Abstract
Eosinophilic granulomatosis with polyangiitis (EGPA) is a type of eosinophilic vasculitis that is mainly limited to small- and medium-sized arteries. Cardiac involvement is the leading cause of death in patients with EGPA. Spontaneous coronary artery dissection (SCAD) is an important cause of acute coronary syndrome in middle-aged women with no or few traditional cardiovascular risk factors. EGPA manifesting as repetitive acute coronary syndrome and SCAD has not been reported. A 45-year-old woman presented with recurrent chest pain and cardiogenic shock associated with coronary vasospasm refractory to common vasodilators. Coronary angiography showed SCAD at the proximal right coronary artery. Blood tests showed significant eosinophilia. In addition to sinusitis as shown by nasal computed tomography and abnormal nerve conduction velocity, the diagnosis of EGPA was made and immunosuppression commenced. During a 20-month follow-up, the patient remained free from symptoms and adverse cardiovascular events. EGPA can involve coronary arteries and may rarely manifest as SCAD or vasospasm. We herein review the mechanism underlying coronary involvement of EGPA and emphasize special clues for its detection. Early recognition and initiation of immunosuppression therapy are important.
Keywords
Introduction
Eosinophilic granulomatosis with polyangiitis (EGPA) is a type of eosinophilic vasculitis that is mainly limited to small- and medium-sized arteries. Cardiac involvement is the leading cause of death in patients with EGPA. Spontaneous coronary artery dissection (SCAD) is an important cause of acute coronary syndrome (ACS) in middle-aged women with no or few traditional cardiovascular risk factors. There is a wide spectrum of clinical presentations and severities of SCAD, ranging from unstable angina to sudden cardiac death, depending upon the grade and severity of the dissection. ST-elevation myocardial infarction is present in up to 50% of patients, and non-ST elevation myocardial infarction (NSTEMI) is observed in the other half. Various conditions and predisposing factors are associated with SCAD, 1 including fibromuscular dysplasia, pregnancy, inherited connective tissue disorders, and vasculitis. EGPA-induced SCAD has not been reported to date. We herein describe a patient with EGPA who presented with repetitive ACS, cardiogenic shock, refractory coronary vasospasm, and SCAD. We review the rare coronary involvement of EGPA and emphasize special clues for its early detection.
Case report
The reporting of this study conforms to the CARE guidelines. 2 Written informed consent for treatment and publication of the case and accompanying images was obtained from the patient. A formal ethical review by an ethics committee was not required because of the nature of this study (case report).
A 45-year-old woman with recurrent chest pain and cardiogenic shock was referred to our center. Eight months previously, the patient had first presented with chest pain, ST-segment depression, and troponin elevation, indicating NSTEMI. She was admitted to a local hospital, but cardiac catheterization revealed a normal coronary angiogram. Coronary spasm was suspected, and the patient was treated with diltiazem. Despite treatment compliance, she still developed recurrent symptoms. Five months later, her chest pain recurred and quickly evolved into cardiogenic shock. An electrocardiogram showed transient ST-segment elevation in the inferior and posterior leads (Figure 1(a)). She was successfully resuscitated with intubation, mechanical ventilation, and vasopressors (epinephrine and others). However, secondary coronary angiography was still inconclusive. After recovery, she was discharged with aspirin, clopidogrel, a statin, and diltiazem. During the next 3 months, she experienced increasingly more frequent episodes of chest pain and required several hospitalizations and outpatient visits (Supplementary Timeline). Three days before admission, she experienced persistent chest paint and dyspnea at home, and she quickly became unconscious and pulseless. Cardiac arrest was diagnosed by the emergency medical services. No ventricular arrythmia was documented. She was successfully resuscitated on site.
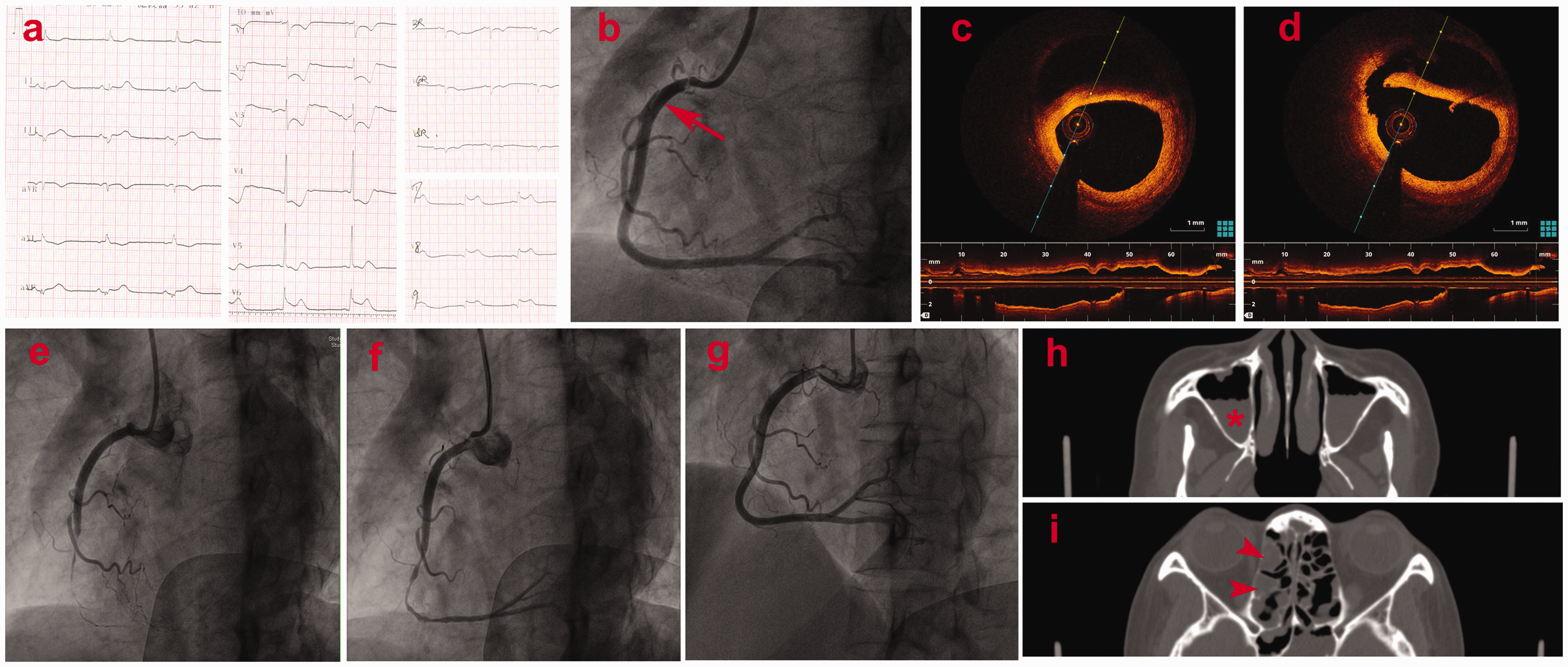
Various imaging findings of the patient. (a) Eighteen-lead electrocardiogram results. (b) Coronary angiography showed a dissection (arrow) at the proximal right coronary artery. Representative cross-sectional optical coherence tomography images of the (c) middle and (d) proximal segment of the dissection. Severe vasospasm of the right coronary artery (e) after stenting and (f) refractory to various vasodilators. (g) Four days later, the fourth angiographic examination revealed a normal angiogram and patent stents. Computed tomography showed changes consistent with chronic sinusitis, including (h) effusion (asterisk) in the maxillary sinuses and (i) mucosal thickening (arrowhead) in the ethmoid sinuses.
The patient had a 3-year history of asthma that had been treated with inhaled salmeterol/fluticasone. Her asthma episodes were previously infrequent but had increased in number since the onset of chest pain. No hypertension, diabetes, dyslipidemia, or allergy to food or drugs was documented.
On admission, the patient had severe chest pain. Electrocardiography revealed bradycardia (51 beats/minute) and significant ST depression in the precordial leads. She was hypotensive with a blood pressure of 75/50 mmHg, and norepinephrine was mandatory. After low-dose nitroglycerin infusion, her symptoms and ST deviation resolved and she became normotensive. Cardiac magnetic resonance imaging revealed small areas of myocardial infarction in both the right coronary artery (RCA) and the left circumflex territories.
The patient underwent her third angiographic examination, which revealed SCAD at the proximal RCA with uncompromised blood flow (Figure 1(b)). Optical coherence tomography (OCT) confirmed that the false lumen involved the outer third of the media and communicated with the true lumen (Figure 1(c) and (d)). Two drug-eluting stents were deployed, and OCT revealed optimal apposition/expansion and full cover of the dissection. About 5 minutes later, however, severe vasospasm with compromised blood flow occurred in the RCA (Figure 1(e)). Repeated OCT showed no distal hematoma squeeze. After repetitive dosing (12 boluses in total) of various types of intracoronary vasodilators (nitroglycerin, nitroprussiate, and verapamil), the blood flow was grade III but the vasospasm persisted (Figure 1(f)). The patient was treated with ticagrelor monotherapy in addition to diltiazem. Her symptom was then well controlled.
Four days later when she was rechallenged with aspirin, her chest pain recurred and a similar electrocardiographic change was observed. Because of concern regarding stent thrombosis, a fourth angiographic examination was performed but revealed a normal angiogram and patent stents (Figure 1(g)).
During the patient’s hospital stay, her eosinophil count fluctuated, peaking at 1.27 × 109/L (18% of total leukocytes). EGPA was highly suspected. Antineutrophil cytoplasmic antibodies (ANCAs), including anti p-ANCA and c-ANCA, were negative. However, computed tomography demonstrated significant nasal sinus effusion and mucosal thickening, suggestive of sinusitis (Figure 1(h) and (i)). Furthermore, a nerve conduction velocity and sensation test revealed polyneuropathies. She refused biopsy because of the bleeding risk. The patient was diagnosed with EGPA according to the American College of Rheumatology criteria. 3
The patient was then referred to the rheumatology team and received methylprednisolone pulse therapy combined with rituximab (anti-CD20 antibody) infusion, followed by tapered-dose prednisolone and cyclosporine. During 20 months of follow-up, the patient remained free from symptoms and adverse cardiovascular events. Her blood eosinophil count remained lower than 0.1 × 109/L.
Discussion
Early in this case, iatrogenic and traumatic coronary dissection were excluded because OCT did not show obvious atherosclerosis in all three coronary arteries. The dissection was unlikely to be the consequence of a penetrating ulcer or plaque rupture secondary to atherosclerotic disease. We also screened the carotid and renal arteries with sonography, but no arterial abnormalities such as stenosis, aneurysm, tortuosity, or dissection were revealed; therefore, fibromuscular dysplasia was excluded as the cause of SCAD. Furthermore, refractory coronary vasospasm persisted despite repetitive administration of different vasodilators. These findings, in addition to the progressive aggravation of the disease course, raised our suspicion of an undetected cause.
EGPA is a rare disease characterized by eosinophil infiltration of the adventitia. Only 40% of patients produce detectable ANCAs. Previous studies have shown that ANCA positivity is associated with more lung and kidney involvement, whereas ANCA negativity tends to have a high incidence of cardiac involvement, 4 as in the present case. Data regarding cardiac involvement of EGPA are limited. Recent cohort studies revealed a 16% to 62% prevalence of cardiac involvement in patients with EGPA.4–6 The cardiac manifestations of EGPA are highly variable, ranging from ACS, myocarditis, pericarditis, heart failure, arrythmia, cardiogenic shock, and ventricular thrombosis to coronary aneurysm. Cardiac involvement of EGPA is associated with a worse prognosis. Therefore, early recognition is critical. The six diagnostic criteria for EGPA are asthma, eosinophilia of ≥10%, mononeuropathy or polyneuropathy, non-fixed pulmonary infiltrates on chest X-rays, nasosinusitis, and extravascular infiltration of eosinophils in biopsy specimens. However, just the presence of asthma in addition to eosinophilia of ≥10% yields high sensitivity of 90.0% and specificity of 99.7%. 3 Therefore, EGPA should be highly suspected in patients with these two manifestations, even when ANCAs are negative, as in this case.
Given the substantial clinical overlap between EGPA and hypereosinophilic syndrome (HES), the differentiation between these two diseases remains challenging in the absence of biopsy-proven eosinophilic vasculitis, especially in ANCA-negative patients. However, two facts support EGPA rather than HES in our case. First, the absolute eosinophil count in our patient was lower than 1.0 × 109/L in most instances throughout her clinical course; this is far from the HES criteria (>1.5 × 109/L persisting for 6 months). Second, Leurs et al. 7 reported that the C-reactive protein (CRP) level was helpful in differential diagnosis of HES from ANCA-negative EGPA in that a low serum CRP level was suggestive of HES. The CRP level in our patient was persistently elevated and in most instances was much higher than 3 mg/L, which was the median level among patients with HES in the cohort of the study by Leurs et al. 7
SCAD due to EGPA has not been reported to date. Although SCAD with significant coronary eosinophilic infiltration at autopsy has been described,8–11 these cases neither showed eosinophilic vasculitis outside of the coronary arteries nor had other manifestations consistent with EGPA, such as asthma or eosinophilia. Therefore, our report describes the first case of SCAD caused by EGPA. Although we did not have pathological evidence of eosinophilic coronary infiltration, the sub-adventitial dissection shown by OCT and the multisystem involvement supported our hypothesis. The mechanism underlying the development of EGPA-induced SCAD remains elusive. Coronary eosinophilic vasculitis has been reported in patients with HES. 12 Lytic substances including lysosomes and proteinases produced by eosinophils destroy collagen, elastin, and smooth muscle cells, resulting in arterial necrosis and disruption. Other potential mechanisms include the production of vasospastic mediators (including leukotrienes C4 and D4) and modulation of mast cell functions by eosinophils. Severe spasm increases arterial wall shear stress, and several different mechanical triggers have been reported in patients with SCAD. 13 Additionally, physical and emotional stress due to chest compression, intubation, repetitive hospital admissions, and repeated angiography procedures may also contribute to SCAD.
A conservative strategy is generally preferred for SCAD except in high-risk patients, such as those with compromised coronary flow, left main artery involvement, or hemodynamic instability. 14 However, the strategy should be tailored on a case-by-case basis. In our patient, SCAD in the proximal RCA, progressive deterioration of chest pain, and the potential risk of dissection progression before control of the coronary vasculitis indicated the need for a more invasive strategy.
Because no ventricular arrhythmia or high-degree atrioventricular block had been documented in our patient, the cardiogenic shock was probably caused by severe and extensive coronary vasospasm or coronary vasospastic crisis by EGPA; this was supported by significant precordial ST-segment deviation after aspirin rechallenge despite RCA stenting. Multivessel SCAD has been reported,9,11 but it was excluded in this case because OCT showed that the structure of the left descending and circumflex arteries was intact. Coronary vasospastic crisis leading to cardiogenic shock and recurrent ventricular fibrillation has also been reported. 15 For safety reasons, we did not perform a provocative test.
Dual antiplatelet therapy (DAPT) is the standard of care after coronary stent implantation. In our patient, the atherosclerotic cardiovascular disease burden was very low in the OCT investigation, and the patient received two stents of large caliber (3.0 and 3.5 mm, respectively); however, she had a low body weight (40 kg). We believe that the ischemic risk was low while the bleeding risk was high. In light of the comparable results of short-term DAPT from the GLOBAL LEADERS 16 and STOPDAPT-2 17 trials, we prescribed ticagrelor monotherapy (90 mg twice daily) in this case.
The reason for the frequent attacks in this patient deserves further scrutinization. Besides EGPA disease progression, the role of aspirin cannot be neglected. Aspirin intake might induce vasoconstriction or spasm, as reported in patients with Kounis syndrome 18 or aspirin-exacerbated respiratory disease. 19 In a retrospective study of 153 patients with aspirin-exacerbated respiratory disease who had no evidence of coronary atherosclerosis, 10 patients had a history of chest pain. Of these 10 patients, 6 reported exacerbation of chest pain while on high-dose aspirin therapy. Their chest pain did not improve with typical antianginal treatments but did respond to corticosteroid therapy, 19 as in our case. This is also the second reason for omitting aspirin from DAPT after stent implantation.
No large-scale clinical trial has addressed the treatment of EGPA complicated with heart involvement. Successful treatment with prednisone or immunosuppressive agents has been reported in some cases.20,21 More recently, the anti-interleukin-5 antibody benralizumab showed promising results in a patient with recurrent cardiac arrest due to eosinophilia-related coronary vasospasm. 22
The major limitation of the present study is the absence of biopsy. However, vasculitis is not always detectable in every biopsy specimen (only 55.6% in MPO-ANCA-positive cases). 23 The diagnosis of EGPA was made in a comprehensive and integrated manner in our patient, even without biopsy-proven vasculitis.
Conclusion
Our case indicates that EGPA can involve the coronary arteries and rarely manifest as SCAD or vasospasm. EGPA should be highly suspected in patients with asthma and eosinophilia, even those with ANCA negativity. Early recognition of the condition and initiation of immunosuppression therapy are important.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221089516 - Supplemental material for Eosinophilic granulomatosis with polyangiitis presenting with repetitive acute coronary syndrome, refractory coronary vasospasm, and spontaneous coronary dissection: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221089516 for Eosinophilic granulomatosis with polyangiitis presenting with repetitive acute coronary syndrome, refractory coronary vasospasm, and spontaneous coronary dissection: a case report by Maohuan Lin, Zizhuo Su, Jianzhong Huang, Jiajie Li, Niansang Luo and Jingfeng Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221089516 - Supplemental material for Eosinophilic granulomatosis with polyangiitis presenting with repetitive acute coronary syndrome, refractory coronary vasospasm, and spontaneous coronary dissection: a case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605221089516 for Eosinophilic granulomatosis with polyangiitis presenting with repetitive acute coronary syndrome, refractory coronary vasospasm, and spontaneous coronary dissection: a case report by Maohuan Lin, Zizhuo Su, Jianzhong Huang, Jiajie Li, Niansang Luo and Jingfeng Wang in Journal of International Medical Research
Footnotes
Acknowledgements
We gratefully acknowledge Dr. Elizabeth Dawn Rockholt for manuscript editing and proofreading. We also thank Dr. Zhuzhi Wen, Dr. Kun Zhang, Dr. Yong Xie, Dr. Woliang Yuan, and Dr. Qianhua Li for their constructive suggestions and support during the care of this patient.
Author contributions
All authors were directly involved in the patient care as well as the performance and interpretation of the patient investigations. Maohuan Lin was the patient’s treating physician and performed the OCT examination and coronary stenting. Zizhuo Su contributed to the data acquisition. Jianzhong Huang was the patient’s primary care physician. Jiajie Li was responsible for the OCT analysis. Niansang Luo and Jingfeng Wang were the consultant physicians who supervised the patient’s care.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Guangdong Basic and Applied Basic Research Foundation [grant No. 2021A1515012042 to Maohuan Lin].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
