Abstract
Terson syndrome refers to intraocular haemorrhage that occurs due to subarachnoid bleeding associated with an acute increase in intracranial pressure. No previous study has reported a delayed macular hole (MH) secondary to Terson syndrome. A 17-year-old boy visited our department and presented with vitreous bleeding and a history of subarachnoid haemorrhage. Sub-internal limiting membrane (ILM) haemorrhage with ILM detachment and intraretinal haemorrhage were detected during pars plana vitrectomy. Additionally, a delayed MH was detected 1 week after the surgery. There was no sign of MH closure during a 2-month follow-up. Subsequently, an MH massage was performed to close the MH. Our findings suggest that a delayed MH can occur secondary to Terson syndrome. Elevated hydrodynamic pressure and hydrostatic pressure, which are caused by sub-ILM and intraretinal haemorrhages of the fovea, contribute to the formation of an MH. Additionally, ILM peeling may cause damage to the macula and facilitate the formation of MHs. Although the MH may close by itself, early surgical intervention is recommended when there is no sign that the MH will close spontaneously because a prolonged MH can lead to retinal damage.
Keywords
Background
Terson syndrome, which was first named in 1900 by the French ophthalmologist Albert Terson, refers to intraocular haemorrhage induced by subarachnoid bleeding associated with acutely elevated intracranial pressure. 1 Vitreous haemorrhage is the major symptom of Terson syndrome. In most cases, vitreous haemorrhage can be easily treated by pars plana vitrectomy, resulting in an immediate improvement of vision.2,3 However, Terson syndrome is associated with multiple complications, such as macular epiretinal membrane, retinal detachment, and retinal folds. 4 A delayed macular hole (MH) secondary to Terson syndrome has not been previously reported. We report a case of Terson syndrome accompanied by vitreous haemorrhage in both eyes, as well as sub-internal limiting membrane (ILM) and intraretinal haemorrhages within the macular region, which were detected during pars plana vitrectomy. Notably, a delayed MH secondary to Terson syndrome occurred after vitrectomy. We also reviewed the literature on an MH secondary to Terson syndrome to determine the characteristics of this complication, thereby enhancing our understanding of this disease and providing guidance for future surgical decisions.
Case report
The reporting of this study conforms to the CARE guidelines. 5 The patient and his parent provided written informed consent for the treatment.
A 17-year-old boy presented to our department with the chief complaint of blurry vision in both eyes for half a month. He suffered from subarachnoid haemorrhage with intracerebral bleeding associated with the spontaneous rupture of intracranial vascular malformation 2.5 months before the presentation. This spontaneous rupture caused a sudden loss of consciousness. The patient then underwent an operation of external ventricular drainage and intracerebral haematoma removal. After he recovered consciousness, 2 months later, he visited our department and complained of decreased vision in both eyes. There was no history of diabetes, hypertension, systemic vascular disease, inflammatory disease, ocular discharge, or ocular trauma. His family history was also unremarkable.
During an ophthalmological examination, his visual acuity was 1/20 in both eyes, and the intraocular pressure was 14.5 mmHg OD (right eye) and 13.7 mmHg OS (left eye). An anterior segment examination was unremarkable. A fundus examination showed dense and diffuse vitreous haemorrhage in both eyes (Figure 1a, b). Ocular ultrasound also revealed vitreous haemorrhage in both eyes with no evidence of retinal detachment (Figure 1c, d). Terson syndrome was diagnosed on the basis of his history of subarachnoid haemorrhage and vitreous haemorrhage in both eyes.
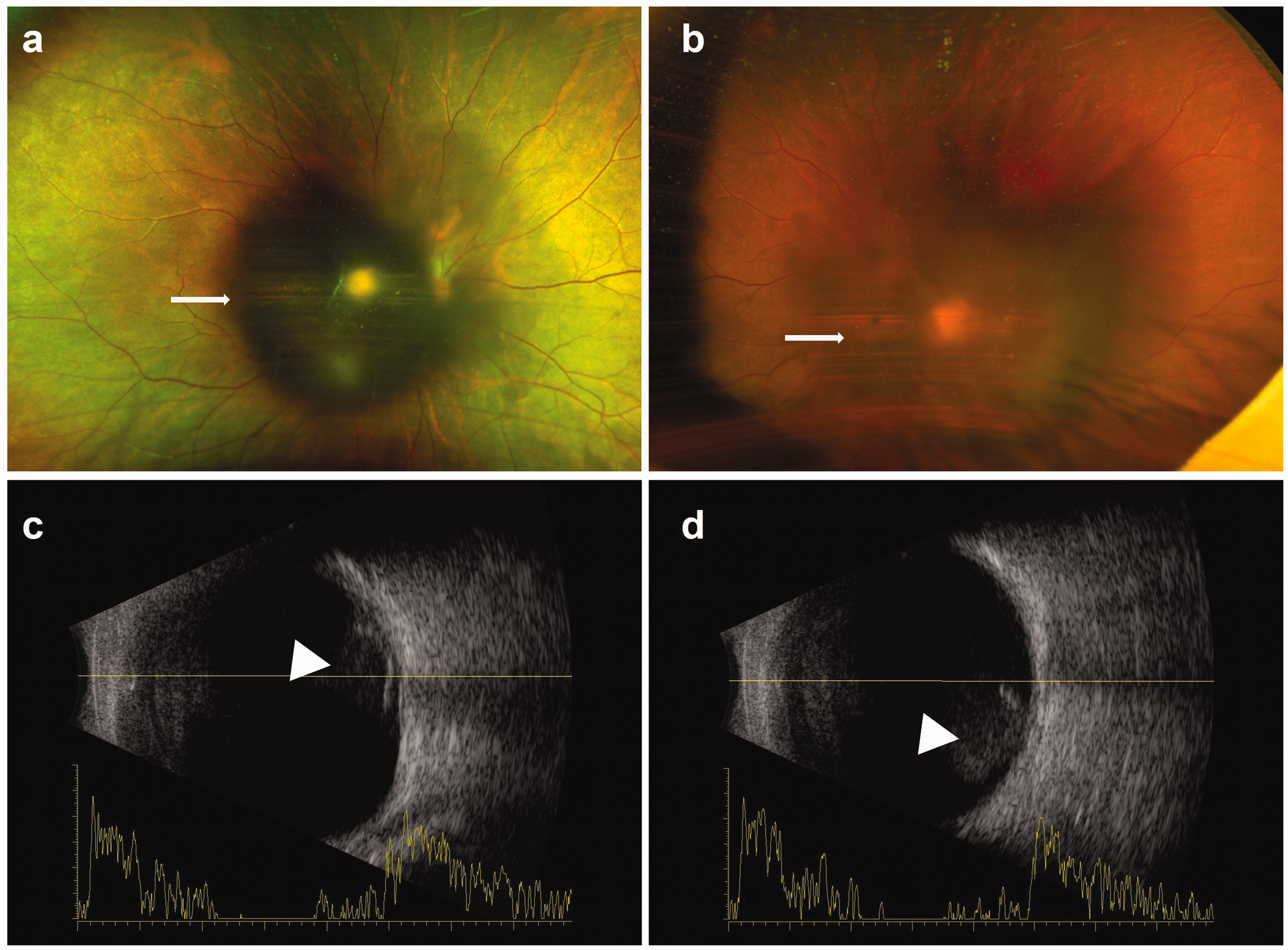
Preoperative fundus photographs and ultrasonographic images. (a, b) Scanning laser ophthalmoscopic images show vitreous haemorrhage in both eyes (white arrows) and (c, d) B-ultrasound images show vitreous opacity in both eyes (white arrowheads).
The patient underwent 25-gauge pars plana vitrectomy in both eyes separately. In his right eye, after removing the vitreous haemorrhage, sub-ILM haemorrhage with ILM detachment and intraretinal haemorrhage were observed in the macular region, but intraoperative optical coherence tomography (OCT) did not show an MH (Figure 2a–c).

Intraoperative optical coherence tomography images. (a) Fundus imaging from the retina’s view during surgery. (b) Intraoperative optical coherence tomography imaging shows a horizontal B-scan of the retina and (c) Intraoperative optical coherence tomography imaging shows a vertical B-scan of the retina. Sub-internal limiting membrane haemorrhage (white arrowhead) with internal limiting membrane detachments (stars) and intraretinal haemorrhage (white arrows) in the right eye can be seen.
Vitrectomy and ILM peeling were performed. One week after the operation, the patient reported slight improvement of his vision, and the best-corrected visual acuity (BCVA) of his right eye was 10/100. However, a full-thickness MH was shown by OCT (Figure 3a, b). One week later, air tamponade was performed on his right eye. However, after intravitreal air absorption, an OCT scan showed a persistent MH in his right eye (Figure 3c, d), and the MH remained at the 1-month (Figure 3e, f) and 2-month follow-ups (Figure 3g, h). We performed an MH massage for his right eye and administered air tamponade because of the persistent existence of MH. Ten days after the last surgery, the MH was closed (Figure 3i, j). One month later, the macular structure showed evidence of gradual reconstruction (Figure 3k, l). Furthermore, the BCVA had improved to 5/20.

Postoperative optical coherence tomography images. The images show changes in an MH in the right eye. (a, b) Optical coherence tomography images show an MH at 1 week after vitrectomy of the right eye. After intravitreal air tamponade of the right eye, the MH remained at the 1-week (c, d), 1-month (e, f), and 2-month follow-ups (g, h). The MH was then completely closed at 10 days (i, j) and 1 month and (k, l) after an MH massage for the patient’s right eye was performed. MH, macular hole.
Vitrectomy and ILM peeling were also performed on his left eye. During the surgery, we did not detect an MH or retinal haemorrhage. At the 1-week postoperative follow-up, the BCVA of the left eye had improved to 20/20, and OCT showed a normal macular structure of his left eye.
Discussion
Terson syndrome is defined as the presence of intraocular haemorrhage associated with intracranial haemorrhage and elevated intracranial pressure. 6 Based on the location of the haemorrhage, the clinical classification of intraocular haemorrhage can be categorized as sub-ILM, preretinal, intraretinal, subhyaloid, or intravitreal. 7 The physiopathology of Terson syndrome and the origin of intraocular blood remain controversial. The most accepted theory for this physiopathology is that a sudden increase in intracranial pressure transmitted to the optic nerve sheath leads to venous congestion and causes the small retinal veins to burst.8,9
To better understand the characteristics of an MH secondary to Terson syndrome, we conducted a literature search. We found that an MH secondary to Terson syndrome was observed in only nine eyes in seven articles4,6,10–14 (Table 1). Although MH occasionally occurs as a complication of Terson syndrome, the exact cause is unclear. Elevated hydrodynamic pressure caused by retinal haemorrhages may be an important cause of an MH. 15 However, there have been no reports of delayed MH secondary to Terson syndrome after vitrectomy. Moreover, secondary MH formation after vitrectomy and ILM peeling is rare, and its pathogenesis is not clearly understood. During the surgery in our case, intraoperative OCT did not show any signs of an MH. A delayed MH was observed only at 1 week postoperatively. The findings in our case indicate that there are other factors, except tractional components, involved in the formation of a secondary MH.
Summary of previously published cases of a macular hole secondary to Terson syndrome.

VA, visual acuity; ILM, internal limiting membrane; MH, macular hole; HM, hand motion; NA, not available; CF, counting fingers.
Notably, we detected sub-ILM haemorrhage with ILM detachment and intraretinal haemorrhage of the fovea during the surgery in our patient. Physicians should be aware of the role of sub-ILM and intraretinal haemorrhages in the formation of a delayed MH. Based on the characteristics of our case, we hypothesize that the hydrodynamic pressure combined with the change in hydrostatic pressure in the fovea contributed to the formation of a delayed MH. The most widely referenced mechanism of an MH suggests that a sudden increase in intracranial pressure causes venous hypertension and stasis, leading to retinal vessel rupture. Sudden bleeding may cause elevated hydrodynamic pressure of the fovea and weaken the fovea. The subsequent long-term presence of blood in the sub-ILM and intraretinal spaces may then also cause hydrostatic pressure changes and retinal degeneration in the fovea. This theory can be proved by clinical manifestation in the other eye of the same patient. During the surgery on our patient’s left eye, we did not detect sub-ILM or intraretinal haemorrhage, and the macular structure remained normal after surgery.
The ILM is important for maintaining the normal structure of the macula. Histological studies of human eyes after ILM removal have shown that it causes damage to a substantial number of Müller cell processes and end-feet. 16 Additionally, changes in the superficial and deep retinal layers after ILM peeling have previously been reported. 17 More tractional force exerted to peel the ILM may induce more damage to ganglion cells and affect Müller cell integrity. 17 Therefore, ILM peeling should be performed with caution because traction may lead to the formation of MHs. During the surgery, we removed the vitreous haemorrhage and ILM, which may have caused a slight pressure change and iatrogenic traction within the unhealthy macular region, thereby resulting in delayed MH formation. The findings of this case study should be considered when planning and performing vitreoretinal surgery for patients with Terson syndrome in whom sub-ILM and intraretinal haemorrhages present within the macular region. Surgeons should be careful in addressing the macular region and consider the potential need to repair an MH.
A previous report showed that delayed closure may occur in MHs secondary to Terson syndrome. 6 In this case, we followed up the patient for 2 months, and there was no sign of MH closure. Therefore, we performed MH massage for this patient, which helped the MH of his right eye to close after the surgery. In addition, the BCVA of his right eye was partially recovered. Even in cases with spontaneous closure of the MH, because of prolonged defects of the choriocapillaris layer, complete visual recovery is uncommon. 18 These findings indicate that early surgical intervention in this situation is advisable.
In conclusion, this is the first study to present a case of delayed MH secondary to Terson syndrome. The MH formed owing to changes in hydrodynamic pressure and hydrostatic pressure caused by sub-ILM and intraretinal haemorrhages of the fovea. Surgical procedures may exacerbate pressure changes, and cause iatrogenic traction in the macula and accelerate delayed occurrence of an MH. Although the MH may close spontaneously, early surgical intervention is recommended when there is no sign that the MH will close spontaneously because a prolonged MH can lead to retinal damage.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241233125 - Supplemental material for Delayed macular hole secondary to Terson syndrome: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605241233125 for Delayed macular hole secondary to Terson syndrome: a case report and literature review by Yi Zhang, Chunyan Lei, Xi Huang and Meixia Zhang in Journal of International Medical Research
Footnotes
Author contributions
MZ, YZ, CL, and XH participated in the diagnosis and treatment of the patient. YZ drafted the manuscript. MZ and CL reviewed the manuscript. All authors provided critical feedback and approved the submitted version.
Data availability statement
All of the data supporting our findings are contained within the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal. No identifiable health information was included in this case report. Therefore, the need for approval was waived.
Funding
This work was supported by the Natural Science Foundation of Science and Technology Department of Sichuan Province (No. 2023NSFSC1665).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
