Abstract
Objective
Most men suffering from depression have different degrees of erectile dysfunction (ED), but the relationship between depression and ED is not clear. This study explored the effect of depression on erectile function in rats and the underlying mechanism.
Methods
The potential targets and key signaling pathways of depression and ED were predicted through bioinformatics analysis, and a depression rat model was established by inducing chronic restraint stress. Pathological changes in rat penis tissue were studied by hematoxylin and eosin staining. The serum dopamine level was quantified by an enzyme-linked immunosorbent assay. The expression of related proteins and mRNA was detected by western blotting and real-time quantitative reverse transcription-polymerase chain reaction.
Results
Hematoxylin and eosin staining showed pathological damage in the penile tissue of the model group rats. The serum dopamine level, dopamine receptor D2 (DRD2) and solute carrier family 6 member 3 (SLC6A3) protein levels in penile tissue, and DRD2 and SLC6A3 mRNA levels were lower in the model group than in the control group.
Conclusion
The decrease in erectile function in the depression rat model was related to dysfunction of the dopamine system and dopaminergic synapse signaling pathway.
Keywords
Introduction
Depression, as one of the most common psychological diseases, is characterized by slow speech and movement, mental retardation, loss of interest in work, self-guilt, self-consciousness, feelings of worthlessness, and even suicidal tendencies. The prevalence of depression in Chinese adults is approximately 6.9%. 1 Although depression has always been a global research hotspot, its etiology and pathogenesis remain uncertain. Erectile dysfunction (ED) refers to a common male sexual dysfunction in which the penis cannot continuously achieve or maintain sufficient erection to obtain satisfactory sexual intercourse. 2 It has a substantial impact on men’s physical and mental health and family harmony. Almost all men suffering from depression have different degrees of ED. 3 Emotional changes can alter the levels of hormones, such as dopamine and catecholamines, and cause hemorheological changes, circulatory disorders, and vascular diseases. 4 Vascular endothelial dysfunction is currently recognized as an important pathological mechanism of ED. 5 The famous Massachusetts aging research data showed that almost all men with depressive symptoms had a certain degree of ED. The incidence rate of moderate to severe ED in patients with depression was 1.82 times that of patients without depression. 6
Given the close relationship between depression and ED, further studying the correlation between depression and ED is essential. Using bioinformatics prediction analysis combined with animal experiments, this study found that the dopaminergic synapse signaling pathway may be the key pathway through which depression causes ED.
Materials and methods
Identification of targets
The GeneCards database (https://www.genecards.org/) 7 and OMIM database (https://omim.org/) 8 were used to identify the targets related to depression and ED. Then, the two targets were crossed to obtain the potential targets of ED caused by depression. These common targets were used for further network construction and analysis.
Protein–protein interaction (PPI) network analysis
The potential targets of ED caused by depression were imported into the STRING database (https://STRING-db.org/), 9 the species was limited to “Homo sapiens,” and the protein interaction relationship was retrieved. Each PPI was screened with the lowest interaction score (unit of 0.40) to ensure the reliability of the data. Targets without a protein interaction relationship were hidden, and the results were saved in the TSV format.
Network construction and analysis
Cytoscape version 3.8.0 software (https://cytoscape.org/) 10 was used to construct a depression–ED–targets network and a PPI network. The PPI network was further analyzed using the Cytoscape plug-in CytoHubba to identify key targets.
Gene Ontology (GO) functional enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis
Using an annotation, visualization, and integrated discovery database (David, https://david.ncifcrf.gov/, version 6.8), the potential targets of depression-induced ED were imported into the functional annotation tool of the David 6.8 database for GO enrichment analysis. The function of the target genes limiting the species to humans (Homo sapiens) was determined. The David 6.8 database was used for KEGG pathway enrichment analysis to further obtain the biological processes and key signaling pathways of depression-induced ED. 11
Ethics approval of the study protocol
All experimental protocols conformed to the requirements, were approved by the Ethics Committee of Medicine and Experimental Animals of Beijing University of Chinese Medicine (No. BUCM-4-2020123303-4150), and performed in accordance with the Guide for the Care and Use of Laboratory Animals, 8th Edition. 12
Experimental animals
Twenty specific pathogen-free grade male Sprague Dawley rats, weighing 220 to 250 g and aged 4 to 5 weeks (Beijing Weitong Lihua Experimental Animal Technology Co., Ltd., Beijing, China, license No. SYXK (Beijing) 2020-0033), were housed in the specific pathogen-free animal laboratory of Beijing University of Chinese Medicine under controlled temperature (23 ± 2°C), humidity (55% ± 10%), and light (12-hour photoperiod) conditions. The rats were provided unrestricted access to food (ordinary full-price nutritional pellet feed) and water.
Reagents and equipment
The following reagents and equipment were used in this study: catechol-O-methyltransferase (COMT) antibody (14754-1-AP; Proteintech, Chicago, IL, USA), dopamine receptor D2 (DRD2) antibody (55084-1-AP; Proteintech), solute carrier family 6 member 3 (SLC6A3) antibody (ab184451; Abcam, Cambridge, UK), glycerol 3-phosphate dehydrogenase (GAPDH) antibody (ab181602; Abcam), SDS-PAGE Gel Kit (GPP1816; GenePool, Beijing, China), BSA Blocking Buffer (GPP1818; GenePool), polyvinylidene fluoride membrane (0.22 µm; Millipore, New York, NY, USA), electrophoresis apparatus (PP-1150; CAVOY, Beijing, China), and double vertical electrophoresis tank (PP-1150; CAVOY).
Groups and model establishment
Twenty male Sprague Dawley rats were randomly divided into the normal group and depression model group, with 10 rats in each group. The internationally recognized chronic restraint stress method was used to construct the depression rat model.13,14 From the first day of the experiment, rats in the model group were placed in a special fine-barbed wire mesh enclosure, which limited their activities without obstructing breathing or inducing oppression, and both ends were fixed with restraint iron clips. The daily restraint time was 9:00 to 15:00. During the restraint period, the rats were forbidden to eat and drink. After the restraint, the rats were placed back into their single-housed cages. The rats were free to eat and drink for 28 days. The blank control group was allowed free feeding and drinking without any restraint stress.
Verification of depression model rats
Sucrose water preference test
Before the experiment, the rats were adapted to drinking sugary water. For the adaptation phase, two drinking water bottles were placed side by side in each cage at the same time. For the first 24 hours, one bottle was filled with 1% sucrose water. For the next 24 hours, one bottle contained 1% sucrose water, and the other bottle contained purified water.
After 24 hours of water and food deprivation, the experiment was started. Each rat was provided two bottles of water weighed in advance. One bottle contained 1% sucrose water, and the other contained purified water. The water bottles were placed randomly on the left and right. After 1 hour, the two bottles were weighed. The sucrose water preference was calculated on days 1, 7, 22, and 28 as follows: sucrose water preference (%) = (sucrose water consumption/total liquid consumption) × 100. 15
Open field test
An open box (80 cm × 80 cm; square bottom) prepared in-house was divided into 25 equal lattices; the box height was 40 cm. The horizontal movement score was based on the number of lattices crossed (a lattice was counted only if all four claws were within that lattice). The vertical movement score was based on the frequency of rearing behavior (1 point was scored if both forelegs were lifted above the ground). Care was taken to keep the environment quiet, appropriately adjust the light intensity, and clean the box before each test to remove any residual smell. Two assessors evaluated the movement of each rat over 3 minutes. This test was conducted before modeling and 1 day before sacrificing the rats and isolating blood and tissue samples. 16
Determination of erectile function in rats
According to the method used by Heaton et al., 17 the two groups of rats were weighed and placed individually in a quiet and dark observation box for 10 minutes. Then, apomorphine (100 µg/kg) was injected subcutaneously into the neck of the rats. The state of the rats in the above environment was photographed by a camera, and the penile erection of the rats was recorded for 30 minutes. One erection episode comprised the swelling or growth of the penis and the exposure of the end of the penis.
Tissue sample preparation
After successful model induction, the rats were weighed and anesthetized with pentobarbital sodium (50 mg/kg, intraperitoneally). 18 The rats were then sacrificed by taking blood from the abdominal aorta. Blood from the abdominal aorta was used to detect the level of serum dopamine, and then the penile tissue was isolated for western blotting, quantitative reverse transcription-polymerase chain reaction (qRT-PCR), and hematoxylin and eosin (HE) staining analyses to observe the changes in tissue morphology. All efforts were made to minimize the number of animals used and decrease their suffering.
Detection of serum dopamine level
Blood from the abdominal aorta was obtained from fasted rats and centrifuged at 3000 g for 15 minutes to obtain the supernatant. An enzyme-linked immunosorbent assay kit (Shanghai Maipu Biotechnology Co., Ltd., Shanghai, China) was used to quantify the serum dopamine level in accordance with the kit instructions.
HE staining
After cleaning the penis tissue, a part of the penis was embedded in paraffin, cut into thin slices (5 μm thick), and then subjected to xylene dewaxing, hematoxylin staining, alcohol differentiation, washing, anhydrous ethanol dehydration, eosin staining, xylene clarification, and sealing with neutral resin. The prepared slides were observed and imaged under an optical microscope equipped with a digital tube (BX51TF; Olympus, Tokyo, Japan).
Western blotting
For western blotting, total protein was extracted from the penile tissue of rats in each group, and the protein samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The protein bands were then electrophoretically transferred to the membrane, blocked, and incubated with the primary antibodies overnight at 4°C. Next, the membrane was washed with Tris-buffered saline with 0.1% Tween 20 three times and then incubated with the secondary antibody overnight at 4°C. After repeat washing, the optical density of the protein bands was quantified using Quantity One v.4.6.2 software (Bio-Rad, Hercules, CA, USA). The relative amount of each protein was expressed relative to the density of GAPDH bands.
qRT-PCR
The penis tissue was added into a mortar along with a small volume of liquid nitrogen and quickly ground into a fine powder. Then, 1 mL of TRIzol reagent (R1000; Lablead, Beijing, China) was added to extract total RNA from the tissue. After checking the quality of the extracted RNA, the RNA was reverse transcribed into cDNA in accordance with the requirements of the reverse transcription kit (GPQ1803; GenePool), and then the target gene product was amplified under the following conditions with a Real-time PCR System (Line Gene 9600 Plus, Bioer Technology, Hangzhou, China): pre-denaturation at 95°C for 15 minutes, denaturation at 95°C for 10 seconds, annealing and extension at 60°C for 30 seconds, and 40 cycles of amplification. The 2−ΔΔCt method was used to determine the relative expression of each gene. GADPH was used as an internal reference. The sequences of primers are shown in Table 1.
The primer sequences for RT-qPCR.

DRD2, dopamine receptor D2; SLC6A3, solute carrier family 6 member 3; GAPDH, glycerol 3-phosphate dehydrogenase.
Statistical analysis
IBM SPSS Statistics for Windows, Version 26 (IBM Corp., Armonk, NY, USA) was used to analyze the measurement data. For normally distributed measurement data, the independent sample t-test was used for comparisons between two groups, and for data not normally distributed, the nonparametric rank-sum test was used. P < 0.05 was considered to indicate a significant difference.
Results
Collection of targets
GeneCards and OMIM searches resulted in 500 depression targets and 500 ED targets. After crossing the two targets, 132 targets related to depression and ED were obtained (Figure 1a).

Bioinformatics analysis. (a) Intersection of targets in depression and erectile dysfunction. (b) Depression–erectile dysfunction–targets network built by Cytoscape. (c) PPI network built by Cytoscape and (d) PPI network processed by the Cytoscape plug-in CytoHubba.
Construction and topological analysis of the depression–ED–targets network and the PPI network
The 106 overlapping targets were further analyzed using the STRING database, and the PPI network and depression–ED–targets network were constructed using Cytoscape (Figure 1b and 1c). According to the CytoHubba analysis, the top 10 targets were regarded as key targets (Table 2 and Figure 1d).
Top 10 key targets.

GO and KEGG enrichment analysis
The results showed that depression and ED involved 618 cell biological processes, 68 cell components, 90 molecular functions, and 99 signaling pathways. The top 10 GO and KEGG pathways based on the P-value are shown in Figure 2.

Analyses of pathway enrichment using GO and KEGG databases. The Y-axis is the name. The X-axis is the richness factor. The size of the node is proportional to the number of genes. The node color is proportional to the P value.
Validation of the depression rat model
Sucrose water preference experiment: There was no significant difference in the sucrose water preference index of rats between the control and model groups on the first day. On the 28th day of the experiment, the sucrose water preference index of rats in the model group was significantly lower than that in the normal group (P < 0.05) (Figure 3a).

Sucrose water preference test and open field test before and after modeling. (a) Sucrose water preference index of rats in the C and M groups. (b) The horizontal movement scores of rats in the two groups and (c) The vertical movement scores of rats in the two groups. Values are the mean ± standard error of the mean (n = 10 animals per group). The t-test was used. Group M was compared with group C, * represents P < 0.05.
Open field test: There was no significant difference in the horizontal and vertical movement scores of rats between the control and model groups on the first day. On the 28th day of the experiment, the horizontal and vertical movement scores of rats in the model group were significantly lower than those in the normal group (P < 0.05) (Figure 3b and 3c).
Number of penile erections
After apomorphine injection, the number of penile erections of rats within 30 minutes was significantly lower in the model group than in the control group (Table 3).
Erectile times of rats in each group.
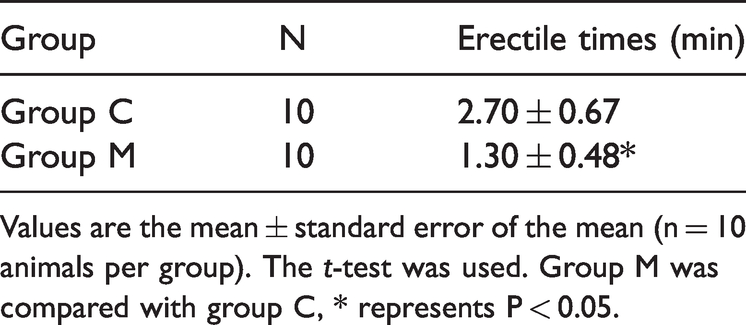
Values are the mean ± standard error of the mean (n = 10 animals per group). The t-test was used. Group M was compared with group C, * represents P < 0.05.
Detection of serum dopamine level
The enzyme-linked immunosorbent assay showed that the level of serum dopamine in the model group was significantly lower than that in the control group (P < 0.05) (Figure 4a and 4b).

Dopamine serum level and histological analysis of penile tissues. (a) The serum level of dopamine in rats in the C and M groups. Values are the mean ± standard error of the mean (n = 10 animals per group). The t-test was used. Group M was compared with group C, * represents P < 0.05. (b) Scatter plot of the serum level of dopamine in rats in the two groups and (c) Analyses of penile tissue in rats using HE staining at the indicated magnifications. The penile tissue of rats was stained with HE to observe pathologic changes in the penis under an optical microscope.
HE staining of penile tissue
Observation of HE-stained sections under a light microscope showed the presence of cavernous trabeculae and evenly distributed blood sinuses in the corpus cavernosum of the penis in both groups. Some red blood cells were observed in the sinus space, and the inner wall of the blood sinuses was covered with endothelial cells. Sinusoidal trabeculae contained abundant smooth muscle, collagen fibers, and some small vessels, and there was no hyperplasia of the interstitial tissue (Figure 4c).
Protein expression of DRD2, SLC6A3, and COMT in the rat penis
Western blotting analysis of protein expression in the penile tissue of rats revealed that the protein expression levels of DRD2 and SLC6A3 in the model group were significantly lower than those in the control group (P < 0.05) (Figure 5a, 5b, and 5c). However, COMT protein expression was significantly increased in the model group (P < 0.05) (Figure 5d).

Analysis of the dopaminergic synapse signaling pathway. (a) Western blotting showing the expression of DRD2, SLC6A3, and COMT proteins. GADPH was used as a loading control. (b), (c), and (d) The light blue bars represent the expression of DRD2, SLC6A3, and COMT in group C. The dark blue bars represent the expression of DRD2, SLC6A3, and COMT in group M. (e) and (f) mRNA expression of DRD2 and SLC6A3 in rat penile samples. Values are the mean ± standard error of the mean (n = 10 animals per group). The t-test was used. Group M was compared with group C, * represents P < 0.05.
mRNA expression of DRD2 and SLC6A3 in the rat penis
The mRNA levels of DRD2 and SLC6A3 in the model group were significantly lower than those in the control group (P < 0.05) (Figure 5e and 5f), indicating that depression significantly alters the dopaminergic synapse signaling pathway in the penile tissue of depression model rats.
Discussion
The fast-paced modern life and increased competitive pressure have resulted in a rising incidence of depression. This common psychological condition imposes a serious burden on families and society.19,20 In addition to affecting the mental health of patients, depression has a significant pathological effect on various systems of the body, including causing ED in men. Several studies have shown that dopaminergic system disorders are closely related to depression and ED.21,22 The results of the present in vivo experiments and bioinformatics analysis showed that the decline in erectile function in the established depression rat model was mediated through the dopaminergic synapse signaling pathway.
In 1999, Sokoloff and Seeman divided DRs into two categories according to the ligand-binding affinity of receptors: D1-like receptors (D1 and D5 receptors) and D2-like receptors (D2, D3, and D4 receptors). 23 D2 receptors (encoded by the DRD2 gene) mainly distributed in the midbrain limbic dopaminergic system are implicated in the pathogenesis of depression. Patients with depression generally show reduced dopamine secretion and impaired DRD2 function.24,25 The dopamine transporter (encoded by SLC6A3) is a presynaptic membrane protein of dopaminergic neuronal terminals. Approximately 80% of the total amount of dopamine is transported into cells through this transporter protein and then released for utilization. The dopamine transporter is exclusively located in dopamine-synthesizing neurons and is considered a unique marker of dopaminergic neurons. A change in its expression reflects the function of the dopaminergic system to a certain extent. 26
After dopamine is secreted into the synaptic space in vesicles from neurons, it specifically binds to DRD2 on the postsynaptic membrane to activate the inhibitory G protein-coupled receptor (Gi). Gi activation inhibits the activity of adenylate cyclase, reducing the intracellular cyclic adenosine monophosphate level. Thus, the level of activated protein kinase A is reduced, followed by decreased phosphorylation of specific serine residues and threonine residues of several proteins. Subsequently, a series of cascade reactions occur, and various physiological effects are observed. 27 COMT transfers a methyl group to the 3- and 4-hydroxyl groups of catecholamine neurotransmitters, including dopamine, to inactivate them. 28 Thus, dopamine bound to the receptor is finally inactivated under the enzymatic hydrolysis of COMT, and its physiological effects are terminated. 29 Depression occurs when the secretion of dopamine, expression of the dopamine transporter, or level of DRD2 is reduced or too much COMT is produced.24–27
The pathological mechanism of ED is multifaceted. Changes in endocrine, nervous, and vascular functions; systemic diseases; local penile diseases; and psychological factors are related to the occurrence and development of ED. 30 DR agonists are a class of drugs that are similar to dopamine in molecular conformation and act directly on DRs. Animal experiments have shown that the binding of dopamine agonists to DRD2 in the hypothalamic paraventricular nucleus leads to penile erection. 31 Moreover, subcutaneous injection of the low-dose semi-synthetic catecholamine alkaloid apomorphine, a D1/D2 receptor dopaminergic agonist, induces penile erection in healthy subjects without sexual stimulation. 32 Therefore, dopamine plays an important role in penile erection.
Antipsychotic treatment in men is associated with a variant of the DRD2 gene, in which the 141C Del allele may be a protective factor. The 141C Ins/Del polymorphism of the DRD2 gene was shown to be significantly associated with sexual dysfunction. Although this may be mediated by prolactin, other factors potentially lead to greater sexual dysfunction in patients receiving typical antipsychotics. 33 In addition, studies have shown that the COMT Met allele leads to higher levels of synaptic dopamine and catechol estrogen compared with the Val allele. The increase in synaptic dopamine and catechol estrogen raises blood pressure.34,35 Hypertension is an independent risk factor for erectile dysfunction. 36 In this study, the level of serum dopamine and the expression of DRD2 and SLC6A3 protein and mRNA in the penile tissue of depression model rats were significantly lower than those in the normal group, whereas the expression of COMT protein was increased. The results of this study are consistent with previously reported results.24–27 Therefore, dysfunction of the dopaminergic system is an important cause of ED in depression model rats. Model rats exhibited decreased SLC6A3 and DRD2 protein expression; reduced dopamine secretion, transport, and receptor binding; increased COMT levels; and elevated dopamine enzymatic hydrolysis. Together, these events led to ED in depression model rats.
According to the literature, this is the first study on the mechanism of ED induced by depression in male rats. The lack of histological differences in the penile tissue between model and control group rats might be because depression directly affects erectile function through the central nervous system and does not substantially affect the penile tissue structure. The combination of bioinformatics analysis and animal experimental research makes the results of this study more robust. The potential mechanism of ED in depression model rats was uncovered, providing a foundation for future experimental and clinical research.
The limitation of this study is that serum levels of sex hormones were not determined. Although sex hormones, especially testosterone, play an important role in erectile function, this study compared the expression of related proteins based on bioinformatics analysis. Sex hormone-related indicators could not be obtained through bioinformatics prediction. The pathogenesis of psychological ED is complex, and the effect of dopamine on erectile function is only one process. Next, we aim to conduct further in-depth research to explore the detailed mechanism of depression-induced decreases in erectile function, providing a reference for clinical treatment. Moreover, this study did not conduct intercourse tests for the determination of erectile function, which is also a limitation. We will address this in future research.
Conclusion
The decrease in erectile function in the depression rat model is related to the dysfunction of the dopaminergic system and the dopaminergic synapse signaling pathway. In depression model rats, the expression levels of dopamine, DRD2, and SLC6A3 decreased, whereas the levels of COMT protein and mRNA increased, resulting in reduced penile erectile function.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shenzhen Key Specialty Construction Project of Traditional Chinese Medicine (grant number: 2019-2023(14)) and Science and Technology Innovation Project of the Chinese Academy of Traditional Chinese Medicine (grant number: C12021A02208).
Author contributions
JW was responsible for the conception and design of the study. SW and JQ were responsible for the acquisition of data. YZ and QW performed the data analysis. ZH, ZC, and JF drafted the manuscript. JW revised and commented on the draft. All authors participated in the interpretation of the findings, and all authors read and approved the final version of the manuscript.
