Abstract
Objective
To compare the initial clinical course and data on 90-day mortality in adults with methanol (MET) or ethylene glycol (EG) poisoning treated with dialysis.
Methods
Data on patient demographics and clinical parameters at intensive care unit (ICU) admission and for the first 24 hours after dialysis initiation were collected, and 90-day outcome data were collected for patients with MET (n = 15) or EG (n = 13) poisoning treated with dialysis in this retrospective cohort study.
Results
In univariate analysis, patients with EG poisoning were older and they had lower hourly urine output during the first 24 hours after the initiation of dialysis. Six (46%) patients with MET poisoning and three (20%) patients with EG poisoning died within 90 days of ICU admission. A larger anion gap and lower pH, bicarbonate levels, base excess, and Glasgow Coma Scale scores on admission, as well as the need for mechanical ventilation, were associated with 90-day mortality.
Conclusions
Metabolic acidosis, a large anion gap, and an altered mental status on admission appear to be associated with mortality in MET or EG poisoning, and EG poisoning may be linked to lower urine output.
Keywords
Introduction
Methanol (MET) and ethylene glycol (EG) are considered extremely toxic substances. MET is a clear liquid that has a similar taste as ethanol, and it is commonly used in industrial products such as windshield washer fluid. 1 EG is an odorless and colorless liquid found in antifreeze and other chemical compounds. 2 Ingested MET and EG are metabolized by alcohol dehydrogenase (ADH) and then aldehyde dehydrogenase. MET is metabolized into formaldehyde and then formic acid, which inhibits mitochondrial respiration, leading to cellular hypoxia and cytotoxicity. EG is oxidized into glycolic acid and finally oxalic acid. Oxalic acid causes end-organ toxicity through the deposition of precipitated calcium oxalate crystals in various tissues. Furthermore, oxalic acid and glycolic acid induce tissue toxicity and metabolic acidosis.3–5
Patients with voluntary or inadvertent MET or EG poisoning often present with analogous symptoms that may resemble those of other common acute illnesses such as diabetic ketoacidosis or sepsis. Furthermore, the laboratory tests needed to diagnose MET or EG poisoning may not be immediately available, leading to potential delays in care. 6 Apart from high serum MET or EG concentrations, the indications for renal replacement therapy (RRT) in patients with MET or EG poisoning include acute kidney injury (AKI), severe metabolic acidosis, and a large anion gap.7,8 MET poisoning typically results in visual impairment and central nervous system damage, 8 whereas AKI is more typical in EG poisoning. 9 Without sufficient care, MET and EG poisoning result in significant morbidity and mortality.8–10
The primary aim of RRT in patients with MET or EG poisoning is to promptly remove the toxin and its injurious metabolites. The preferred RRT modality is intermittent hemodialysis (IHD). IHD is considered superior to continuous renal replacement therapy (CRRT) because of its more rapid toxin clearance, 8 whereas CRRT is usually preferred in hemodynamically unstable patients. 11 Even with sufficient treatment, MET and EG poisoning are associated with poor survival, often because of late presentation.9,12
In this observational retrospective cohort study, we compared the characteristics and outcomes of patients with MET or EG poisoning treated with RRT in the intensive care unit (ICU) of a tertiary academic medical center during a 10-year period.
Methods
This retrospective single-center cohort study included consecutive patients admitted to the ICU of Turku University Hospital (Turku, Finland) between 1 January 2010 and 31 September 2019 because of MET or EG poisoning requiring RRT. MET or EG poisoning was confirmed by a corresponding serum MET or EG concentration of at least 3 mmol/L on ICU admission. Patients younger than 18 years and those with a history of chronic severe liver disease (Child–Pugh A or worse) or chronic kidney disease (eGFR < 20 mL/minute or the need for maintenance dialysis) were excluded.
Blood pH; glucose, lactate, bicarbonate, and electrolyte levels; hemodynamics; and vasoactive medication were recorded on ICU admission and at 6-hour intervals for the first 24 hours after RRT initiation. Patient demographics, disease history, medications, and biochemical data were also extracted from the hospital medical records. Creatinine and eGFR levels were assessed on admission and at hospital discharge using the Chronic Kidney Disease Epidemiology Collaboration formula.
Laboratory assays
All blood pH, lactate, bicarbonate, glucose, and electrolyte samples were collected using lithium heparin syringes (Radiometer Medical ApS, Brønshøj, Denmark) from an arterial line within 5 minutes of venous blood sample collection, and analyses were conducted using the ABL90 point-of-care cartridge-based blood gas analysis system (Radiometer Medical ApS, Brønshøj, Denmark). The analysis of lactate levels was performed using an amperometric method based on lactate oxidase. Venous blood samples were analyzed in the central laboratory of Turku University Hospital (TYKSLAB).
CRRT and IHD
The selection of the RRT modality between CRRT and IHD was under the discretion of the attending ICU physician according to clinical care standards and patient hemodynamics. CRRT was chosen for hemodynamically compromised patients.
IHD was performed using Cordiax 5008 dialysis monitors (Fresenius, Bad Homburg, Germany) with a treatment duration of 5 to 10 hours, blood flow rates of 250 to 300 mL/minute, a dialysate flow rate of 500 mL/minute, and low-molecular-weight heparin (LMWH) anticoagulation, as appropriate, depending on the clinical condition of the patient.
CRRT was performed according to a standard protocol employed in our center using Multifiltrate CRRT monitors (Fresenius) and Ultraflux AV1000 or Ultraflux EMiC2 1.80 m2 polysulfone hemofilter membranes (Fresenius) with Ci-Ca® dialysate K2 (Fresenius) and 4% trisodium citrate to achieve regional citrate anticoagulation. Blood and dialysate flow rates were set according to the weight of the patient and by the caring ICU physician to target a dialysis dose of 30 mL/kg/hour.
Compliance with ethical standards
The study was conducted in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans. This retrospective register-based study used data from the hospital medical records to produce an anonymized dataset for analyses. Informed consent was not required because of the retrospective nature of the study. The study was approved by the ethics committee of South-West Finland Hospital District (ref T143/2016). All patient details were de-identified before conducting the analyses and reporting. The reporting of this study conforms to STROBE guidelines. 13
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request after receiving permission from the Ethics Review Board.
Statistical analysis
Continuous covariates were expressed as the mean and standard deviations for normally distributed variables and as the median and interquartile range (IQR) when the normality assumption was not met. Categorical covariates were reported with absolute and relative (percentage) frequencies. The Shapiro–Wilk test (P > 0.05) was used to assess normality assumptions for continuous covariates.
Repeated-measures analysis of variance was used to compare data for pH, bicarbonate and lactate levels, the anion gap, the norepinephrine dose, and hourly urine output during the first 24 hours after RRT initiation between the study subgroups (MET poisoning vs. EG poisoning; 90-day survivors vs. non-survivors). The correlation/clustering between repeated measurements within a subject was considered using autoregressive or unstructured correlation structures based on Akaike and Bayesian information criterion examinations. Student’s t-test was used to compare continuous normally distributed covariates, and the chi-squared test or Fisher’s exact test was used to compare categorical covariates among the study subgroups. For skewed variables, groupwise comparisons were performed using a non-parametric Kruskal–Wallis test. Because of the limited number of deceased patients in the dataset, multivariable logistic regression models for 90-day mortality could not be constructed because of the increased risk for overfitting.
P < 0.05 (two-tailed) was considered statistically significant. The analyses were performed using JMP Pro 13.0 for Mac (SAS Institute Inc., Cary, NC, USA).
Results
Altogether, 28 patients with EG (n = 15 [54%]) or MET poisoning (n = 13 [46%]) treated with RRT in the ICU between 2010 and 2019 were included in the study. The mean age of the study population was 52 ± 13 years, the mean BMI was 25.7 ± 5.9 kg/m2, and five (17.9%) patients were women.
Twenty-three patients underwent IHD, and five patients underwent CRRT because of instable hemodynamics. The median time from ICU admission to RRT initiation was 2.0 (IQR = 0.5–10) hours (Table 1). Furthermore, 10 (36%) patients received an ethanol infusion (5 patients in each group), and 12 (43%) patients received fomepizole (6 patients in each group). One patient with MET poisoning received both an ethanol infusion and fomepizole, whereas six patients received no antidote. There was no difference in the use of antidotes between the EG and MET poisoning groups (P = 0.33).
Baseline measurements on intensive care unit admission.
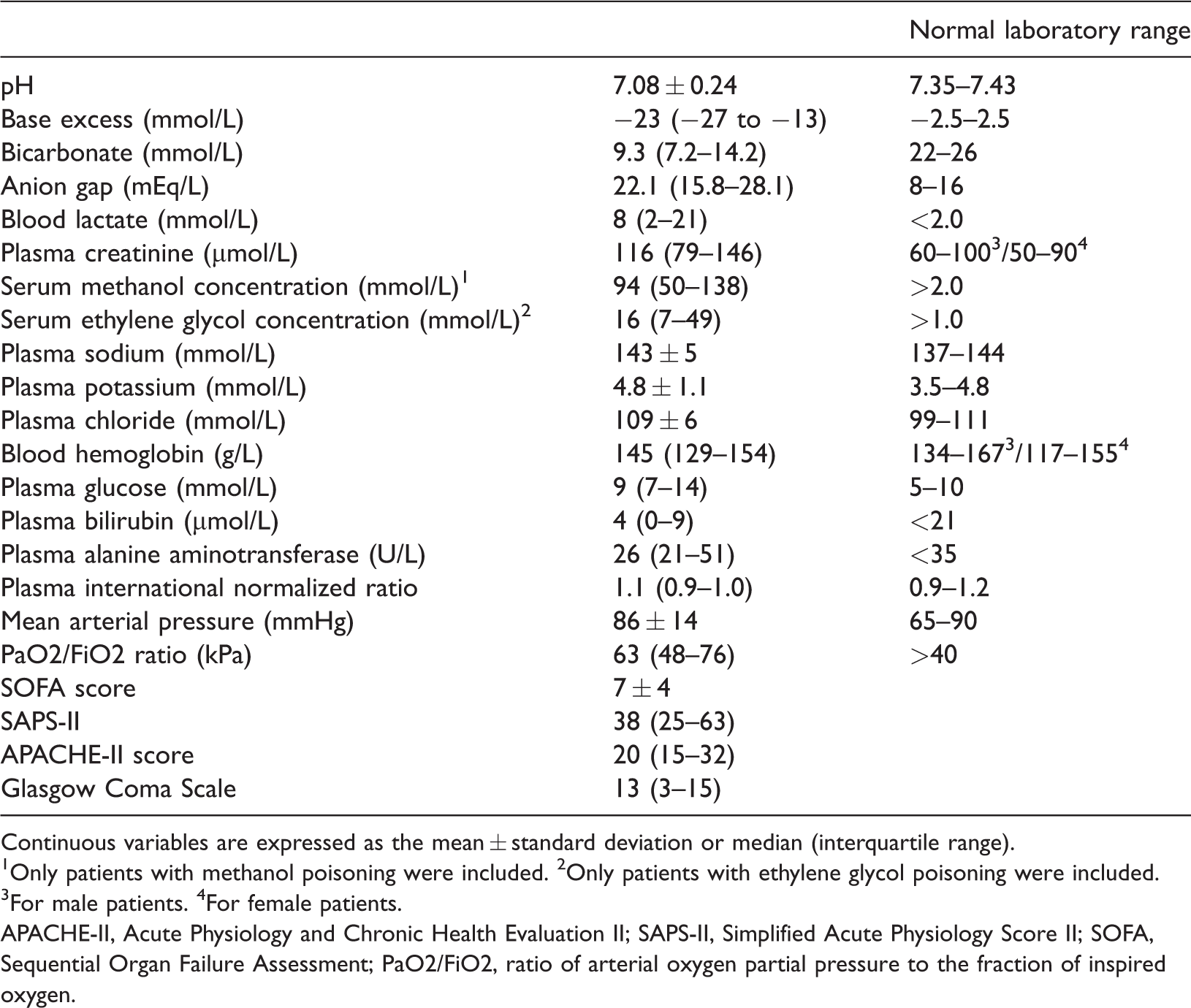
Continuous variables are expressed as the mean ± standard deviation or median (interquartile range).
1Only patients with methanol poisoning were included. 2Only patients with ethylene glycol poisoning were included.
3For male patients. 4For female patients.
APACHE-II, Acute Physiology and Chronic Health Evaluation II; SAPS-II, Simplified Acute Physiology Score II; SOFA, Sequential Organ Failure Assessment; PaO2/FiO2, ratio of arterial oxygen partial pressure to the fraction of inspired oxygen.
The median duration of ICU stay was 43 (IQR = 22–60) hours, 18 (62%) patients required mechanical ventilation, and eight (29%) patients required vasopressor support. The median duration of RRT was 15 (IQR = 10–26) hours. The ICU-related, in-hospital, 90-day, and 365-day mortality rates were 18% (5 patients), 25% (5 patients), 32% (9 patients), and 36% (10 patients), respectively (Table 2).
Treatment specifics and patient outcomes in the study.

Continuous variables are expressed as the mean ± standard deviation or median (interquartile range).
SOFA, Sequential Organ Failure Assessment; PaO2/FiO2, ratio of arterial oxygen partial pressure to the fraction of inspired oxygen.
Patients with EG poisoning were older (57.0 ± 13.2 years vs. 44.9 ± 11.6 years; P = 0.03) and they had higher blood lactate levels (21.0 mmol/L [IQR = 5.4–28.0] vs. 4.5 mmol/L [IQR = 2.1–8.5]; P = 0.003) on admission than those with MET poisoning (Table 3), as well as higher lactate levels (P = 0.01) and lower hourly urine output (P = 0.003) during the first 24 hours after RRT initiation (Figures 1 and 4).
Univariate comparisons between the study subgroups.

Values are presented as the mean ± standard deviation or median (interquartile range).
ALAT, plasma alanine aminotransferase; APACHE-II, Acute Physiology And Chronic Health Evaluation II; BMI, body mass index; EG, ethylene glycol; ICU, intensive care unit; INR, international normalized ratio; MAP, mean arterial pressure; MET, methanol; MV, mechanical ventilation; NE, norepinephrine; RRT, renal replacement therapy; SOFA, Sequential Organ Failure Assessment; SAPS-II, Simplified Acute Physiology Score II; PaO2/FiO2, ratio of arterial oxygen partial pressure to the fraction of inspired oxygen.

Hourly urine output (mL/hour) measured every 6 hours during the first 24 hours after the initiation of renal replacement therapy in patients with methanol and ethylene glycol poisoning (panel A) and in survivors and non-survivors (panel B).
Six (46%) patients with MET poisoning and three (20%) patients with EG poisoning died within 90 days of ICU admission, and there was no difference in the mortality risk between the study groups (P = 0.23). Patients who died within 90 days had higher rates of mechanical ventilation (89% vs. 47%; P = 0.04), a larger anion gap (30 ± 9 vs. 21 ± 8; P = 0.02), and lower pH (6.9 ± 0.2 vs. 7.1 ± 0.2; P = 0.03), bicarbonate (7.5 mmol/L [IQR = 5.6–9.0] vs. 13.2 mmol/L [IQR = 7.6–19.3]; P = 0.01), and base excess (−15 [IQR = −26 to −5] vs. −27 [IQR = −31 to −23]; P = 0.01) at baseline than survivors (Table 3).
Lactate levels and urine output during the first 24 hours after RRT initiation were not different between patients who died within 90 days and survivors (Figures 1 and 4). Moreover, there were no statistically significant differences in pH (Figure 2), norepinephrine requirements, the anion gap (Figure 3), or bicarbonate levels during the first 24 hours after RRT initiation between the study groups or between survivors and non-survivors. Toxin concentrations on admission were not associated with 90-day mortality in patients with EG (16 mmol/L [IQR = 7–49]; P = 0.26) or MET poisoning (94 mmol/L [IQR = 50–148]; P = 0.13; Table 1).

Blood pH measured every 6 hours during the first 24 hours after the initiation of renal replacement therapy in patients with methanol and ethylene glycol poisoning (panel A) and in survivors and non-survivors (panel B).

The anion gap (mmol/L) measured every 6 hours during the first 24 hours after the initiation of renal replacement therapy in patients with methanol and ethylene glycol poisoning (panel A) and in survivors and non-survivors (panel B).

Blood lactate levels (mmol/L) measured every 6 hours during the first 24 hours after the initiation of renal replacement therapy in patients with methanol and ethylene glycol poisoning (panel A) and in survivors and non-survivors (panel B).
Reduced LMWH anticoagulation was used in six (46%) patients with MET poisoning for clinical reasons. However, in most patients with MET poisoning managed with IHD, circuit anticoagulation was not applied as per current treatment guidelines because of case reports of intracranial hemorrhage.
Discussion
The present study revealed that patients with EG poisoning requiring RRT may have lower hourly urine output on ICU admission and within 24 hours after RRT initiation than patients with MET poisoning. Mortality was similarly high in the two groups. Patients who died within 90 days had a larger anion gap and lower pH, bicarbonate levels, base excess and Glasgow Coma Scale (GCS) at baseline than survivors. To our knowledge, this is the first study to compare outcomes between patients with EG and MET poisoning managed with RRT in the ICU using extensive repeated-measures clinical and biochemical data during the first 24 hours after RRT initiation.
Fomepizole and ethanol can be used as antidotes in the treatment of EG or MET poisoning because both agents inhibit the function of ADH and thereby decrease the production of toxic metabolites. 7 However, IHD has been found to be the most effective RRT modality in the management of MET or EG poisoning, and CRRT is recommended only for hemodynamically unstable patients.11,14 Nevertheless, hemodialysis efficiently reduces the half-life of circulating MET, EG, and their metabolites regardless of the chosen modality. 8 In our study, 23 patients underwent IHD, and five patients underwent CRRT because of instable hemodynamics. Although circuit anticoagulation was used in a subset of patients in our cohort, in general, current guidelines recommend against the use of circuit anticoagulation when treating MET poisoning with hemodialysis because of case reports of intracranial hemorrhage. 8
It remains under debate whether an antidote such as fomepizole or ethanol infusion should be administered to patients with EG or MET poisoning who are undergoing RRT. Of the two possible antidotes, fomepizole has been considered the first-line choice because of its stronger inhibitory effects on ADH and easier administration and monitoring protocol. 15 However, the efficacy of the antidotes in managing EG or MET poisoning is limited by the formation of deleterious metabolites prior to antidote administration because fomepizole and ethanol can only inhibit ADH but not remove or cancel the effects of already formed metabolites. It has been reported that a large anion gap and severe acidosis in patients with EG or MET poisoning are associated with the delay between toxin ingestion and hospital admission.9,12 Accordingly, the patients in the present study had a large anion gap and severe metabolic acidosis on admission, suggesting a prolonged delay between ingestion and presentation. Thus, hemodialysis was required to remove both the toxin and formed metabolites and correct the metabolic acidosis.
The prompt initiation of care is essential for the management of EG or MET poisoning. However, delays in initiating care can be substantial for several reasons. First, ascertaining the toxic agent in patients presenting with poisoning can often be difficult, and this is particularly true for patients with altered levels of consciousness. Conversely, conscious patients may be hesitant to admit the ingestion of MET or EG and seek help because of embarrassment or self-destructive motivations. 2 Second, the availability of the blood gas chromatography–mass spectrometry instruments required for diagnosis varies between centers.15,16 Third, the interval between ingestion and presentation at the hospital has been found to average 24 to 48 hours.16–18 Fourth, many clinicians rely on the osmolar gap to screen for toxic alcohol poisoning, but the osmolar gap, though often helpful, is neither 100% sensitive nor 100% specific for detecting toxic alcohol poisoning. 16 For these reasons, it is often mandatory to initiate treatment based on the patient history and clinical condition before the detrimental toxin is identified. Moreover, the observed clinical condition, laboratory findings, and disease characteristics in patients with EG or MET poisoning were similar in prior studies. 19 When surrogate alcohol poisoning is suspected, the rapid initiation of RRT, despite not being a toxin-specific treatment, is the most effective strategy for removing both the toxin and the produced toxic metabolites and prevent end-organ damage. Furthermore, both EG and MET can cause permanent neurologic damage, and therefore, prompt toxin removal is necessary to minimize adverse central nervous system sequelae.8,20
The associations of a larger baseline anion gap with lower pH, bicarbonate levels, base excess, and GCS scores observed in the present cohort were in line with the results of previous observational studies of patients with severe MET or EG poisoning.9,19,21 Furthermore, high serum creatinine levels,6,22 high blood glucose levels,17,23 and hypothermia 22 at baseline have been demonstrated to be associated with mortality in patients with MET or EG poisoning in prior studies. Notably, baseline lactate levels appeared to be higher in the EG group. However, we measured blood lactate levels using an oxidase-based system, which has been reported to produce a falsely elevated lactate level in the presence of glycolic acid, which is structurally similar to lactate. Although true lactate levels could not be reported for patients with EG poisoning in our study, we believe that it is important to inform the healthcare community about this diagnostic issue with EG poisoning because point-of-care assays using oxidase-based methods for blood lactate measurements are widely in use in Western countries. 24
The limitations of this study included its retrospective design and relatively limited number of patients with EG or MET poisoning treated in our unit. The lactate dehydrogenase method, which is more specific for lactic acid and which is not affected by the presence of glycolic acid, would have been a more precise method for analyzing lactate content. Regrettably, this method was not available in our study. Thus, no direct conclusions can be drawn concerning the lactate levels in patients with EG poisoning because we used the oxidase-based analysis to measure blood lactate levels. However, the study included all critically ill patients with EG or MET poisoning requiring RRT during a 10-year period at our ICU. Furthermore, patients treated in the ICU of the research hospital are monitored continuously, and all vital parameters are recorded in the electronic patient database. Moreover, the study included comprehensive serial measurements of a vast array of hemodynamic and biochemical variables recorded on admission and every 6 hours during the first 24 hours after RRT initiation, enabling the use of repeated-measures analyses to examine differences between patients with MET or EG poisoning and between 90-day survivors and non-survivors.
To conclude, patient survival was poor and comparable between patients with severe MET or EG poisoning treated with RRT in the ICU, and severe metabolic acidosis, a larger anion gap, and altered mental status on admission were associated with mortality. Patients with EG may have lower urine output over the first 24 hours of RRT than patients with MET poisoning.
Footnotes
Acknowledgements
The authors are grateful to Mrs. Eveliina Loikas, RN for help with the data collection.
Declarations of conflicting interests
The authors declare no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Turku University Hospital Education and Research Foundation.
