Abstract
The inferior vena cava, the principal retroperitoneal venous trunk, carries deoxygenated blood from the lower extremities, pelvis, and abdominal viscera to the right atrium. Complex inferior vena cava malformations can radiologically mimic pulmonary space-occupying lesions, substantially complicating clinical diagnosis and therapeutic planning. We present the case of a patient in whom contrast-enhanced chest computed tomography incidentally revealed a right-sided intrathoracic mass, initially suspected to be a primary or metastatic pulmonary malignancy. Subsequent dynamic contrast-enhanced computed tomography demonstrated heterogeneous, avid enhancement during the venous phase, raising the possibility of a vascular anomaly. Dedicated magnetic resonance imaging ultimately delineated the mass as a congenital inferior vena cava malformation. This case underscores that intricate inferior vena cava anomalies may masquerade as intrapulmonary masses; misguided surgical intervention could precipitate severe hemorrhagic or thromboembolic sequelae. Multimodality imaging—integrating high-resolution computed tomography and magnetic resonance imaging—is therefore indispensable for accurate differential diagnosis.
Keywords
Introduction
The widespread adoption of high-resolution computed tomography (CT) has markedly increased the detection rate of pulmonary space-occupying lesions. Such findings universally prompt immediate concern for primary or secondary malignancy, representing a common and challenging scenario in contemporary pulmonary and thoracic surgical practice. 1 Current diagnostic algorithms therefore prioritize exclusion of primary lung cancer or metastatic disease, typically necessitating a stepwise combination of advanced imaging and, frequently, invasive tissue-sampling procedures. 2
Although neoplastic processes dominate clinical suspicion, the differential diagnosis of a pulmonary mass is broad and encompasses infectious, inflammatory, and congenital benign entities. Among these, pulmonary vascular malformations (PVMs) and other vascular anomalies—including arteriovenous malformations, cavernous hemangiomas, and inferior vena cava (IVC) variants—constitute a rare yet clinically significant subset.3,4 Owing to their propensity for intense contrast enhancement, lobulated contours, spiculated margins, or associated ground-glass opacities, these vascular lesions can closely mimic malignant tumors on imaging, thereby posing substantial diagnostic challenges.
Preoperative identification of such tumor-mimicking vascular malformations remains exceedingly uncommon and is fraught with difficulty. Misdiagnosis may precipitate inappropriate percutaneous transthoracic needle biopsy or unwarranted surgical resection; given the inherent hypervascularity of these lesions, such interventions carry a high risk of catastrophic hemorrhage and potentially fatal outcomes. 5
We herein present a paradigmatic case in which a complex congenital IVC malformation manifested as a highly suspicious pulmonary mass on chest CT, ultimately elucidated solely through multimodality imaging. This report aimed to heighten awareness among clinicians, radiologists, and thoracic surgeons regarding vascular anomalies masquerading as pulmonary neoplasms. We emphasize their distinctive radiologic features and differential diagnostic indications to avert severe iatrogenic complications and guide optimal diagnostic and therapeutic strategies. The novelty of this case lies in its exceptionally rare clinical presentation (a pulmonary mass as the initial manifestation) and the complexity of the malformation (hepatic segment of the IVC anomalously draining into the right inferior pulmonary vein territory with accompanying azygos vein dilation). It offers invaluable imaging experience for differential diagnosis and underscores the decisive role of multimodality imaging and multidisciplinary collaboration in averting misdiagnosis and inappropriate treatment.
Case presentation
In October 2024, a male patient in his early 20s presented with a 4-day history of cough and expectoration at the Department of Cardio-Thoracic Surgery of Chengdu Second People’s Hospital, Sichuan, China. Four days prior to admission, he developed an intermittent cough productive initially of scant, watery, white sputum that subsequently became yellowish and tenacious, increasing to a volume of approximately 10 mL per day. He denied fever, rigors, night sweats, hemoptysis, chest tightness, dyspnea, palpitations, or chest pain. A noncontrast chest CT obtained at an outside facility revealed a well-circumscribed, lobulated soft-tissue mass in the right hemithorax measuring 5.9 cm × 4.6 cm × 6.3 cm. The patient reported no weight loss and was otherwise in good general condition.
His medical history was notable for nasal surgery for rhinitis conducted 2 years ago. He denied allergies, trauma, transfusions, or significant family history. He was a nonsmoker and a full-time student.
On admission, the patient was afebrile (36.3°C), hemodynamically stable (blood pressure (BP), 120/86 mmHg), with a regular pulse of 96 bpm and a respiratory rate of 19 breaths per minute. Physical examination revealed mild thoracic asymmetry with subtle leftward prominence. Auscultation demonstrated symmetric breath sounds bilaterally, although slightly diminished on the right; no rales, rhonchi, or pleural friction rubs were noted. The cardiac apex was palpable in the right fourth intercostal space; heart sounds were normal and regular without murmurs or pericardial rub.
The results of laboratory tests, including complete blood count, comprehensive metabolic panel, procalcitonin, coagulation profile, erythrocyte sedimentation rate, and tumor markers (carcinoembryonic antigen (CEA), neuron-specific enolase (NSE), squamous cell carcinoma antigen (SCCA), cytokeratin-19 fragment (CYFRA21-1), and pro-gastrin-releasing peptide (ProGRP)), were all within normal limits. These tumor markers are intended to screen for common primary lung malignancies. CEA is a broad-spectrum marker for adenocarcinoma and other cancers; NSE is associated with small-cell lung cancer; SCCA targets squamous cell carcinoma; CYFRA21-1 shows high sensitivity for non–small-cell lung cancer, especially squamous types; and ProGRP is highly specific for small-cell lung cancer. Combined measurement of these markers provides a serologic first step to exclude malignant pulmonary masses. Electrocardiography showed sinus rhythm with abnormal Q waves in leads I and aVL. Transthoracic echocardiography confirmed dextro-position of the heart without structural or functional abnormalities; left-ventricular ejection fraction was preserved.
Contrast-enhanced chest CT (Figure 1(a) to (c)) demonstrated a large solid mass adjacent to the right hilum. The right lung lacked discernible fissures and was markedly hypoplastic; the right middle-lobe bronchus could not be identified on sequential images. The mediastinum and heart were displaced rightward. No pleural effusion was present.

(a) A rounded, perihilar mass was present in the right lung (yellow arrow). (b) Yellow arrows indicate the orifices of the left and right upper-lobe bronchi and (c) green arrows indicate the orifices of the left and right lower-lobe bronchi.
Mediastinal-phase CT (Figure 2(a)) revealed mild heterogeneous enhancement of the mass with small feeding vessels. Venous-phase imaging (Figure 2(b)) showed intense, persistent enhancement. Contrast-enhanced CT venous-phase three-dimensional (3D) reconstructions (Figure 2(c) and (d)) clearly depicted the exact location of the lesion and its adjacent structures, explaining why it appears as a “mass” on axial images. Multiplanar reconstruction (Figure 3) demonstrated dilated vascular channels coursing leftward to the left atrium, closely associated with the right inferior pulmonary vein.

(a) Arterial-phase contrast-enhanced CT. The lesion showed mild enhancement; the yellow arrow highlights a small opacified vessel. (b) Venous-phase contrast-enhanced CT. The lesion exhibited marked heterogeneous enhancement. (c) Contrast-enhanced CT venous-phase coronal reconstruction diagram. The red arrow indicates the lesion and (d) contrast-enhanced CT venous-phase sagittal reconstruction diagram: The red arrow indicates the lesion. CT: computed tomography.

In the left-sided region of the lesion, an enlarged vascular structure was seen draining into the left atrium and was closely related to the right inferior pulmonary vein (yellow arrow).
Subsequent contrast-enhanced magnetic resonance imaging (MRI) (Figures 4(a) and 5(b)) delineated multiple tortuous, enlarged vessels. The intrahepatic segment of the IVC was absent; instead, the infrarenal IVC drained into the mediastinal mass, which ultimately emptied into the hepatic veins and right inferior pulmonary vein. MRI further revealed an enlarged azygos vein (Figure 4(b)), consistent with azygos continuation of the IVC.
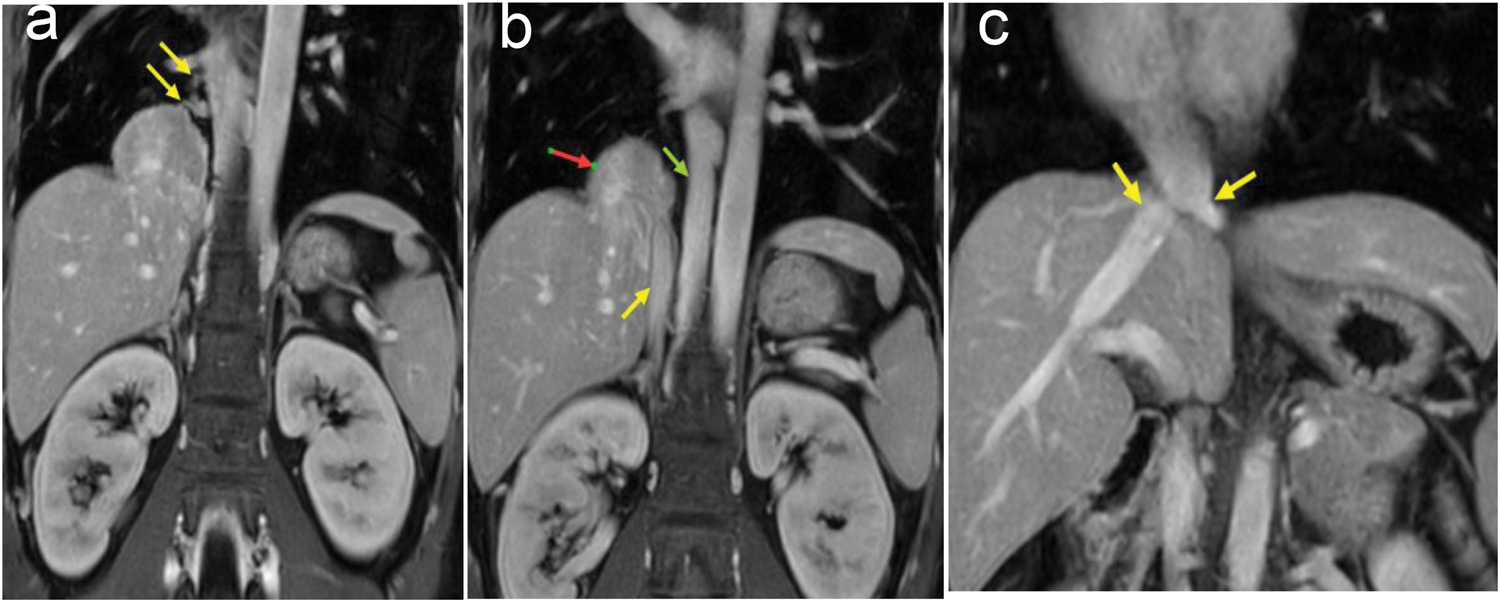
MRI coronal plane. (a) The yellow arrow indicates that the lesion is closely related to the right inferior pulmonary vein. (b) The red arrow indicates the lesion; the yellow arrow indicates the inferior vena cava; and the green arrow indicates the dilated azygos vein. (c) The yellow arrow indicates the hepatic venous tributary draining into the inferior vena cava. MRI: magnetic resonance imaging.
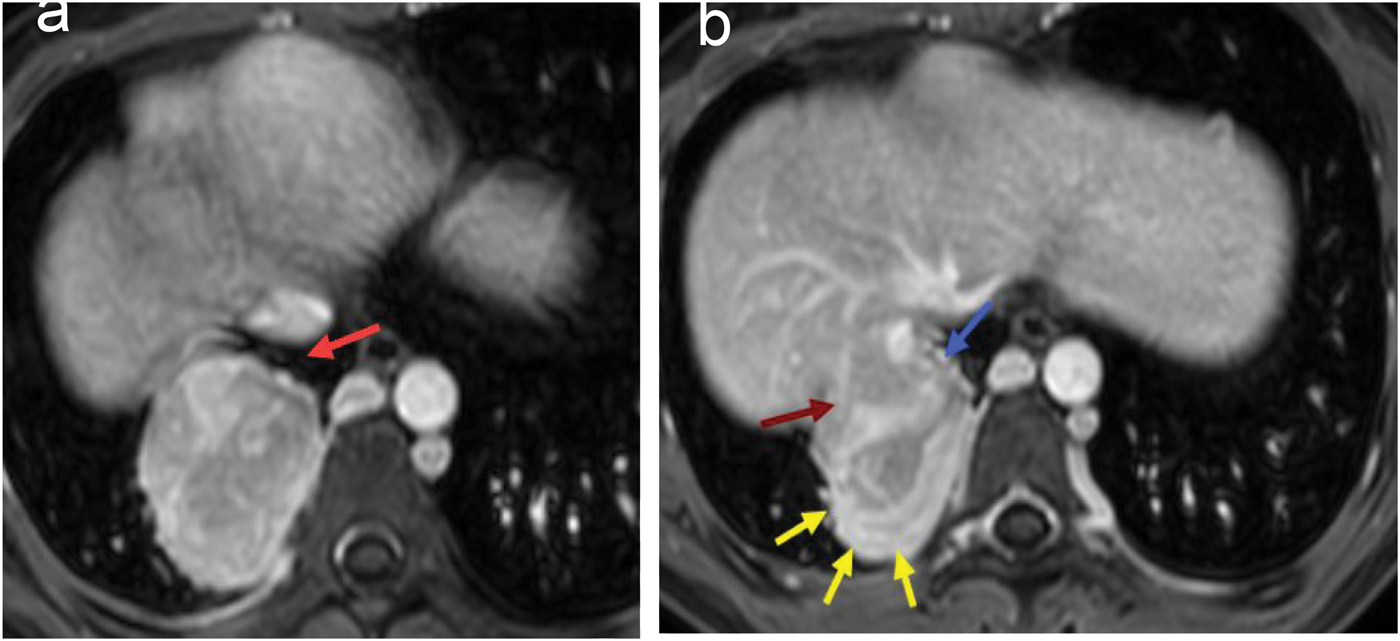
MRI transverse plane. (a) The red arrow indicates a distinct gap between the suprahepatic segment of the inferior vena cava and the lesion and (b) the yellow arrow indicates multiple dilated and tortuous vascular shadows; the red arrow indicates that the lesion is closely related to the hepatic veins; and the blue arrow indicates the anomalous morphology of the hepatic segment of the inferior vena cava. MRI: magnetic resonance imaging.
Multidisciplinary consultation among the thoracic surgery, vascular surgery, hepatobiliary surgery, and radiology departments concluded the diagnosis of (a) congenital complex IVC malformation with azygos continuation and (b) right pulmonary hypoplasia (developmental anomaly).
Given the absence of symptoms and the benign nature of the malformation, conservative management with annual serial imaging surveillance was recommended. The patient and his family were thoroughly counseled regarding potential complications and warning symptoms. He was discharged in stable condition for outpatient follow-up. The reporting of this study conforms to the Case Report (CARE) guidelines. 6 All patient details have been de-identified from this case report, and patient consent to treatment was obtained.
Discussion
The IVC is the principal retroperitoneal conduit for systemic venous return, collecting deoxygenated blood from the lower extremities, pelvis, and a substantial portion of the abdominal viscera and delivering it to the right atrium. Under normal circumstances, the IVC is formed by the confluence of the right and left common iliac veins anterior to the fifth lumbar vertebra. It ascends along the right anterolateral aspect of the spine; courses dorsal to the duodenum, portal vein, and liver; penetrates the central tendon of the diaphragm at the T8 level; and, after a brief intrathoracic course, enters the right atrium. 7
Embryologically, the definitive venous system is formed between the 6th and 8th gestational weeks8 and is derived from three paired embryonic veins: vitelline, umbilical, and cardinal systems. The superior vena cava arises from the right anterior cardinal vein, with well-recognized anomalies including persistent left superior vena cava and duplicated caval channels, while the IVC is assembled from multiple segments of the cardinal system. Classically, the IVC is subdivided into five developmental components: 9 (a) infrarenal (iliac) segment, derived from the right supracardinal vein; (b) renal segment, formed by anastomosis between the right sub- and supracardinal veins; (c) suprarenal segment, derived from the right subcardinal vein; (d) hepatic segment, arising from the right vitelline (hepatocardiac) channel; and (e) intrahepatic segment, which is a continuation of the hepatic veins into the right atrium.
Anatomic variants of the IVC are encountered in approximately 4% of the general population 10 and result from aberrations in regression, fusion, or persistence of the embryonic venous channels.
In the present case, contrast-enhanced CT and MRI demonstrated the absence of normal intrahepatic IVC. Instead, the infrarenal IVC drained into a complex, tortuous mediastinal venous plexus that ultimately emptied into the right inferior pulmonary vein and hepatic veins. Concurrent severe dilatation of the azygos system was evident. The azygos vein normally serves as a collateral pathway between the superior and inferior caval systems; in the setting of IVC interruption or obstruction, it becomes a critical venous decompressive channel. 11
We interpreted the hepatic segment anomaly as a congenital IVC malformation with azygos continuation, most likely attributable to failed formation or premature regression of the right subcardinal–hepatocardiac anastomosis. Chronic obstruction of the IVC with subsequent recruitment of the azygos–hemiazygos system cannot be entirely excluded; however, the absence of clinical signs of caval hypertension—including hepatic congestion, lower-extremity edema, or cutaneous collateral circulation—strongly indicates a developmental rather than acquired etiology.
Budd–Chiari syndrome (BCS) is defined as any obstruction to hepatic venous outflow situated between the terminal hepatic venules and the right atrial orifice of the inferior vena cava. 12 Anatomically, the obstruction may be classified as suprahepatic IVC, hepatic vein, or combined (mixed) type. 13 The resultant outflow block elevates sinusoidal pressure, producing post-sinusoidal portal hypertension. 14 In the acute phase, diminished portal inflow may precipitate portal vein thrombosis; chronically, persistent venous stasis and hypoxia provoke centrilobular necrosis, fibrosis, nodular regeneration, and ultimately cirrhosis. 15 Clinically, patients with BCS present with abdominal pain, hepatosplenomegaly, and ascites; when the IVC itself is stenosed, additional signs such as flank or back varices and bilateral lower-extremity edema may appear. 16
In the present case, although the malformation existed in close proximity to the right hepatic vein, high-resolution MRI sequences (Figures 4(c) and 5(a)) demonstrated unobstructed hepatic venous drainage, with no evidence of luminal narrowing, thrombosis, or extrinsic compression. Moreover, the patient lacked any clinical or biochemical markers of BCS; specifically, no abdominal pain, ascites, peripheral edema, or abnormal liver function tests were noted. Consequently, BCS was excluded with a high degree of certainty. To further highlight the clinical value and educational significance of this case, its key features are compared with those of previously reported similar cases in Table 1.
Comparison of different cases of inferior vena cava malformations in terms of clinical imaging characteristics and management.

IVC: inferior vena cava; CT: computed tomography; MRI: magnetic resonance imaging.
Pulmonary sequestration (PS) is a rare congenital pulmonary malformation, accounting for 0.15%–6.4% of all congenital lung anomalies, characterized by nonfunctional pulmonary tissue that lacks normal bronchial communication and receives an aberrant systemic arterial supply. 17 In the present case, although chest CT confirmed right lung hypoplasia, contrast-enhanced imaging demonstrated no identifiable systemic feeding vessel supplying the lesion; therefore, PS was excluded. In contrast, the uniqueness of the present case lies in its initial presentation as a deceptive “pulmonary mass,” a pattern markedly different from the usual asymptomatic detection prompted by unrelated symptoms of IVC anomalies. The complexity of the malformation, involving the hepatic segment of the IVC, hepatic veins, pulmonary veins, and azygos system, together with ipsilateral pulmonary hypoplasia, is exceptionally rare. Successful diagnosis underscores the critical value of scrutinizing the venous-phase enhanced CT and employing MRI for differentiation when a pulmonary neoplasm is suspected, thereby providing essential evidence to avoid high-risk invasive procedures.
Conclusion
In summary, the final diagnosis in this case was a complex congenital malformation of the IVC accompanied by right pulmonary hypoplasia. Such anomalies are exceptionally rare and anatomically intricate; precise characterization and broad differential diagnosis are therefore imperative. Multidisciplinary collaboration is essential to obtain an accurate diagnosis. Misidentification as an intrapulmonary neoplasm followed by an unwarranted resection could precipitate catastrophic hemorrhage and life-threatening complications.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251391919 - Supplemental material for Complex inferior vena cava malformation initially manifesting as a pulmonary space-occupying lesion: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605251391919 for Complex inferior vena cava malformation initially manifesting as a pulmonary space-occupying lesion: A case report by Shuang Feng, Xiaowei Wang, Maosen Zhao, Yongjie Guan, Yi Dong, Mingming Wang and Ke Gao in Journal of International Medical Research
Footnotes
Acknowledgments
All authors contributed equally to this manuscript and are considered as first co-authors.
Author contributions
SF: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
XW: Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing–original draft, Writing–review & editing.
MZ: Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing–original draft, Writing–review & editing.
YG: Conceptualization, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
YD: Conceptualization, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
MW: Conceptualization, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
KG: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
Availability of data and materials
Not applicable; please contact the corresponding author for data requests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The protocol was approved by the Chengdu Second People’s Hospital Clinical Research Ethics Committee. Written informed consent was obtained from the participant for the publication of this case report.
Funding
Not applicable.
Statement of competing interests
The authors declare that they have no competing interests.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
