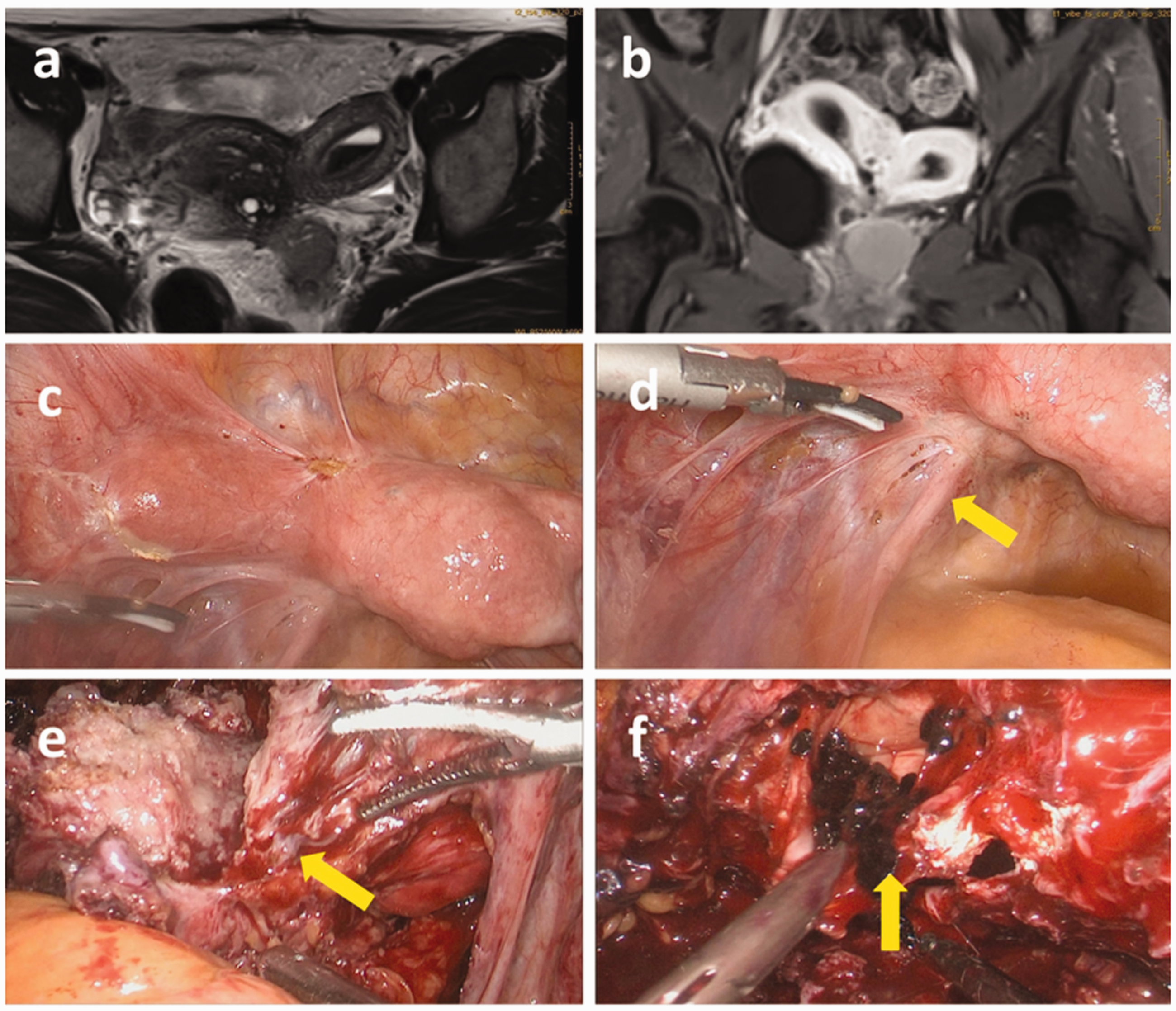
Magnetic resonance images (MRI) and laparoscopic findings in case 1. MRI showed a double uterus and a mass in the left posterior pelvis. The mass was composed mainly of solid components and showed a slightly low signal on T1-weighted imaging (a), an uneven slightly high signal on T2-weighted imaging (b), and uneven enhancement on an enhanced scan. There was hematocele in the left uterine cavity. Laparoscopy found a double uterus in the pelvis (c). The tumor in the left uterosacral ligament is indicated by the yellow arrow (d, e) and The oblique septum on the left side of the vagina is indicated by the yellow arrow (f).
Considering DIE, she underwent laparoscopy on 15 August 2019. The hysteroscopy was performed first. Only one cervix was observed in the vagina, and the cervix was slightly small. One uterine cavity was observed, and only one oviduct opening was found on the right side of the uterus. During the laparoscopic surgery, a double uterus was found (Class U3b, according to the ESHRE/ESGE classification system 9 ). However, the left uterus was considerably smaller. Her left ovary and fallopian tube were absent. The mass was in the left uterosacral ligament, with a diameter of 4 cm (Figure 2). The mass tightly adhered to the posterior wall of the uterus and rectum. When the left anterior wall of the vagina was incised, chocolate cyst fluid flowed out. An oblique septum on the left side of the vagina was also observed. The other cervix (left) was found above the septum. Therefore, the patient had two cervixes: the right one was what we observed during hysterectomy, and the left one was located behind the oblique septum on the left side of the vagina. Samples of the uterosacral ligament mass were collected by laparoscopy and sent for frozen sections. After three frozen biopsies, the mass was finally confirmed to be epithelial cancer. Thus, the patient underwent open cytoreductive surgery. Combined resection of the mass, uterus, right tube and ovary, part of the rectum, pelvic and para-aortic lymph nodes and omentum was performed. Preventive ileostomy was performed.

Histopathological findings of the tumor in the left uterosacral ligament and rectum in case 1. Hematoxylin and eosin staining of the high-grade serous cancer (HGSC) in the left uterosacral ligament at low magnification, 100×. The yellow arrow indicates the remote hemorrhage and transition from endometriotic tissue to a cancerous lesion (a). HGSC in the left uterosacral ligament at high magnification, 100× (b). HGSC in the seromuscular layer of the rectum, 20× (c). HGSC in the seromuscular layer of the rectum, 100× (d). Scale bars = 0.25 mm for 20× and 40 µm for 100×.
Pathology
The final pathology demonstrated HGSC arising from endometriosis in the left uterosacral ligament. The serosa of the rectum was also involved. Immunohistochemistry results showed paired box gene 8 (PAX-8) (+), cytokeratin 7 (CK7) (+), P53 (+, 80%), Wilms tumor protein (WT1) (+), P16 (+), CA125 (+, partly), estrogen receptor (ER) (+, 20%), progesterone receptor (PR) (+, 3%), mesothelial cell (MC) (+, slightly), CK5/6 (+, a few), napsin A (−), hepatocyte nuclear factor-1β (−), calretinin (CR) (−), vimentin (−), CK20 (−) and Ki-67 (+, 70%). Adenomyosis was found in the uterus. No tumor was detected in the uterus, right ovary, fallopian tube, omentum or lymph nodes. Genomic analysis of the tumor sample using an OncoDeficiency® panel (Precision Scientific, Beijing, China) found a missense mutation in exon 7 of the BRCA1 gene named NM_007294.3 (BRCA1): c.446A > C (p.Glu149Ala). No homologous recombination deficiency was present.
Adjuvant therapy and follow-up
A final diagnosis of stage IIB HGSC resulting from left uterosacral ligament endometriosis was made based on the International Federation of Gynecology and Obstetrics staging system. The concomitant diagnosis included Herlyn–Werner–Wunderlich syndrome (U3b/C3/V2, according to the ESHRE/ESGE classification system), pelvic endometriosis and adenomyosis. She received six cycles of chemotherapy with a regimen of paclitaxel (175 mg/m2) and carboplatin (area under the curve =5 mg/mL/min) and underwent follow-up. She received enterotomy anastomosis 6 months later. Her CA125 level was 14 U/mL, and computed tomography results were normal in April 2022. She has remained disease-free for >29 months.
Case 2
Clinical history
The patient was a 52-year-old postmenopausal Chinese woman, gravida 1, para 1. She had been postmenopausal for 5 years and had vaginal bleeding for 2 months. She had dysmenorrhea for over 15 years before menopause. Her past medical history indicated that she underwent laparoscopic right ovarian cystectomy for endometriosis in 2011. Her family history included her father with nasopharyngeal cancer and her mother with breast cancer.
The patient was admitted to our hospital for postmenopausal vaginal bleeding in March 2021. Her CA125 and HE4 levels were 78.9 U/mL and 133.90 pmol/L, respectively. Ultrasound showed a posterior uterine mass. MRI showed an irregular soft tissue mass in the lower right ureter with an unclear boundary of approximately 3.2 × 2.7 × 3.6 cm (Figure 3). Right hydronephrosis and upper right ureter dilation were present. Single-photon emission computed tomography revealed that the glomerular filtration rate of the right kidney was 13.7 mL/min, and that of the left kidney was 82.7 mL/min, indicating atrophy of the right kidney and severely impaired renal function.

Imaging findings in case 2. Magnetic resonance imaging (MRI) showing an irregular soft tissue mass (yellow arrow) in the lower right ureter on T1-weighted images. Right hydronephrosis and upper right ureter dilation were present (a) and Single-photon emission computed tomography (99m Tc-DTPA 6mCi imaging) showing a low level extension line for the right kidney, which demonstrated atrophy of the right kidney and severely impaired renal function. The green line indicates the right kidney, and the red line indicates the left one (b).
The patient underwent laparoscopy on 12 March 2021. During the operation, a cystic and solid mass of approximately 6 × 5 × 5 cm was located in the right uterosacral ligament and adhered tightly to the rectum (Figure 4). Dilatation of the upper segment of the right ureter was found. The lower ureter was narrow and surrounded by a tumor. Samples of the mass were obtained, and frozen sections were made. The pathology of frozen-section biopsies could not differentiate a tumor from endometriosis. Therefore, laparoscopic resection of DIE, right segmental resection of the ureter with bladder implantation, partial rectal resection and anastomosis, right uterosacral ligament mass dissection and laparoscopic hysterectomy and bilateral adnexectomy were performed.

Histopathological findings of the tumor in the right uterosacral ligament and rectum in case 2. Hematoxylin and eosin staining of high-grade serous cancer (HGSC) in the right uterosacral ligament at low magnification, 20 × (a). HGSC in the right uterosacral ligament at high magnification, 100× (b). HGSC in the seromuscular layer of the rectum, 40× (c). HGSC in the seromuscular layer of the rectum, 100× (d). The yellow arrows in (a) and (b) indicate a transition from endometriotic tissue to a cancerous lesion. Scale bars = 0.25 mm for 20×, 125 µm for 40× and 40 µm for 100×.
Pathology
The final pathology confirmed HGSC in the right uterosacral ligament, surrounded by endometriosis. HGSC and endometriosis were also observed in the serosa of the rectum. Adenomyosis was found in the uterus and cervix. No tumor was detected in the uterus, bilateral ovaries or fallopian tubes. Immunohistochemistry results indicated CK (+), CK7 (+), PAX-8 (+), P16 (+), WT1 (+), vimentin (+), PAX-2 (+), ER (+, ∼10%), PR (−), CD10 (−), P53 (−) and Ki-67 (+, ∼70%). No homologous recombination deficiency was found by Genomic Scar Analyses of the tumor sample (Beijing Genomics Institution, Beijing, China) with a score of <1.
Adjuvant therapy and follow-up
The patient and her family refused restaging surgery. According to preoperative imaging and intraoperative findings, the final diagnosis of stage IIB HGSC (clinical stage) resulting from right uterosacral ligament endometriosis was made. The concomitant diagnosis included pelvic endometriosis and adenomyosis. Adjuvant treatment with paclitaxel (175 mg/m2), carboplatin (area under the curve = 5 mg/mL/min) and bevacizumab (7.5 mg/kg) was given intravenously for six cycles. To date, she has completed six cycles of chemotherapy and is still receiving bevacizumab maintenance therapy. Her last CA125 level was 7.1 U/mL, and her computed tomography results were normal in December 2021. There has been no sign of recurrence to date.
Discussion
Endometriosis is one of the most common gynecologic diseases, but the rate of malignant transformation in endometriosis is relatively low. 10 Moreover, the most frequent histotype of epithelial cancer developing from endometriosis has an endometrioid or clear cell-type histology and is defined as endometriosis-associated cancer. HGSC has a different oncological origin, and direct malignant transformation from endometriosis to HGSC is extremely rare. The HGSC in our cases arose from endometriosis, particularly in extraovarian sites, such as the uterosacral ligament. To our knowledge, these are the first two reported cases of HGSC arising from the uterosacral ligament, and they might support a new concept regarding the origin of HGSC.
The criteria required to diagnose a malignant tumor originating from endometriosis were described by Sampson in 1925. 11 The criteria include: (i) the demonstration of both cancer and endometriosis in the same ovary; (ii) the cancer must arise from the endometriosis and not invade it from another site; and (iii) it must be possible to recognize the characteristic histological structures of endometriosis in both the glands and surrounding stroma. In 1953, Scott added an even stricter criterion: the morphological demonstration of continuity between the benign and malignant epithelium within endometriosis. 12 In 2004, Van Gorp et al. defined endometriosis-associated ovarian cancer and classified the coexistence of endometriosis and ovarian cancer into three categories. 13 Cancers meeting Sampson and Scott’s criteria belonged to category A, and those showing a histological pattern compatible with an origin in endometriosis belonged to category C. In the review by Van Gorp et al., no studies meeting Sampson and Scott’s criteria for serous and mucinous carcinoma were identified. The aggressive growth pattern of HGSC, which destroys the endometriotic tissue from which it arises, might be part of the reason. In our cases, the diseases of both patients met Sampson and Scott’s criteria. Endometriotic invasion of >5 mm of the peritoneal surface, which is termed DIE, was noted. 14 In both cases, HGSC in the uterosacral ligament was contiguous with endometriosis, and in the second case, there was even infiltration throughout the right ureter. Additionally, we found no tumors in the fallopian tubes or ovaries during the operation or pathological analyses. Therefore, both cases were diagnosed as malignant transformation caused by uterosacral DIE.
Although the etiology of endometriosis is unclear, Sampson’s retrograde menstruation theory that endometriosis originates from the implantation and invasion of menstrual endometrial cells due to reflux through the fallopian tubes into the peritoneal cavity is widely accepted. 7 Estrogen stimulation and chronic inflammation might stimulate abnormal growth of ectopic endometrial tissue. 15
Patients with endometriotic malignant transformation usually have a long-term history of endometriosis or endometriosis-associated infertility. 16 Our reported cases confirmed this theory. The first patient in our study had oblique vaginal septum syndrome and dysmenorrhea for over 20 years due to poor menstrual blood drainage, and the second patient had a long-term history of dysmenorrhea.
The dualistic model of epithelial ovarian carcinogenesis was proposed in 2004 and is generally accepted. 17 These two types of ovarian cancer have different precursors. Endometrioid and clear cell carcinoma, which are included in the type I category, are the most frequent morphological subtypes associated with endometriosis. 18 In contrast, HGSC (referred to as type II cancer) is rarely associated with endometriosis. Both the fallopian tube epithelium and ovarian surface epithelium are candidates for the origin of HGSC cells. 19 However, based on the findings during the operation and pathological analyses in our cases, there were no carcinomas in either the fallopian tube epithelium or ovarian surface epithelium, suggesting that the tumor originated only from the uterosacral ligament. Therefore, HGSC from endometriosis might indicate a new origin of ovarian type II carcinoma.
The neoplastic transformation of extraovarian endometriosis frequently involves the rectovaginal septum, colon and vagina, accounting for >50% of cases. 20 These locations are most commonly affected by DIE. 21 In our cases, the tumors were located in the uterosacral ligaments, and one even involved the serosa of the rectum.
Preoperatively diagnosing the malignant transformation of endometriosis is difficult because of a lack of sensitive tumor markers. Recently, Anglesio et al. reported that endometriosis without cancer was associated with somatic mutations in ARID1A, PIK3CA, KRAS or PPP2R1A genes, all of which are cancer-driver mutations. 22 These mutations may lead to extraovarian endometriosis-related carcinomas, such as those from the vagina, fallopian tube or mesosalpinx, pelvic sidewall, colon or parametrium. However, neither of our patients showed any somatic mutations, which suggested sophisticated mechanisms of their endometriosis-related carcinomas.
Considering the rarity of the malignant transformation of endometriosis at extraovarian sites, optimal management of these tumors is not well defined. 20 However, when feasible, primary surgical treatment should involve complete resection of all disease-containing tissue. Nevertheless, the adjuvant therapy regimens were not uniform and depended on patient and tumor characteristics. Considering the aggressive behavior of HGSC, after multidisciplinary team discussion, the postoperative chemotherapy regimen of carboplatin plus paclitaxel was administered in accordance with the National Comprehensive Cancer Network ovarian cancer guideline. 23 The oncological outcomes for endometriotic malignant transformation are favorable, with a 5-year survival rate of approximately 80% reported in the literature. 24 Lymph node metastasis and peritoneal dissemination have been reported to be poor prognostic factors. 24 Long-term follow-up is imperative to adequately assess the outcome of these patients with HGSC. The mechanism of HGSC arising from endometriosis needs to be explored in the future.
There are some limitations to our study. We did not provide a photograph of the gross specimens because they were not properly preserved. We did not collect frozen samples during operation to make whole-exome sequencing (WES) of different components of the tumor. The small sample size of this rare disease made the definition of the optimal treatment difficult. More research needs to be done on this rare and challenging gynecological malignancy.
Conclusions
Here, we report two rare cases of HGSC arising in the uterosacral ligament, notably transforming from DIE lesions, which are the first two reported cases worldwide. These cases might indicate a new origin of ovarian type II carcinoma. Our findings show that patients with a long-term history of endometriosis and sudden aggravation of dysmenorrhea or vaginal bleeding should be aware of the possibility of endometriotic malignant transformation. The main treatment for this disease is optimal surgery followed by chemotherapy. Long-term follow-up is necessary.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221109373 - Supplemental material for Primary high-grade serous cancer arising from uterosacral ligament endometriosis: two case reports
Supplemental material, sj-pdf-1-imr-10.1177_03000605221109373 for Primary high-grade serous cancer arising from uterosacral ligament endometriosis: two case reports by Ming Chen, Yuedi Zhang, Manman Xu, Duo Liu, Yang Yang and Shuzhong Yao in Journal of International Medical Research
Footnotes
Author contributions
Conceptualization: Ming Chen and Shuzhong Yao. Data curation: Ming Chen, Yuedi Zhang, Manman Xu, Duo Liu and Yang Yang. Writing-original draft: Ming Chen, Yuedi Zhang. Writing-review & editing: Ming Chen and Shuzhong Yao.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The study was granted ethical exemptions by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University because it is a case report. Written informed consent forms were obtained from the two patients for the publication of this paper and accompanying images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CSCO Cancer Research Foundation (Y-sy2018120) and Beijing Kanghua Foundation (KH-2021-LLZX-052).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
