Abstract
Rosia–Dorfman disease (RDD) is a rare benign disease with exceedingly rare neural involvement. In this report, we describe a very rare case of intracranial RDD mimicking a subdural hematoma. The patient presented with headache and a suspected history of head injury. Head computed tomography showed a well-demarcated extra-axial crescentic hyperdense lesion along the right hemisphere convexity, and this lesion was initially mistaken for a subacute subdural hematoma with brain contusion. However, the follow-up examinations during the subsequent month did not show the natural changes characteristic of a subdural hematoma. Thus, a magnetic resonance imaging scan was performed, which showed a crescentic lesion with T1 isointensity, T2 hypointensity, and obvious homogeneous enhancement. Differential diagnoses such as lymphoma and metastases were considered, and surgery was performed. The final histopathologic examination confirmed the diagnosis of RDD. This report describes a rare presentation of RDD. RDD can mimic the appearance of a subdural hematoma and should be considered as a differential diagnosis even in patients with a history of head injury and a “classic” imaging appearance of a subdural hematoma.
Keywords
Introduction
Rosai–Dorfman disease (RDD) is a rare benign disease characterized by sinus histiocytosis with massive lymphadenopathy. It was first described by Rosai and Dorfman in 1969. 1 This lymphoproliferative disorder particularly affects the head and neck region, and it is associated with extranodal involvement in 43% of cases. 1 Central nervous system (CNS) involvement of RDD is exceedingly rare, accounting for approximately 7.8% of all reported cases. 2 In this report, we describe a very rare case of CNS involvement of RDD mimicking a subdural hematoma. The reporting of this study conforms to the CARE guidelines. 3
Clinical presentation
A 32-year-old man presented with an almost 1-month history of headache. He had a suspected history of head injury caused by falling down in the morning 1 month previously. His physical examination findings were normal. His erythrocyte sedimentation rate (ESR) was elevated at 38.00 mm/hour (reference range, 0.00–15.00 mm/hour). Head computed tomography (CT) showed a well-demarcated extra-axial crescentic hyperdense lesion along the right hemisphere convexity with marked perilesional edema (Figure 1). This finding was thought to be consistent with a subacute subdural hematoma with brain contusion and edema because of the classic imaging appearance and history of injury. There was no evidence of bone fracture or hyperostosis.

A series of head computed tomography scans on (a, b) day 1, (c) day 21, and (d) day 42 after admission showed a well-demarcated extra-axial crescentic hyperdense lesion along the right hemisphere convexity with marked perilesional edema. The density of the lesion was stable without any changes in its course. The density of the adjacent skull was increased without bone fracture, erosion, or hyperostosis.
After several follow-up examinations during the subsequent month, the crescentic hyperdense shadow remained without any changes in its density (Figure 1). Thus, the patient was suspected to have a chronic subdural hematoma. However, to exclude other possibilities, a magnetic resonance imaging (MRI) scan was performed. The MRI scan showed a crescentic lesion with T1 isointensity and T2 hypointensity in the right hemisphere convexity with perilesional edema. The lesion and the dura adjacent to the lesion were obviously homogeneously enhanced after administration of gadolinium contrast. Additionally, the lesion showed pseudopod-like enhancement deep into the sulcus. The adjacent skull was enhanced as well. The adjacent brain parenchyma showed perilesional edema (Figure 2). Considering that the density of the lesion was not consistent with the evolution of a subdural hematoma, differential diagnoses such as lymphoma and metastases were considered. Craniotomy was conducted to clarify the diagnosis. The lesion appeared as white fish-like tissue that had no obvious boundary with the dura and adhered closely to the adjacent brain parenchyma. It was very difficult to completely resect the lesion, and partial excision was therefore conducted. Histopathologic examination demonstrated infiltration of lymphocytes, plasma cells, and histiocytes in the stroma with proliferative fibrous connective tissue. Some histiocytes exhibited features of emperipolesis (enlarged histiocytes enclosing well-preserved mononuclear inflammatory cells within their cytoplasm) (Figure 3). Immunohistochemical analysis showed that the histiocytes stained diffusely positively for S100, CD68, and CD163 and stained negatively for CD1α, CD21, and CD23. 4 Therefore, a final diagnosis of RDD was made. A whole-body CT scan ruled out any other organ involvement. The patient’s headache was relieved after the surgery.
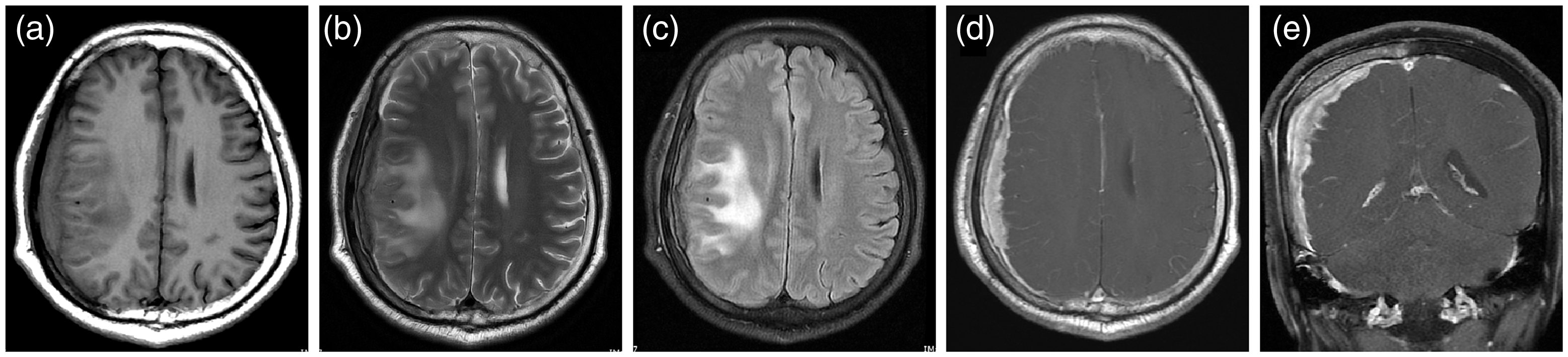
Preoperative magnetic resonance imaging, (a) T1-weighted imaging, (b) T2-weighted imaging, (c) T2 fluid-attenuated inversion recovery imaging, (d) axial T1 contrast, and (e) coronal T1 contrast showed a T1 isointense and T2 hypointense crescentic lesion in the right hemisphere convexity with perilesional edema. The lesion and the dura adjacent to the lesion were obviously homogeneously enhanced. The lesion exhibited pseudopod-like enhancement deep into the sulcus. The adjacent skull was enhanced as well.
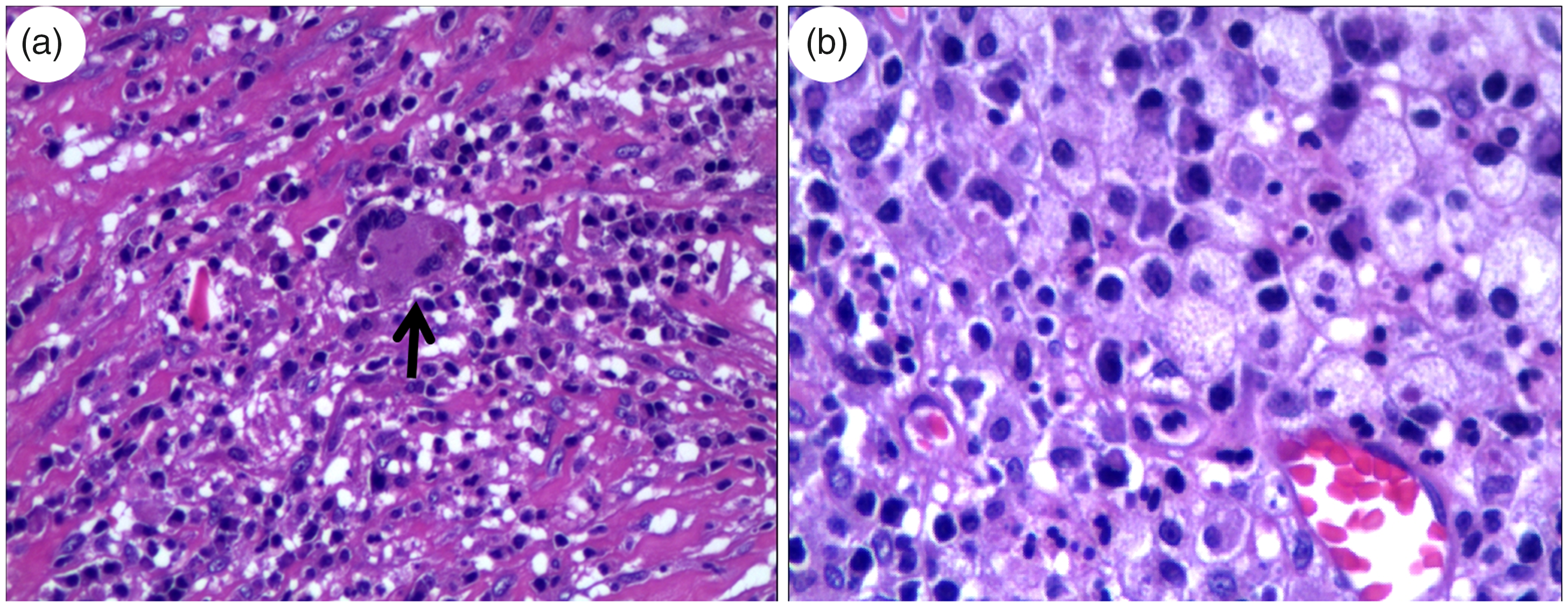
(a) Photomicrograph (hematoxylin and eosin; original magnification, ×200) showed emperipolesis (large histiocytes with prominent intracytoplasmic lymphocytes) (arrow). (b) Photomicrograph (hematoxylin and eosin; original magnification, ×400) showed numerous proliferative, large pale histiocytes with eosinophilic cytoplasm on a background of many plasma cells and lymphocytes.
Discussion
CNS involvement of RDD is very rare. In approximately 30% of cases, RDD manifests with fever, a systemic inflammatory state, anemia, polyclonal hypergammaglobulinemia, and an elevated ESR. 5 Intracranial RDD presents with varied symptoms, such as headache, dizziness, seizure, visual disturbances, numbness, and cranial nerve deficits according to the location of the lesion. 6 The most common appearance of intracranial RDD is the presence of well-defined dural-based extra-axial masses associated with surrounding vasogenic edema. The masses often exhibit homogeneous enhancement after contrast administration. 7 This typical imaging feature of intracranial RDD makes it difficult to differentiate from meningioma. 8 Approximately 90% of all cases of intracranial RDD involve the leptomeninges. 9 The present case is exceptionally rare because it presented with CNS involvement and mimicked a subdural hematoma. To the best of our knowledge, this is the second reported case of RDD that was misdiagnosed as a subdural hematoma. The first such case was reported by Kumar et al. 7 in 2008.
The presence of headache and an elevated ESR is consistent with the manifestations of intracranial RDD, although these findings are not specific. This case was misdiagnosed as a subdural hematoma because of the history of trauma and the presence of a crescentic high-density shadow on CT. However, after careful consideration of this case, we determined that two main factors can be used to differentiate RDD from a subdural hematoma: the density transformation on CT and the signal on MRI.
First, with respect to the density transformation on CT, the density of a subdural hematoma depends predominantly on the age of the hemorrhage and stage of blood degradation. Therefore, most subdural hematomas follow a fairly predictable course on serial CT scanning. Subdural hematomas are hyperdense compared with the cerebral cortex in the acute stage, gradually decrease in density over time, and become hypodense in the chronic stage. During this chronic process, they may also exhibit other densities such as isodensity, gradation density, and mixed density. 10 The lesion in the present case remained hyperdense on CT with no changes in more than 1 month. This was inconsistent with a subdural hematoma.
Second, with respect to the signal on MRI, the lesion in this case showed isointensity on T1-weighted imaging and isointensity with hypointensity on T2-weighted imaging and T2 fluid-attenuated inversion recovery. The hypointense signal on T2-weighted imaging is reportedly caused by free radical production during phagocytosis as well as necrotic and fibrotic changes. 6 The MRI appearance of a subdural hematoma is related to the age of the hematoma. In the chronic stage, most subdural hematomas exhibit a hypointense T1 signal and isointense to hypointense T2 signal. Some chronic subdural hematomas remain hyperintense on T2-weighted imaging. Repeat hemorrhage, which is common in subdural hematomas, contributes to the heterogeneous appearance on MRI. 10 Therefore, there is some overlap in the signal appearance on non-contrast MRI between RDD and subdural hematomas. A contrast-enhanced scan could aid in the differentiation. The fibrous capsule and internal septations of chronic subdural hematomas are vividly enhanced on postcontrast T1-weighted images. Unlike a subdural hematoma, our patient’s lesion was markedly and homogenously enhanced after the administration of contrast agent and demonstrated a dural tail sign. Additionally, the lesion showed pseudopod-like enhancement deep into the sulcus, suggesting leptomeningeal involvement, which is rare in subdural hematomas. The adjacent skull was enhanced as well.
The imaging appearance of this case required differentiation from certain neoplasms, such as lymphomas and dural-based metastases, as well as from granulomatous disorders such as sarcoidosis.
Conclusion
RDD can mimic the appearance of a subdural hematoma and should be considered as a differential diagnosis even in patients with a history of head injury and the “classic” imaging appearance of a subdural hematoma. Careful dynamic observation of the intensity or signal of the lesion is essential to reach a correct diagnosis.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics and consent
The requirement for ethical approval was waived because of the nature of the study (retrospective case report). The patient described in this report provided verbal informed consent.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
