Abstract
Takotsubo cardiomyopathy (TC) is a rare disease with unclear etiology that is characterized by wall motion abnormalities of the left ventricle. We report a 64-year-old woman who presented with cardiac arrest 6 hours after ureteral stenting, with no history of heart disease. Notably, she had a urinary tract infection preoperatively. TC was diagnosed with characteristic apical ballooning on the left ventriculogram. The hemodynamics and cardiac function recovered quickly within 1 day after conservative treatment and controlling the infection. TC should be considered when a patient presents with decreased cardiac function after ureteral stenting, especially in patients with potential concurrent infection. A review of the literature documenting cases of TC related to urological surgery in the past decade was conducted using PubMed. The results were summarized in a table.
Keywords
Introduction
Takotsubo cardiomyopathy (TC) is an acute reversible heart failure syndrome typically triggered by stressful events. TC is similar to acute coronary syndrome (ACS); however, there is no angiographic evidence of obstructive coronary artery disease (CAD) or acute plaque rupture in TC. 1 Although TC is considered to have a benign course, arrhythmias, heart failure, and cardiac arrest (CA) are severe complications. TC has been described with various surgeries; however, to our knowledge, no cases associated with ureteral stenting have been reported. Furthermore, cases of TC leading to CA are rare. Here, we report a rare case of postoperative CA due to TC after ureteral stenting, and we performed a review of TC cases related to urological procedures. The reporting of this case conforms to the CARE guidelines. 2
Case presentation
A 64-year-old woman (weight: 53 kg, height: 151 cm) with right lumbar pain without an obvious cause, accompanied by hematuria, nausea, vomiting, and an elevated body temperature of 39.0°C, was admitted to the authors’ hospital. She was scheduled for emergency operation (ureteral stenting) under general anesthesia.
Upon admission, the results of preoperative evaluations comprising pulmonary and airway examinations were unremarkable. Electrocardiography (ECG) showed a prolonged QT interval. There were noticeable abnormalities for some laboratory values, such as the white blood cell count: 19.57 × 109/L, neutrophils: 18.5 × 109/L, lymphocytes: 0.61 × 109/L, and C-reactive protein: 110.67 mg/L. Her medical history indicated hypertension for 16 years and diabetes for 6 years, which were poorly controlled with drug therapy. She had no family history of cardiopulmonary disease, and her American Society of Anesthesiologist (ASA) score was IIE. In the operating room, uneventful rapid-sequence induction of general anesthesia was performed, and anesthesia was maintained with propofol and remifentanil. The patient’s vital signs were stable, with blood pressure maintained at 120–140/68–84 mmHg and with a heart rate of 67–85 beats/minute, intraoperatively. The operation proceeded smoothly, and the tracheal tube was removed after the patient was fully awake. She was then transferred to the ward with a heart rate of 79 beats/minute, blood pressure of 155/75 mmHg, and blood oxygen saturation (SpO2) of 100% (oxygen flow of 2 L/minute through a nasal cannula; fraction of inspired oxygen (FiO2) = 28%).
Six hours after being transferred to the ward, the patient developed chest tightness and profound bradycardia followed by asystole requiring cardiopulmonary resuscitation (CPR), comprising chest compressions and injection of 1 mg epinephrine intravenously (IV). An anesthetist was urgently requested to perform endotracheal intubation. After active CPR, the patient's heart rate increased to 130 beats/minute, a peripheral arterial pulse was palpable, and SpO2 was 92%. Blood gas analysis showed the following: pH: 7.00, partial pressure of arterial oxygen (PaO2): 11.32 mmHg, partial pressure of arterial carbon dioxide (PaCO2): 116.32 mmHg, K+: 4.09 mmol/L, Ca2+: 1.02 mmol/L, lactate (Lac): 7.38 mmol/L, base excess (BE): −5.96 mmol/L, glucose (Glu): 7.57 mmol/L. The ECG was notable for ST-segment elevations in leads V1, V2, and V3, which indicated a possible acute myocardial infarction pattern. Transthoracic echocardiography showed a severely reduced left ventricular ejection fraction (LVEF) of 26%, akinesia of the left ventricular anterior wall to the apex, moderate tricuspid regurgitation, and pulmonary hypertension. Laboratory examinations showed noticeable abnormalities in troponin-T and creatine kinase-MB fraction (CK-MB) values (Table 1). Emergency percutaneous coronary intervention (PCI) was performed 8 hours postoperatively, which demonstrated normal coronary anatomy without obstructive lesions. However, the left ventriculogram showed apical ballooning, which was consistent with TC (Figure 1) (Video 1). Based on these findings, TC was diagnosed.
Troponin-T and CK-MB values.

CK-MB, creatine kinase-MB fraction.

Coronary angiography (a) showing no evidence of coronary artery disease. The left ventriculogram (b and c) shows the characteristic apical ballooning and hypercontraction of the basal segments.
The patient was then transferred to the cardiac care unit (CCU) while mechanically ventilated. Norepinephrine and levosimendan were administered IV for cardiovascular support with infusion rates of 0.08–0.2 μg/kg/minute and 0.1 μg/kg/minute, respectively. The patient also received the following treatments: aspirin 100 mg daily, atorvastatin 20 mg daily, spironolactone 20 mg daily, and rabeprazole 20 mg daily via nasogastric tube, with 4,000 IU of subcutaneous low-molecular-weight heparin every 12 hours and one dose of IV furosemide 10 mg. Fortunately, the patient's clinical and hemodynamic conditions gradually improved; therefore, norepinephrine, levosimendan, and low-molecular-weight heparin were discontinued. Chest radiography showed less exudation in both lungs than that before treatment. Repeat transthoracic echocardiography revealed that the anterior wall of the left ventricle and the apex of the heart showed decreased motion. On the 4th day postoperatively, echocardiography revealed normal heart function with an LVEF of 61% (Figure 2), and the patient was transferred to a general ward. Myocardial perfusion imaging showed no obvious abnormalities (LVEF: approximately 75%) (Figure 3) (Video 2A and Video 2B). On the 10th day postoperatively, the patient was discharged home with aspirin 100 mg once daily, atorvastatin 20 mg once daily, and bisoprolol fumarate 2.5 mg once daily. At the 1-week follow-up, she had fully recovered with continued medications. One month after discharge, she was hospitalized again to treat a ureteral calculus, and she underwent holmium laser lithotripsy without apparent complications.
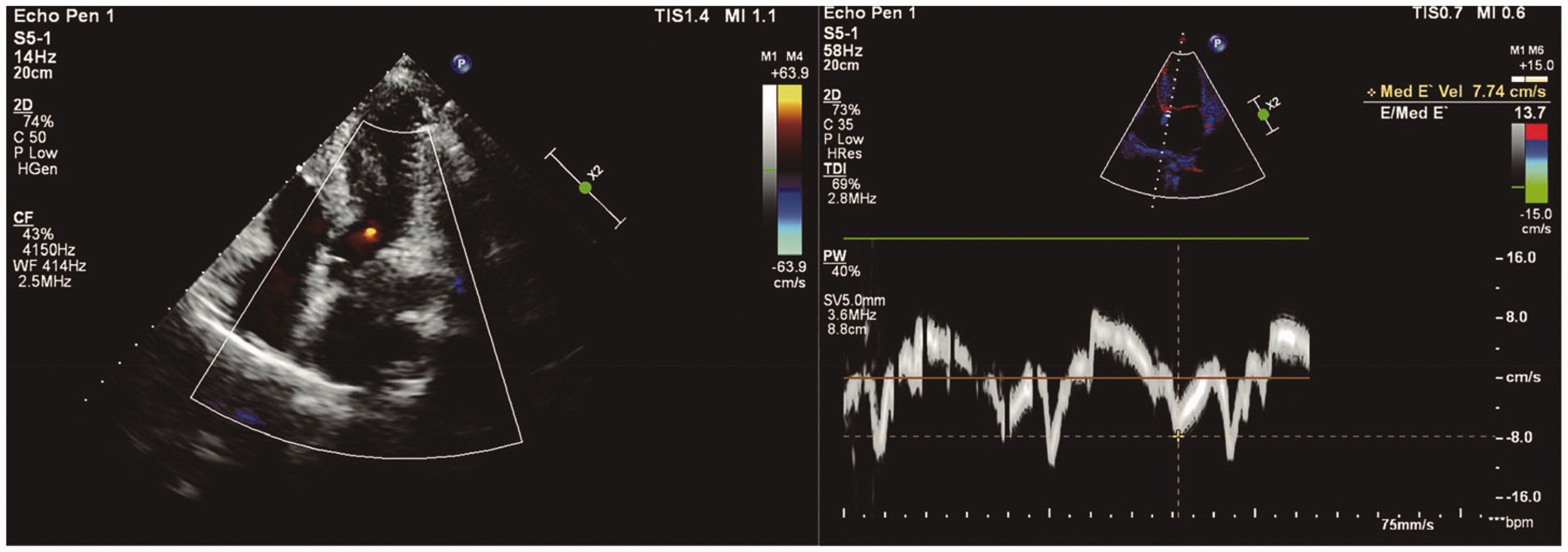
Transthoracic echocardiography after resolution of TC. No obvious abnormality of wall motion is visible. Aortic valve regurgitation was minimal.

Myocardial perfusion imaging showed no apparent myocardial damage.
Discussion
TC, also known as stress cardiomyopathy, apical ballooning syndrome, octopus pot cardiomyopathy, and broken heart syndrome, is characterized by wall motion abnormalities of the left ventricle. According to the abnormal distribution of left ventricular wall motion, there are four main types of TC: apical, midventricular, basal, and focal. 1 TC is estimated to constitute 2% of patients with ACS; 3 therefore, TC should be considered in patients suspected of having ACS. Although multiple diagnostic criteria have been proposed for TC, 4 the 2008 revised Mayo Clinic criteria 5 are still the most widely referenced, and are as follows: (1) transient LV midsegment hypokinesis, akinesis, or dyskinesis that extends beyond a single epicardial vascular distribution (a stressful trigger and apical involvement may or may not be present); (2) absence of obstructive CAD or angiographic evidence of acute plaque rupture; (3) new electrocardiographic changes (i.e., ST-segment elevation and/or T-wave inversion or an elevated troponin level); and (4) exclusion of pheochromocytoma and/or myocarditis.
In our case, sepsis-induced cardiomyopathy was also a consideration owing to the presence of infection; however, several clinical features suggested TC, instead of ACS or sepsis-induced cardiomyopathy. First, the ECG showed ST-segment elevation in the precordial leads without the development of Q waves at symptom onset, indicating TC rather than ischemic myocardial injury. 3 Second, the increase in serum cardiac biomarkers was relatively insignificant compared with the concentrations seen with ACS. Third, the cardiac wall motion abnormality resolved completely only 1 day after TC, which is not common in ACS but is a usual evolution of TC. Fourth, the regional wall movement abnormality supported TC and was different from that seen with sepsis-induced cardiomyopathy, which is characterized by overall ventricular insufficiency and expansion. 6
The specific etiology responsible for TC has not been described definitively. The main possible mechanisms are catecholamine toxicity, microvascular dysfunction, inflammation, decreased estrogen levels, genetic predisposition, and others. 7 Additionally, direct toxicity of catecholamines on cardiomyocytes was suggested as a leading factor. 7
The incidence of TC has been increasing worldwide since its discovery in Japan in 1990, but its association with urologic surgery is poorly described in the literature.7,8 PubMed was searched for TC related to urologic surgery in the past decade, and only eight cases have been reported. The results are shown in Table 2. Among the eight cases, perioperative stress (six cases),9–14 local anesthetic and epinephrine infiltration (one case), 15 and pheochromocytoma (one case) 16 were possible triggers. Furthermore, TC occurred preoperatively (one case), 14 intraoperatively (four cases)9,13,15,16 and postoperatively (three cases).12–14 Previous patients also underwent different procedures, such as transurethral resection of a bladder tumor (TUR-BT), adrenalectomy, robotic radical cystectomy, and others. However, no case report mentioned CA resulting from TC after ureteral stenting.
Previous cases of Takotsubo cardiomyopathy after urologic surgery.

ETT, endotracheal tube; LMA, laryngeal mask airway; TUR-BT, transurethral resection of a bladder tumor; LBBB, left bundle branch block; Postop, postoperative; Intraop, intraoperative; Preop, preoperative.
In our patient, TC developed after ureteral stenting, with the initial presentation of chest tightness and profound bradycardia, followed by asystole. It is important to note the presence of infection perioperatively in our patient, although without a diagnosis of sepsis. Infection is a common cause of TC, accounting for 30.7% of cases with physical triggers. 17 Our patient’s hemodynamics stabilized, and cardiac function recovered quickly within 1 day after conservative treatment and controlling the infection. In our opinion, we should be alert to the possibility of TC after ureteral stenting, especially in patients with potential concurrent infection, even though ureteral stenting is a minimally invasive urologic surgery.
Particularly, in our patient, myocardial perfusion imaging was used to evaluate cardiac function, which is an approach that can quantitatively assess myocardial perfusion and obtain the LV functional parameters. A previous study showed that myocardial perfusion imaging can predict the prognosis of patients with coronary heart disease, 18 and this tool might also be helpful in patients with TC.
Currently, there is no established guideline for the treatment for TC. The main goal of treatment focuses on supportive treatment and maintaining hemodynamic stability. Owing to bradycardic arrest induced from reversible heart disease, our patient was not implanted with a pacemaker. 19 Although angiotensin converting enzyme inhibitors/angiotensin receptor inhibitors (ACEI/ARBs) have been shown to reduce recurrence and benefit prognosis in TC, whether β-blockers are beneficial to long-term outcomes remains controversial. 20 The calcium-sensitizing inodilator levosimendan may improve left ventricular systolic function and myocardial oxygen supply. 21 Alpha-2 agonists, such as dexmedetomidine and clonidine, may be beneficial to prevent recurrence in at-risk patients by reducing sympathetic outflow. 22
In conclusion, we describe the first known case of a postmenopausal female with postoperative CA after ureteral stenting due to TC. We emphasize the importance of maintaining a high suspicion of TC perioperatively, and we hope to increase awareness of the various risk factors for this syndrome.
Footnotes
Acknowledgement
The authors thank the patient in this report for providing permission to use her medical information.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Ethics statement
The study protocol was approved by Hebei General Hospital Ethics Committee (approval number: 2022029). We obtained written consent from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editor of this journal.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Key Research and Development Program of Hebei Province (Grant No. 19277714D).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
