Abstract
Objective
Diabetic macular edema (DME), the most common cause of diabetes-related visual impairment, may occur following pars plana vitrectomy (PPV) to manage proliferative diabetic retinopathy complications. This retrospective single-centre case series evaluated the efficacy and safety of injecting 4 mg/0.1 ml triamcinolone into the suprachoroidal space using a custom-made needle to treat DME post PPV.
Methods
Data regarding central macular thickness (CMT) using spectral domain-optical coherence tomography and best-corrected visual acuity (by Snellen chart) at baseline (pre-injection), and at 1, 4, and 8 weeks following injection were analysed, along with intraocular pressure (IOP), cataract progression, and ocular safety.
Results
Eleven eyes in 10 patients received 11 suprachoroidal injections. Vision improvement was noted (0.75 log minimum angle of resolution (MAR) at baseline to 0.40 log MAR after treatment). CMT reduced significantly from 456.45 ± 113.42 μm at baseline to 247.63 ± 53.40 μm at 8 weeks following injection. No rise in IOP, or cataract development in the single treated phakic eye, was observed during 8 weeks of follow-up.
Conclusion
Suprachoroidal injection of triamcinolone using a custom-made needle to treat DME post PPV shows promising results with acceptable safety outcomes. Large clinical trials with longer follow-up are needed to evaluate this treatment option for countries with limited health-care resources.
Introduction
Pars plana vitrectomy (PPV) is an essential procedure to manage proliferative diabetic retinopathy complications, 1 such as tractional retinal detachment and non-clearing vitreous haemorrhage and tractional diabetic maculopathy presenting with moderate reduction of visual acuity. PPV increases oxygenation and offers mechanical removal of vascular endothelial growth factor (VEGF) and inflammatory cytokines from the vitreous cavity, resulting in clearer media and releasing the tractional forces of retinal and macular tissues.2,3
Diabetic macular edema (DME) is the most common cause of diabetes-related visual impairment worldwide, and may occur post PPV. 3 Intravitreal anti-VEGF and steroids have been shown to effectively treat DME, and vitreous is an excellent medication reservoir that increases the half-life of drugs. 4 Therefore, intravitreal medications, including triamcinolone acetate or anti-VEGFs, will have a reduced half-life with increased rapid clearance in vitrectomized versus non-vitrectomized eyes. 5 Yanyali et al., reported no improvement in macular thickness or visual acuity in a short-term follow-up study of vitrectomized eyes treated with bevacizumab, 6 while Boyer et al., demonstrated effective reduction in DME with intravitreal dexamethasone implant in the vitrectomized eye. 7 However, dexamethasone implant cannot be reimbursed or is unavailable for patients in many low- and middle-income countries, such as Syria, due to high costs and lack of supply.
The suprachoroidal space is a potential reservoir that holds the unique position of being anatomically bound with choroid and sclera, which may translate to slower washout and better absorption of drug into the target tissue. Pharmacokinetic studies in animal models have shown a 12-fold increase in the concentration of drug in the retina and choroid, and up to 3% of the drug enters the anterior chamber, reducing the risk of intraocular pressure (IOP) spikes, cataract formation, and progression or exacerbation of pre-existing glaucoma. The systemic exit of corticosteroids is also demonstrated to be lowest with the suprachoroidal route compared with other local ocular injections. Porcine models have demonstrated a 10-fold reduction in the dosage required for a therapeutic effect similar to the intravitreal route.8–11
Human trials evaluating the efficacy and safety of the suprachoroidal drug delivery route have been conducted for various conditions, such as macular edema secondary to diabetes, non-infectious uveitis, and retinal vein occlusions. 12 A phase 3 trial of a suprachoroidal injection of proprietary preservative-free, terminally sterilized aqueous suspension of triamcinolone acetonide (Clearside Biomedical, Alpharetta, GA, USA) delivered with a customized system (Xipere; Clearside Biomedical) has been shown to have a high safety index in cases of non-infectious uveitis, with the incidence of IOP spike being 11.5% in patients. 13
Thus, the suprachoroidal route holds great promise in providing a therapeutic effect with a better theoretical safety profile than posterior subtenon and intravitreal routes. The suprachoroidal drug delivery system is not commercially available, however, use of a customized needle with a pre-set depth may enable a drug to be deposited into the suprachoroidal space.
The aim of the present study was to retrospectively analyse the safety and efficacy of 4 mg/0.1 ml triamcinolone acetonide injected into the suprachoroidal space using a custom-made needle, to manage DME in a series of vitrectomized eyes, in a setting with limited resources.
Patients and methods
Study population
This single-centre retrospective case series included data from patients with DME post PPV, who were treated with a suprachoroidal injection of triamcinolone at Arabi Hospital, Aleppo, Syria, between July 2020 and November 2020. Eligibility criteria for inclusion of patient records comprised DME with central macular thickness (CMT) >275 μm; best-corrected visual acuity (BCVA) better than 20/320; no history of glaucoma or IOP lowering medication; IOP <21 mm Hg; DME that presented ≥6 months following PPV; no treatment with injection or laser for at least 4 months prior to triamcinolone injection; and no coexisting recurrent haemorrhage.
Approval to undertake this retrospective interventional case series study was provided by the Ethics Review Board of Arabi Hospital. As this was a retrospective analyses of de-identified patient data, the requirement for informed consent from patients was waived by the ethics committee. The reporting of this study conforms to STROBE guidelines. 14
Treatment
All patients included in the study had received treatment for post-PPV DME with a 0.1 ml volume of 40 mg/ml triamcinolone acetonide (Cadila Pharmaceuticals, Ahmedabad, India), a non-preservative free suspension used for intravitreal injections, that was injected into the suprachoroidal space by a single physician (AM) using a custom-made needle (assembled by AM). The needle was devised from a 1-inch, short bevelled 30G needle with a sleeve obtained from a rubber plastic stopper. The 30G needle was passed through the stopper, which was fashioned in a manner that allowed only 1000 µm of the 30G needle to jut out from it. Vernier callipers were used to determine the length of the 30G needle extruding out of the stopper (Figure 1). 15
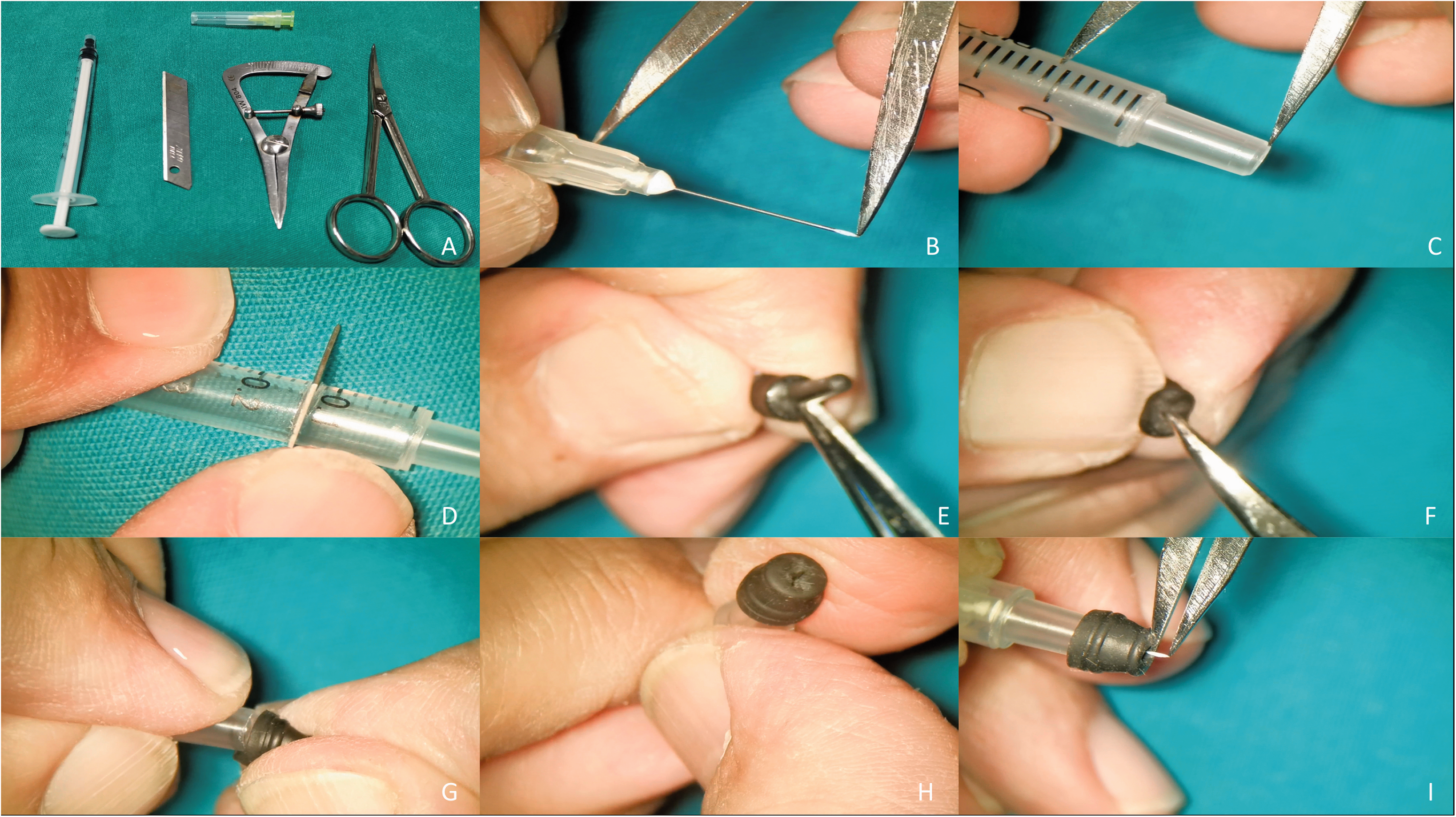
Representative images of the process of manufacturing the custom-made needle for suprachoroidal injection, showing: (a) instruments needed for manufacturing; (b) measurement of the needle length, including the section embedded in the plastic, using callipers; (c) measurement of the plastic stopper 2.5 mm less (1 mm for the needle to deliver into the suprachoroidal space and 1.5 mm for the thickness of the rubber stopper); (d) blade cutting the syringe according to the measurements; (e) removal of the rubber seal's ramification; (f) removal of the rubber seal from the plunger, and making a central cut in the rubber seal throughout the leading ring with scissors, essentially ensuring that the leading ring's surface is smooth and even; (g) installing the rubber stopper onto the prepared plastic Luer slip; (h) passing the 30-gauge needle through the previously made hole in the leading ring of the rubber seal, then fitting the plastic Luer slip onto the plastic part of the 30-gauge needle; and (i) checking that the exposed part of the 30-gauge needle is only 1 mm. 16
The needles were autoclaved before the injection. The periocular area was painted with 10% povidone-iodine while a drop of 4% povidone-iodine was instilled into the conjunctival cul-de-sac. Under local anaesthesia, after placing the drape and eye speculum, the injection site was marked 4 mm away from the limbus in the supero-temporal quadrant. Then, 0.1 ml of triamcinolone acetonide (40 mg/ml) was drawn into a tuberculin syringe with the custom-made needle. The bevel of the 30G needle was kept away from the limbus and was inserted into the sclera perpendicularly until the stopper touched the conjunctiva (Figure 2). 15 The drug was then injected into the suprachoroidal space and the needle was withdrawn obliquely to prevent reflux.

The injection technique, showing: (a) measuring 4 mm from the limbus; (b) the injection by placing the needle perpendicular to the ocular surface; and (c) withdrawing the needle obliquely from the eye. 16
The patient's pupil was dilated post-injection to facilitate peripheral retinal examination to check for any triamcinolone penetration in the intravitreal cavity, or suprachoroidal haemorrhage, or choroidal detachment.
Study outcomes
The primary outcomes were BCVA (by Snellen chart, presented as log minimum angle of resolution [MAR]); CMT, measured by spectral domain-optical coherence tomography (SD-OCT) using a Nidek RS-330 Retina scan duo (Gamagori, Aichi, Japan), and IOP recorded at the baseline (prior to suprachoroidal injection of triamcinolone) and at 1, 4 and 8 weeks post triamcinolone injection.
Statistical analyses
Data are presented as mean ± SD, median (range), or n (%) prevalence, and were statistically analysed using Microsoft Excel software, 2013 (www.Microsoft.com). Differences between time-points were analysed using Wilcoxon signed–rank test, and a P value <0.05 was considered statistically significant.
Results
A total of 11 eyes of 10 patients were eligible for study inclusion (mean age, 60.72 ± 7.74 years). Nine patients (90%) were female and one patient was male (10%), and all patients had DME post PPV for a median of 11.72 (±4.1) months before treatment with suprachoroidal triamcinolone injection. One eye was phakic (9.1%), and the other 10 (90.9%) were pseudophakic; four eyes (36.36%) were on the right of the face, and seven (63.63%) were on the left. Mean baseline glycosylated haemoglobin (HbA1c) for the 10 patients was 7.62 ± 0.39%, and mean baseline IOP was 14.54 ± 1.44 mmHg.
All cases had received anti-VEGF and panretinal photocoagulation (PRP) treatment during, or prior to, PPV. During PPV, three eyes had intraoperative internal limiting membrane peeling. Three eyes had no remnants of the epiretinal membrane (ERM). The other eight eyes had ERM remnants. At the time of injection, no eye had silicone oil in the vitreous cavity; however, three out of 11 eyes had silicon oil post PPV, which was removed at least 4 months before considering suprachoroidal injection. There were no posterior hyaloid remnants in any of the 11 eyes.
Eleven suprachoroidal injections of triamcinolone (one administration per eye) were administered by the technique described above and were followed-up for 8 weeks. No additional treatment was required to manage the DME during follow-up.
Mean BCVA (log MAR) improved from 0.75 ± 0.40 (20/80) at baseline to 0.40 ± 0.33 (20/50) at 8 weeks (P = 0.003; Figure 3). Mean CMT reduced from 456.45 ± 113.42 μm at baseline to 294 ± 90 μm (35.5% reduction; P = 0.003) at 1 week following injection, with further reduction at week 4 to 256.36 ± 70.12 μm (43.9% reduction from baseline; P = 0.003 versus baseline), and at week 8 to 247.63 ± 53.40 μm (45.74% reduction from baseline; P = 0.003 versus baseline; Figure 4). There was no statistically significant change in mean IOP: 14.54 ± 1.4 mmHg at baseline and 14.72 ± 1.9 mmHg at week 8 of follow-up (P = 0.794).
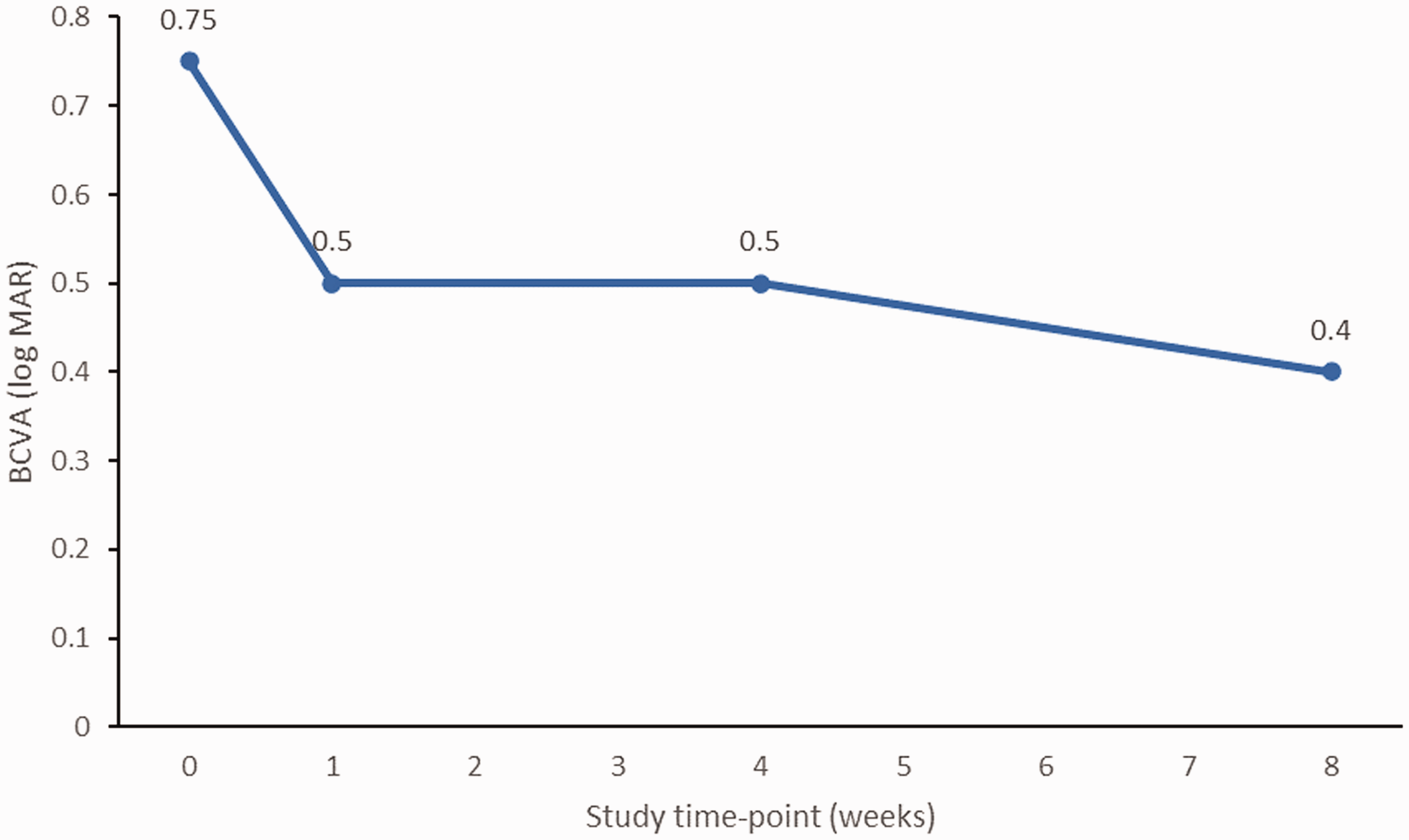
Changes in mean best-corrected visual acuity (BCVA) in 11 eyes, presented as log minimum angle of resolution [MAR] units, in response to suprachoroidal triamcinolone injection.

Changes in mean central macular thickness (CMT) in 11 eyes in response to suprachoroidal triamcinolone injection.
No complications occurred, such as increased IOP, intraocular triamcinolone penetration, central retinal artery occlusion, vitreous or suprachoroidal haemorrhages, retinal or choroidal detachments, conjunctival necrosis, endophthalmitis, retinal toxicity, or intraocular inflammation. The single phakic eye did not develop a cataract during the 8-week follow-up period.
Discussion
The present case series study retrospectively evaluated the efficacy and safety of a single suprachoroidal injection of triamcinolone acetonide to treat DME in 11 vitrectomized eyes, over an 8-week follow-up period.
A significant reduction in mean CMT from baseline was noted at 1 week post-injection (35.5% reduction from baseline), and further reduction was noted at 4 weeks post injection (43.9% reduction from baseline). The CMT remained significantly reduced versus baseline at week 8 of follow-up (45.74% reduction from baseline), with no statistically significant change in CMT between week 4 and week 8 post injection. Also, BCVA improved by approximately two lines during the 8-week post-injection follow-up.
Intravitreal injection of anti-VEGF in vitrectomized eyes has shown a lack of efficacy in reducing the CMT and visual acuity; 16 in contrast, the suprachoroidal injection of triamcinolone acetonide in the present case series showed improvement of both CMT and BCVA.
Diabetic macular edema is caused by thickened basement membrane and loss of pericytes, which lead to a compromise of the inner retinal-blood barrier, causing fluids to leak interstitially. 7 This mechanism is governed by the upregulation of VEGF and inflammatory cytokines, the latter of which has a more prominent role in chronic DME. 16 Steroids may play a role in the downregulation of inflammatory cytokines and VEGF, 17 and thus, intravitreal steroids can be useful in treating diabetic macular edema. 18
Other DME treatments are available, such as focal laser photocoagulation of the leaking microaneurysms, and are considered for non-central clinically significant macular edema. 19 Intravitreal anti-VEGF downregulates VEGF and enhances vascular permeability, which is considered first-line therapy for naïve or de novo cases. 20 PPV is considered in cases of tractional DME to relieve traction; 21 however, PPV in non-tractional DME can be considered when all other treatment fails, which may offer anatomical improvement as it increases oxygenation and mechanical VEGF removal. 21
In cases of vitrectomized eyes, the vitreous cavity lacks the vitreous, which acts as a reservoir for intravitreal anti-VEGF and steroids; hence, the half-life will be shortened. 5 In such cases, an intravitreal dexamethasone implant will provide sustained release of dexamethasone from a degradable polymer. However, targeting the DME from the suprachoroidal space using triamcinolone acetonide may be effective. For persistent cases of DME in particular, 22 as the suprachoroidal space is full of aqueous and is 1 ml in volume, 23 the triamcinolone should be cleared from the suprachoroidal space within 8 weeks. 24 Targeting the suprachoroidal space may offer increased triamcinolone efficacy, as it will be less diluted in the suprachoroidal space versus the vitreous cavity, with less interaction with the angle of the anterior chamber. 25 Hence, the suprachoroidal space may be an alternative route to the intravitreal cavity to treat DME post PPV.
In the present case series, DME presented in eyes that had undergone vitrectomy at a median of 11.72 (± 4.1) months previously, which may have impacted the visual recovery post PPV. Figure 5A shows an OCT cross-section of the eye of a 63 female patient included in the present case series. The eye was pseudophakic with DME at 19 months post PPV and presented with increased CMT of 406 μm, cystic changes, and some residual ERM prior to suprachoroidal injection treatment. Figure 5B shows a reduction of CMT to 265 μm, with most cystic changes resolved post suprachoroidal injection of triamcinolone, and vision improved from 20/50 to 20/32 within 8 weeks of follow-up.

Optical coherence tomography B-scan cross-sectional images of the eye of a 63 year-old female patient: (a) pseudophakic with diabetic macular edema at 19 months post pars plana vitrectomy who presented with increased central macular thickness (CMT) of 406 μm, cystic changes, and some residual epiretinal membrane at baseline; and (b) reduction of CMT to 265 μm, with most cystic changes resolved post suprachoroidal injection of triamcinolone, and improved vision from 20/50 to 20/32 within 8 weeks of follow-up.
The majority of eyes included in the present case report were pseudophakic, and intravitreal steroids are often recommended as a treatment for pseudophakic eyes. A study that investigated the efficacy of treatment to improve vision showed comparable results between intravitreal ranibizumab and triamcinolone adjunctive to focal/grid laser. 26 However, a study that compared focal/grid laser with intravitreal triamcinolone showed that triamcinolone is more effective in pseudophakic eyes. 27
Boyer et al. 7 showed that a dexamethasone implant improved vascular leakage and visual acuity in DME cases post PPV with an acceptable safety profile. However, Lee et al. 28 reported that a dexamethasone pellet might cause retinal injury during injection due to the absence of vitreous to slow down the dexamethasone pellet speed while injecting.
There were no complications during or after suprachoroidal triamcinolone injection in the present case series. The mean IOP was 14.54 ± 1.4 mmHg at baseline and 14.72 ± 1.9 mmHg at week 8 of follow-up, and no cataract developed in the single phakic eye. In the 11 eyes, there was no incidence of endophthalmitis, suprachoroidal haemorrhage, choroidal or retinal detachment, or penetration of triamcinolone into the intravitreal cavity. The absence of suprachoroidal haemorrhage is due to injection into the pars plana, which is avascular tissue.
To the best of our knowledge, this is the first case series to evaluate the safety and efficacy of suprachoroidal injection of triamcinolone for DME post PPV. Compared with intravitreal dexamethasone implant, the suprachoroidal injection of triamcinolone is cheaper and the delivery device can be hand-made with available material, which may be of particular use in low-resourced countries.
Limitations
The present results may be limited by several factors. Suprachoroidal injections carry a theoretical risk of choroidal haemorrhage and detachment. Patients with a thin scleral profile may receive an inadvertent injection of the drug into the vitreous cavity due to the fixed penetration depth of the needle. Patients need to be follow-up for a relatively long duration to conclusively prove the low incidence of changes in IOP and cataract formation, thus, the short follow-up duration in the present study is a limitation. The present study also lacked a control or sham group and was conducted in a single-centre, which limits the interpretation of the results. The safety results should be interpreted with caution due to the small sample size and short-term follow-up.
A larger multicentre prospective randomized clinical trial with a larger patient sample size and longer duration of follow-up is needed to evaluate the safety and efficacy of suprachoroidal injection of triamcinolone and to compare this treatment modality with other treatment options, such as intravitreal or posterior sub tenon triamcinolone, intravitreal dexamethasone implant, anti-VEGF, or focal Laser for DME in vitrectomized patients.
Conclusions
Suprachoroidal injection of triamcinolone to treat DME in vitrectomized eyes using a custom-made needle may reduce the macular thickness and improve vision with acceptable safety outcomes, and may be a useful treatment option for countries with limited resources. With the avoidance of delivering the drug into the vitreous cavity, the patients will not experience floaters, which is a frequent complaint post intravitreal triamcinolone acetonide. The authors aim to continue evaluating the utility of suprachoroidal triamcinolone injection in various other indications in ongoing clinical trials with a longer follow-up duration.
Footnotes
Acknowledgements
Special thanks to Ashish Sharma and Nilesh Kumar for their support in improving the structure of this manuscript.
Author contributions
AM and AZ searched, extracted and analysed the data and drafted the manuscript. AZ contributed to data interpretation and critical revision of the manuscript.
Declaration of conflicting interest
The Authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
