Abstract
Abstract
Objective
To assess the relationship between chronic obstructive pulmonary disease (COPD) severity and bone mineral density (BMD) in the whole body and different body areas.
Methods
This retrospective, cross-sectional study included patients with COPD. Demographic and lung function data, COPD severity scales, BMD, and T scores were collected. Patients were grouped by high (≥–1) and low (<–1) T scores, and stratified by body mass index, airway obstruction, dyspnoea, and exercise capacity (BODE) index. The relationship between whole-body BMD and BODE was evaluated by Kendall’s tau-b correlation coefficient. Risk factors associated with COPD severity were identified by univariate analyses. BMD as an independent predictor of severe COPD (BODE ≥5) was verified by multivariate logistic regression. BMD values in different body areas for predicting severe COPD were assessed by receiver operating characteristic curves.
Results
Of 88 patients with COPD, lung-function indicators and COPD severity were significantly different between those with high and low T scores. Whole-body BMD was inversely related to COPD severity scales, including BODE. Multivariate logistic regression revealed that BMD was independently associated with COPD severity. The area under the curve for pelvic BMD in predicting severe COPD was 0.728.
Conclusion
BMD may be a novel marker in predicting COPD severity, and pelvic BMD may have the strongest relative predictive power.
Introduction
Chronic obstructive pulmonary disease (COPD) is a serious health burden and a major cause of mortality worldwide. 1 In a study of 59 906 US veterans with COPD, the mean respiratory-related total healthcare costs were approximately 10 times higher in patients than in controls without COPD. 2 A systemic review in China showed that COPD prevalence varied between 1.20% and 8.87% in different provinces/cities across China, and COPD ranked among the top three leading causes of death. 3 COPD is a condition characterized by progressive and not fully reversible airflow limitation, 4 and is thought to affect around 10% of the adult population aged 40 years and older. 5
Patients with COPD have a high risk of osteopenia and osteoporosis.6,7 According to a Japanese database, bone mineral density (BMD) may be expressed as an absolute value, T scores (standard deviations from a young, sex-specific reference mean BMD), and Z-scores (standard deviations from age- and sex-specific reference mean BMD). 8 Osteopenia (T score between –1.0 and –2.5) and osteoporosis (T score equal to or below –2.5) are conditions characterized by decreased bone mass and density, leading to bone fragility. 9 The percentage of patients with COPD and osteoporosis varies widely, ranging from 18%–65%, while the percentage of patients with COPD and osteopenia ranges from 14%–66%. 10 As a pulmonary disease with many systemic manifestations, one of the most important factors related to inflammation is tobacco smoke, which directly induces systemic damage, for instance, by lowering the pH of bone tissue, resulting in absorption of bone salts. 11 Cigarette smoking is the most commonly encouraged, yet readily identifiable risk factor for COPD, which presents in almost 15% of smokers. 12 Long-term cigarette smoke exposure is shown to deteriorate bone quality of the lumbar vertebrae, as illustrated by disorientation of collagen fibres and the biological apatite c-axis. 13 Smoking causes exposure to cadmium, which is a known risk factor for osteoporosis. 14 In addition, alterations in bone metabolism may occur as indirect or direct effects of nicotine, or other constituents of cigarette smoke, on osteoblastic bone formation and osteoclastic bone resorption. 15 Indirect systemic effects, through the induction of lung inflammation and the production of systemic inflammatory mediators, have an impact on bones and other organs. 16 Furthermore, patients with COPD have difficulty breathing during fatigue, which may lead to inactivity and have a negative impact on BMD. 17
Although the prevalence of osteoporosis in COPD is very high, its standardized treatment is often ignored, and the effects of BMD in different body areas on severity of COPD have not been fully described. The present study aim was to provide a better understanding of osteoporosis in patients with COPD, to enable more precise body part protection and treatment for this patient group, by examining the associations between BMD measured in the whole body and in different parts of the body, and COPD severity.
Patients and methods
Study population
This retrospective cross-sectional study included patients who were diagnosed with COPD and treated at the Third Affiliated Hospital of Wenzhou Medical University, Wenzhou, China, between February 2018 and February 2019. The study inclusion criteria were: (1) age >40 years; and (2) diagnosis of COPD as defined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines. 1 Exclusion criteria comprised: (1) history of cardiac surgery; and (2) history of malignant tumours during the previous 5 years. 18 Patients were divided into two groups according to T score cut-off point of –1.0: high BMD group (T score ≥ –1.0) and low BMD group (T score < –1.0 [encompassing osteopenia/osteoporosis classifications]). 9 The study was approved by the Institutional Ethics Committee review board of the Third Affiliated Hospital of Wenzhou Medical University (approval No. YJ20170015; 24 November 2017) and was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from study participants or their legal proxies and patient details were de-identified for the study. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 19
Data collection
Data regarding age, sex, body mass index (BMI), current smoking status, current drinking status, duration of disease, comorbidities, and current treatment were collected by self-completed questionnaires provided to patients during a hospital visit. Forced expiratory volume in 1 second (FEV1), forced expiratory volume in 1 second in percent of the predicted value (FEV1%), forced vital capacity (FVC) and FEV1/FVC of patients with COPD were recorded within 7 days of hospital admission as significant indicators of pulmonary function. Data were collected regarding BMD in different body areas, including whole-body BMD, and BMD of the head, upper limb, thigh, trunk, ribs, pelvis, and spine that were determined by dual-energy X-ray absorptiometry using a GE Lunar Prodigy DXA system (General Electric Company, Boston, MA, USA). 20 T scores, based on whole-body BMD, were also obtained. Data for grading the severity of COPD in patients were also gathered within 7 days of hospital admission, including GOLD stage, BMI, airway obstruction, dyspnoea, and exercise capacity (BODE) index; 6-min walk test (6MWT) score; modified Medical Research Council (mMRC) dyspnoea scale; age, dyspnoea and airflow obstruction (ADO) score; and COPD assessment test (CAT) score. BODE stages 1, 2, 3 and 4 were defined as BODE score of 0–2, 3–4, 5–6, and 7–10 points, respectively. 21 Higher COPD severity was defined as BODE ≥5 points. 22
COPD diagnostic criteria
Patients with COPD were categorized into four COPD stages according to GOLD spirometry criteria: GOLD stage 1, FEV1 ≥80% predicted (mild); GOLD stage 2, 50% ≤ FEV1 < 80% predicted (moderate); GOLD stage 3, 30% ≤ FEV1 < 50% predicted (severe); and GOLD stage 4, FEV1 < 30% predicted (very severe). 23
Statistical analyses
Statistical analyses were performed using SPSS software, version 26.0 (IBM, Armonk, NY, USA). Continuous variables with normal distribution are presented as mean ± SD, and variables with skewed distribution are presented as median (interquartile range). Categorical variables are presented as n (%) prevalence. Differences in continuous clinical characteristics between patients with high (T ≥ –1) and low (T < –1) T scores were analysed using Student’s independent samples t-test (for normally distributed data) or Mann–Whitney U-test (for skewed data). Categorical data were compared using χ2-test or Fisher’s exact test. The relationship between whole-body BMD and various parameters, including patient demographics, lung function and COPD severity was evaluated by Pearson’s correlation coefficient, Spearman’s rank correlation coefficient or Kendall’s tau-b correlation coefficient. Univariate analyses were performed to identify risk factors associated with COPD severity. Multivariate logistic regression analyses were performed to identify the contribution of BMD as an independent predictor of severe COPD (BODE ≥5 points). The value of BMD in different body areas for predicting COPD severity was estimated using receiver operating characteristic (ROC) curves according to low (<5) or high (≥5) BODE score. Two-sided P values <0.05 were considered to be statistically significant in all analyses.
Results
Study population characteristics
A total of 88 patients with COPD were included in the study: high BMD group (T score ≥ –1.0), n = 58 (66%); and low BMD group (T score <–1.0 [encompassing osteopenia/osteoporosis classifications]), n = 30 (34%). Statistically significant between-group differences were observed regarding sex, smoking and drinking status, FEV1, FVC, BODE score, 6MWT score, mMRC dyspnoea scale, ADO score and CAT score (P < 0.05; Table 1). Furthermore, BMD in all body areas were significantly different between the two groups (P < 0.05; Table 1). FEV1 and FVC were higher in the high T-scores group than in the low T-scores group. Additionally, BODE, mMRC dyspnoea scale ADO and CAT scores were significantly higher (P < 0.05; Table 1), and 6MWT score was significantly lower (P = 0.011) in the low T-scores group versus the high T-scores group. These results showed that BMD is related to respiratory function and COPD severity, where the lower the BMD (represented by T scores), the worse the patient's respiratory function and the higher the severity of COPD. Rates of type 1 diabetes mellitus and postmenopausal osteoporosis were significantly higher in the low-T score group than in the high T-scores group (P = 0.020 and P = 0.004, respectively), but there were no statistically significant between-group differences in rates of other comorbidities (Table 1).
Comparison of demographic and clinical characteristics, disease severity, lung function, BMD and other functions between patients with COPD, categorised according to low (T < –1) or high (T ≥ –1) T scores.
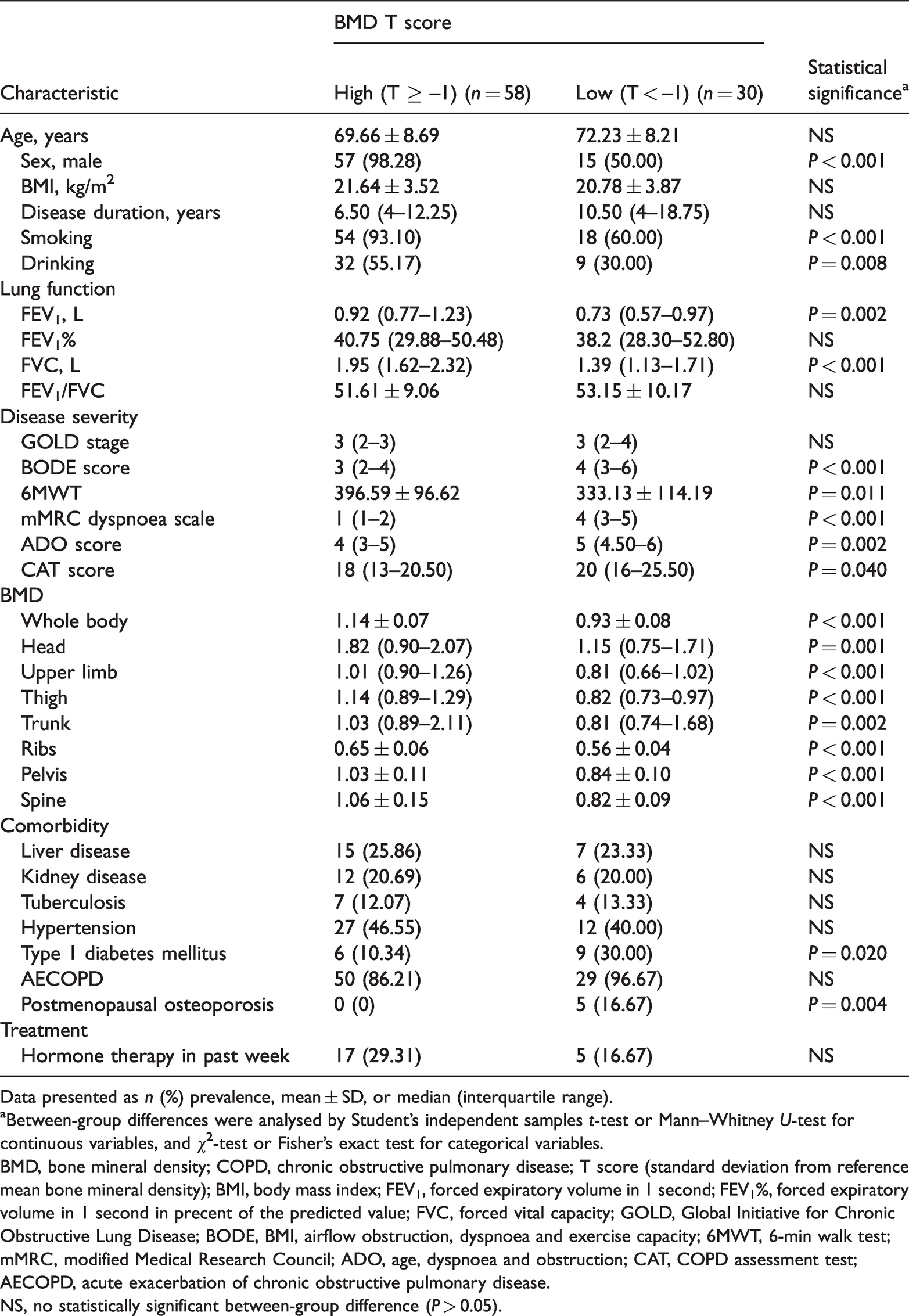
Data presented as n (%) prevalence, mean ± SD, or median (interquartile range).
aBetween-group differences were analysed by Student’s independent samples t-test or Mann–Whitney U-test for continuous variables, and χ2-test or Fisher’s exact test for categorical variables.
BMD, bone mineral density; COPD, chronic obstructive pulmonary disease; T score (standard deviation from reference mean bone mineral density); BMI, body mass index; FEV1, forced expiratory volume in 1 second; FEV1%, forced expiratory volume in 1 second in precent of the predicted value; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; BODE, BMI, airflow obstruction, dyspnoea and exercise capacity; 6MWT, 6-min walk test; mMRC, modified Medical Research Council; ADO, age, dyspnoea and obstruction; CAT, COPD assessment test; AECOPD, acute exacerbation of chronic obstructive pulmonary disease.
NS, no statistically significant between-group difference (P > 0.05).
Correlations between BMD and lung function or COPD severity
Analyses of the correlation between whole-body BMD and various characteristics, including demographics, lung function and COPD severity scales, showed that BMD was significantly inversely related to disease severity scores, including BODE score (r = –0.271, P = 0.001), mMRC dyspnoea scale (r = –0.239, P = 0.004) and ADO score (r = –0.222, P = 0.006), and was positively related to markers of lung function, such as FEV1 (r = 0.376, P < 0.001) and FVC (r = 0.461, P < 0.001). These results indicate that BMD may be a good biomarker of COPD in terms of disease severity and pulmonary function. Whole-body BMD was also significantly related to BMI (r = –0.273, P = 0.010) and smoking (r = 0.370, P < 0.001) (Table 2).
Correlation between BMD and various parameters, including patient demographics, lung function and COPD severity indices in 88 patients with COPD.

aThe relationship between whole-body BMD and various parameters was assessed using Pearson’s correlation coefficient, Spearman’s rank correlation coefficient or Kendall’s tau-b correlation coefficient.
BMD, bone mineral density; COPD, chronic obstructive pulmonary disease; BMI, body mass index; FEV1, forced expiratory volume in 1 second; FEV1%, forced expiratory volume in 1 second as precent of the predicted value; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; BODE, BMI, airflow obstruction, dyspnoea and exercise capacity; 6MWT, 6-min walk test; mMRC, modified Medical Research Council; ADO, age, dyspnoea and obstruction; CAT, COPD assessment test.
NS, no statistically significant correlation (P > 0.05).
To determine the relationship between various factors and severity of COPD, patients were stratified into two groups according to BODE score (BODE < 5 points or BODE ≥5 points) and associated variables were subjected to univariate logistic regression. BMI (P = 0.003), disease duration (P = 0.030), drinking status (P = 0.008), FEV1 (P < 0.001), FEV1% (P < 0.001), FVC (P < 0.001), FEV1/FVC (P < 0.001), and BMD, including BMD of the whole body (P = 0.022), thigh (P = 0.038), ribs (P = 0.005), pelvis (P = 0.003) and spine (P = 0.034), were found to be significantly correlated to COPD severity (Table 3).
Univariate logistic regression analyses to assesses the association between different factors and COPD in 88 patients with COPD stratified according to BODE score (BODE < 5 points or BODE ≥5 points).

COPD, chronic obstructive pulmonary disease; BODE, BMI, airflow obstruction, dyspnoea and exercise capacity; BMI, body mass index; FEV1, forced expiratory volume in 1 second; FEV1%, forced expiratory volume in 1 second as percent of the predicted value; FVC, forced vital capacity; BMD, bone mineral density; OR, odds ratio; CI, confidence interval.
NS, no statistically significant correlation (P > 0.05).
To further assess the relationship between whole-body BMD and COPD severity (by BODE score), multivariate logistic regression analyses were performed, controlling for potential confounding variables (Table 4). In Model 1, nothing was adjusted (odds ratio [OR] 0.006, 95% confidence interval [CI] 0.000, 0.484; P = 0.022). After adjusting for age and smoking status in Model 2, the link between BMD and BODE remained significant (OR 0.003, 95% CI 0.000, 0.407; P = 0.020). On the basis of Model 2, adjustments were made for sex in Model 3, and the link between BMD and BODE remained significant (OR 0.005, 95% CI 0.000, 0.984; P = 0.049). To control for the effects of comorbidities, Model 4 adjusted for liver and kidney disease, and postmenopausal osteoporosis in addition to age, sex and smoking status, and the relationship remained statistically significant (OR 0.001, 95% CI 0.000, 0.407; P = 0.023). These results suggest that BMD may be an independent risk factor of COPD severity. Similar results were shown in patients stratified according to T scores and BODE score (BODE 0–2, BODE 3–4, BODE 5–6, and BODE 7–10), in which patients with low T scores (< –1) had a higher proportion of BODE score ≥5 (BODE 5–6 and BODE 7–10) indicating higher disease severity (44.82% versus 19.3%; P < 0.001; Figure 1).
Adjusted odds ratios and 95% confidence intervals for the association between whole-body BMD and COPD severity (by BODE score).

BMD, bone mineral density; COPD, chronic obstructive pulmonary disease; BODE, body mass index, airflow obstruction, dyspnoea and exercise capacity.
Model 1, unadjusted univariate analysis; Model 2, adjusted by age and smoking status; Model 3, adjusted by age, sex, and smoking status; Model 4, adjusted by age, sex, smoking status, liver disease, kidney disease, and postmenopausal osteoporosis.
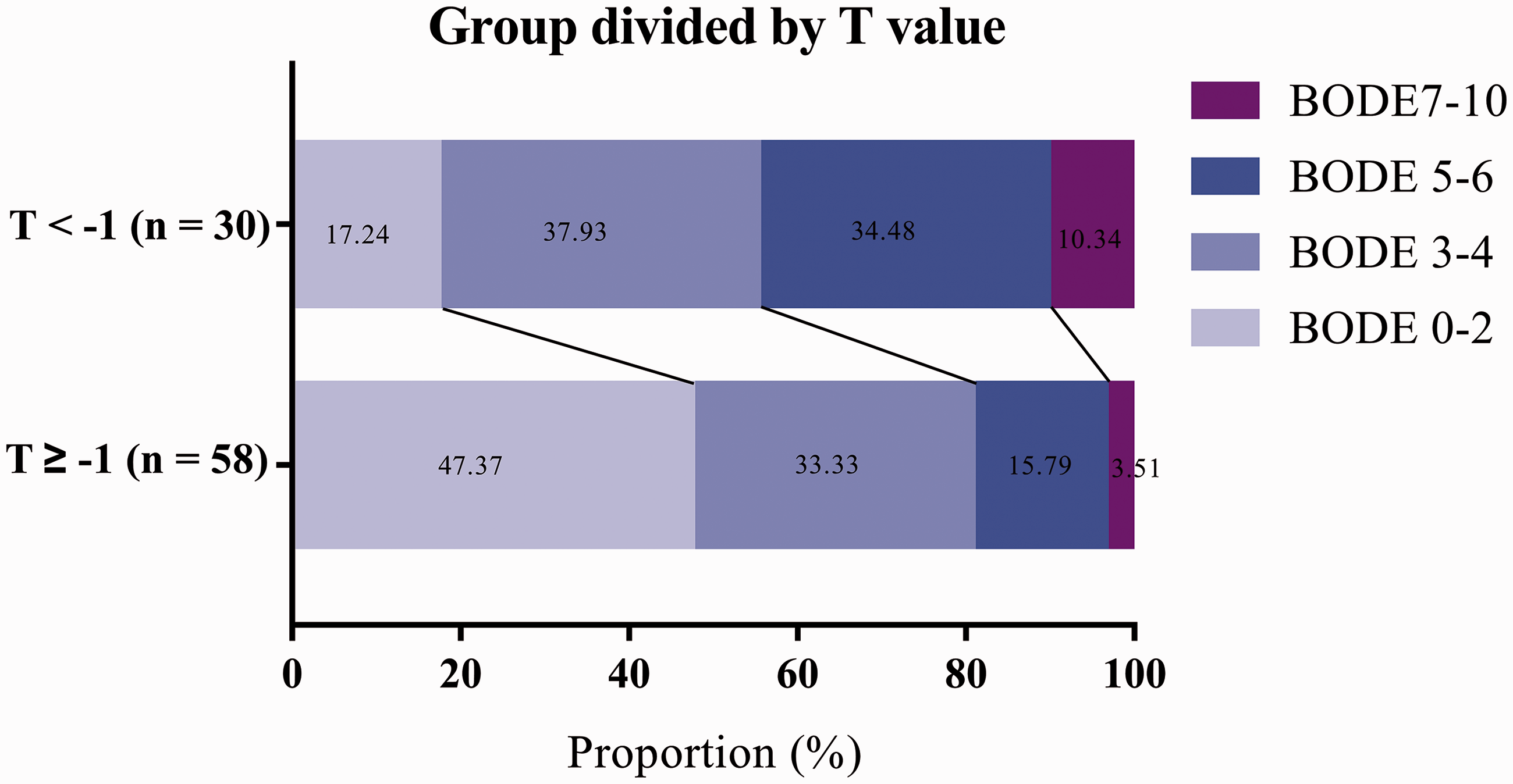
Composite bar chart showing rates of different BODE score grades in 88 patients with COPD grouped according to low (<–1) or high (≥–1) T scores. BODE, body mass index, airway obstruction, dyspnoea, and exercise capacity; COPD, chronic obstructive pulmonary disease; T score, standard deviation from reference mean bone mineral density.
Comparison of BMD in different body areas in predicting COPD severity
The utilities of BMD in different body areas for predicting COPD severity were analysed by ROC curves (Figure 2). The area under the curve (AUC) value for whole-body BMD was 0.670 (95% CI 0.539, 0.800; P = 0.015). Among all the body areas, pelvic BMD displayed the largest AUC (0.728, 95% CI 0.610, 0.846; P = 0.001), and at a cut-off value of 0.425, the specificity and sensitivity for predicting COPD severity were 87.5% and 55.0%, respectively. This result suggests that BMD may be a significant marker for predicting COPD severity, with pelvic BMD being the strongest predictor.

Receiver operating characteristic curves of bone mineral density (BMD) in different body areas for predicting severe chronic obstructive pulmonary disease (BODE score ≥5 points). AUC, area under the curve; BODE, body mass index, airflow obstruction, dyspnoea and exercise capacity.
Discussion
The results of present study indicated that decreased respiratory function and increased disease severity were associated with decreased BMD, which could be observed according to patient’s T scores. In addition, the relationship between BMD and COPD severity differed depending on where in the body BMD was measured, with pelvic BMD displaying the strongest predictive power.
In the present study population with COPD, there was a high prevalence of low BMD (34% of patients with T score < –1), which concurs with previous studies that have reported a relationship between BMD and lung function in patients with COPD.9,24,25 Lekamwasam et al.26,27 showed that FEV1 was positively and independently associated with BMD in 4830 females aged 45–76 years, and with hip BMD in 947 males aged 65–76 years, from the general population of the United Kingdom. Another study reported that 68% of 62 patients with COPD and a mean FEV1 of 33% predicted, had osteopenia or osteoporosis, assessed by DEXA or spinal X-ray. 6 In addition, bone density has been reported to differ significantly among patients with different stages of COPD, even after adjusting for BMI and corticosteroid usage, 28 and compared to patients with stage 2 disease, patients with COPD stage 4 were found to have a 7.6 times greater risk of having low BMD, as measured by quantitative ultrasound. 29 The present research supports the understanding that low BMD is associated with both reduced lung function and increased disease severity. Despite having a recognized role in morbidity and mortality associated with COPD, osteoporosis remains a neglected condition during the follow-up of patients affected with this pulmonary disease, and there is a lack of attention to BMD in patients with COPD. 30 Osteoporosis and osteopenia are often neglected in patients with COPD and cannot be effectively treated, which may result in more severe COPD and increased mortality. Therefore, the present authors believe that measuring BMD as a risk predictor of COPD is valuable, and accurate prevention can be carried out according to the degree of BMD in different parts of the body. For example, in the present study, pelvis BMD was found to be closely related to COPD severity, therefore, in the later treatment plan, more attention can be paid to the pelvis to prevent the possibility of fracture caused by osteoporosis, potentially leading to a better quality of life for patients with COPD.
There may be several explanatory mechanisms for BMD as a risk indicator of COPD and the particular association of pelvis BMD. The first is an increased level of systemic inflammation, as reduced lung function is found to be closely related to increased levels of systemic inflammatory markers, 31 which present as significantly elevated levels of C-reactive protein (CRP), fibrinogen, leucocytes, and tumour necrosis factor-α. 32 In a study of patients with type 1 diabetes, CRP was inversely associated with BMD of the whole body, the pelvis and leg, 33 which may be one of the reasons why patients grouped by T score in the present study differed in terms of diabetes prevalence. The second mechanism may be a low level of physical activity, because patients with COPD have difficulty breathing during fatigue, which may lead to lack of activity and a negative impact on BMD. Physical disuse is not only shown to be related to osteoporosis in the upper limb, but also in the hip after leg injury.34,35 Furthermore, in a spaceflight study into the effects of disuse on the body, BMD changes were statistically significant, with losses averaging 1% per month in the pelvis. 36 The third mechanism may be corticosteroid-induced loss of bone mass. For example, a meta-analysis that included 16 randomized controlled trials with 17 513 subjects and seven observational studies with 69 000 subjects indicated that fracture risk appeared dose-dependent. 37 However, in the present study, there was no significant difference between the two groups of patients with different T values in whether they had used corticosteroid hormones in the previous week, which may have been due to a lack of long-term hormone use history by the corresponding patients, and the fact that corticosteroid hormones affect bone density over time. Another potential mechanism may be hypercapnia, which is supported by previously published research showing that lower arterial pH and higher arterial carbon dioxide levels correlate with lower BMD in patients with COPD. 38 High consumption of carbonated beverages have also been shown to exert a moderate effect on BMD. 39 Finally, hormone levels may be another mechanism, as Carlson et al. 40 found that in both pre- and postmenopausal women, hormone replacement therapy and increased circulating oestrogen levels had a protective effect on pulmonary function. In the present study, there was also a significant difference in BMD among postmenopausal females with osteoporosis, however the results may have been biased by the small sample number, and needs to be tested in a larger population.
Interestingly, previous studies have shown that low BMD is closely related to chronic liver disease, chronic kidney disease, tuberculosis and acute exacerbation of chronic obstructive pulmonary disease,41–44 which is inconsistent with the present results. The small number of subgroups in the present study may be one reason for the above contradiction, however, other studies have also shown that chronic kidney disease is not associated with low BMD at the spine, suggesting that BMD in different parts of the body may possess different associations with chronic disease. 45 In addition, no significant difference in BMD has been shown between patients with alcoholic liver disease and controls. 46
The present results may be limited by several factors. Firstly, a major limitation is the small sample size, and the present findings require verification by investigating a larger database of patients. Secondly, due to the lack of BMD data in healthy people, patients with COPD could not be compared with a healthy control group. Finally, the study lacks the support of follow-up data, due to difficulties in obtaining BMD values during patient follow-up. However, data regarding significant areas of BMD, such as BMD of the whole body, head, upper limb, thigh, trunk, ribs, pelvis and spine, were included to provide comprehensive data to support the present study.
Conclusion
To conclude, BMD may be a novel marker of COPD. Lower levels of BMD may be independently associated with higher levels of disease severity in patients with COPD, while BMD of the pelvis may possess the strongest predictive power.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221094644 - Supplemental material for Associations between bone mineral density and chronic obstructive pulmonary disease
Supplemental material, sj-pdf-1-imr-10.1177_03000605221094644 for Associations between bone mineral density and chronic obstructive pulmonary disease by Xiaodiao Zhang, Keke Ding, Xiaqi Miao, Jianing Wang, Binbin Hu, Jiamin Shen, Xueting Hu, Yage Xu, Beibei Yu, Tingting Tu, Aiju Lin, Xianjing Chen and Yiben Huang in Journal of International Medical Research
Footnotes
Acknowledgements
The authors thank all participants in the study and the staff at the Third Affiliated Hospital of Wenzhou Medical University for their contribution in obtaining the data and assisting in the successful completion of the research.
Author contributions
XC and YH participated in the study conception and design; XZ, KD, XM, JW, BH, JS, XH, YX, BY, TT, and AL acquired the data; XZ, KD, XM, and JW analysed and interpreted the data; XZ, KD, and XM drafted the manuscript; XC, YH, XZ, KD, XM, JW, BH, JS, XH, YX, BY, TT, and AL critically revised the article for important intellectual content. All authors read and approved the final manuscript.
Data accessibility
Data included in the current study are not publicly available to ensure confidentiality of the patients, but are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Wenzhou Municipal Sci-Tech Bureau Program (Y20210842). The Funding body had no role in the study design, data collection, analysis, interpretation of data or writing of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
