Abstract
Objective
C-X-C motif chemokine ligand 5 (CXCL5), a member of the chemokine family, is associated with remodeling of connective tissues. However, its role in formation of intrauterine adhesions (IUA) remains unclear. We aimed to investigate the expression and mechanism underlying the role of CXCL5 in IUA.
Methods
Expression of CXCL5 in IUA was detected by immunohistochemistry in a rat model of IUA and by real-time PCR and western blotting in patients with IUA. The protein levels of matrix metalloproteinase 9 (MMP9) and transcription factor p65 in human endometrial cells were assessed by western blotting after CXCL5 overexpression.
Results
Protein expression of CXCL5 was significantly decreased in the endometria of IUA rats compared with that of control and sham-operated rats. Real-time PCR and western blotting in patients with IUA showed similar results to those from the rat model. After overexpression, CXCL5 significantly upregulated expression of MMP9 and slightly upregulated expression of p65 in human endometrial cells.
Conclusions
CXCL5 plays an important role in IUA formation after endometrial injury. We propose a molecular mechanism to explain formation of IUA, including downregulation of MMP9 by low CXCL5 expression. These findings provide valuable information for the prevention and targeted therapy of IUA.
Introduction
Intrauterine adhesion (IUA), also known as Asherman syndrome, is characterized by adhesions and fibrosis in endometrium within the uterus or cervix and partial or complete obliteration of the uterine cavity.1,2 IUA occurs most commonly after infection or trauma, such as a uterine surgery, especially during the postpartum period of a cesarean section, when estradiol levels are low. 3 IUA often leads to infertility, recurrent miscarriage, and various complications of pregnancy, such as placenta implantation, placental abruption, preterm premature rupture of membranes, and developmental abnormalities in fetuses.4–6 In recent years, IUA has become a common cause of female infertility in China, and its incidence has risen with the increase in frequency of uterine cavity surgery.7,8 Thus, the prevention and treatment of IUA are important in maintaining women’s health and meeting fertility requirements.
The basal layer in the human endometrium contains various progenitors and stem cells.9,10 Once the basal layer of the endometrium is severely damaged, the ability of the endometrium to regenerate is lost, usually leading to IUA.11,12 Importantly, a broad range of inflammatory chemokines, together with cytokines and cell adhesion molecules, are abnormally expressed during the development of IUA and help regulate the secretion and degradation of extracellular matrix components in endometrial fibrosis.12,13 Therefore, a better understanding the role of inflammatory chemokines in the process of adhesion formation might help to identify new therapeutic targets for IUA.
C-X-C motif chemokine ligand 5 (CXCL5), also known as epithelial neutrophil-activating peptide-78 (ENA-78), is a member of the CXC subfamily of chemokines and was first cloned in 1994 by Chang et al. 14 After binding with its receptor CXCR2, CXCL5 enables the recruitment of neutrophils and promotes angiogenesis and remodeling of connective tissues. 15 Recently, Konduru et al. reported that the knockdown of endogenous transcription factor p65 suppresses CXCL5 gene transcription in vivo, 16 indicating that CXCL5 expression is regulated by the nuclear factor (NF)-κB signaling. The mRNA and protein levels of p65 have been shown to significantly increase and be associated with the degree of adhesion in patients with IUA,17,18 suggesting that CXCL5 might be involved in the development of IUA mediated by the NF-κB pathway. Moreover, matrix metalloproteinase 9 (MMP9) plays an essential role in the degradation of extracellular matrix and fibrous tissue of patients with IUA. The low expression of MMP9 in patients with IUA results in excessive deposition of extracellular matrix, and the normal endometrium is replaced by fibrous connective tissue, ultimately leading to endometrial fibrosis and formation of adhesions in the endometrium.19,20 Although the CXCL5/CXCR2 axis is essential for cell adhesion and migration, followed by matrix remodeling by activating PI3K/AKT-induced upregulation of MMP9 in bladder cancer, 21 the role of CXCL5 in the process of the adhesion formation and fibrosis proliferation in IUA remains unclear.
In this study, we investigated the expression of CXCL5 in the pathogenesis of IUA by analyzing the CXCL5 expression in a IUA rat model and endometria of patients with IUA. To uncover the molecular mechanism underlying IUA formation, we used an in vitro model of Ishikawa cells to analyze the signal pathways involving CXCL5 and its potential role in IUA.
Methods
Ethics approval and consent to participate
The experimental design of genomic and expression studies was reviewed and approved by the Ethics Committee of the Anhui Medical University, China (No. 20160034) and complied with the Declaration of Helsinki. Each participant provided written informed consent to be included in the study. Animal experiments were reviewed and approved by the Ethics Committee on Animal Research of the Anhui Medical University (No. LLSC20180106).
Patients and tissue samples
Intrauterine adhesion and control tissues were collected from the Second Affiliated Hospital of Anhui Medical University between October 2017 and July 2018. In total, six IUA and six non-IUA endometrial tissues were collected from patients with IUA diagnosed by hysteroscopy and patients with secondary infertility who did not have IUA, respectively. We chose non-IUA tissues from patients with secondary infertility as controls instead of tissue from healthy individuals because of ethical concerns. Patients with secondary infertility undergo uterine examinations to confirm whether their infertility is caused by uterine disease, and some were confirmed as not having IUA and were able to provide endometrial tissue samples for this study. There was no significant difference in age, weight, or parity between patients in the IUA group and the control group.
Establishment of an IUA rat model
Adult female Sprague–Dawley rats aged 8 weeks (weight: ∼220 g) were obtained from the Experimental Animal Center of Anhui Medical University and used to create the IUA model using phenol mucilage according to the model description.17,22 All rats were raised in the laboratory for 1 week of adaptation. Vaginal smear was used to evaluate the phases of the estrous cycle, and all surgeries were performed when animals were in diestrus. Forty rats were randomly assigned to three groups: IUA group (20 rats for phenol mucilage treatment), sham-operated group (10 rats with vehicle treatment), and the control group (10 rats with no treatment). First, 5% pentobarbital sodium was injected intraperitoneally to anesthetize the rats. Then, intrauterine injections of 0.04 mL of phenol mucilage (liquefied phenol 25% [vol/vol]; Arabian gum 5% [wt/vol]; and glycerin 20% [vol/vol]) were administered to rats in the IUA group to cause endometrial injury. Rats in the sham-operated group were administered an equal volume of phosphate-buffered saline (PBS). The rats were housed with free access to food and water in an environment-controlled room at 22°C with a 12-h light/dark cycle. Two weeks after surgery, all rats were euthanized using CO2 at a flow rate of 10 L/min for 7 minutes, and endometrial tissue samples were collected for further analyses. This study was performed in strict accordance with recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (Bethesda, MD, USA).
Hematoxylin and eosin staining and Masson staining
Endometrial tissues underwent standard paraffin embedding and were cut into 5-µm-thick serial sections. 23 Hematoxylin and eosin (HE) staining and Masson staining were performed as previously described. For HE staining, the sections were incubated with hematoxylin solution for 5 minutes and then stained with eosin solution for 3 minutes. For Masson staining, after deparaffinization and rehydration, the sections were stained with hematoxylin for 5 to 10 minutes, and Masson staining mixture for 5 minutes; then, the sections were immersed in phosphomolybdic acid solution for 1 minute and aniline blue solution for 5 minutes and examined and photographed by microscopy.
Immunohistochemistry and scoring
Immunohistochemistry was performed according to the manufacturer’s instructions (Beijing Zhongshan Golden Bridge, Beijing, China). The sections were fully processed with 3% H2O2 for 15 minutes, washed with PBS buffer, and then blocked with 10% goat serum for 30 minutes at room temperature. Antibody against CXCL5 (ab198505, Abcam, Cambridge, UK) was incubated with the sections overnight at 4°C. After incubation with biotinylated secondary antibody and biotinylated horseradish peroxidase complex for 15 minutes, the sections were stained with diaminobenzidine for visualization.
The number of CXCL5-positive cells was counted in three randomly chosen fields using a semi-quantitative method by multiplying the number of positive cells by their positive intensity. 17 The proportion of positive cells was classified into five grades (0–4): 0%, 25%, 50%, 75%, and 100%. The intensity of the stain was scored from 0 to 3 points: 0 = no staining, 1 = weak, 2 = moderate, 3 = strong staining. The score was calculated by multiplying the proportion and intensity score (total score range: 0–12). The samples were observed under a light microscope (at 40×, 100×, 200×, and 400× magnification).
RNA isolation and real-time PCR analysis
Total RNA from the endometrial tissues was isolated, as previously described. 24 RNA was extracted using RNAiso Plus reagent (TaKaRa Biotechnology (Dalian) Co. Ltd., Dalian, China). RNA samples (2 µg) were further reverse transcribed with the PrimeScriptP RT Master Mix (TaKaRa Biotechnology) according to the manufacturer’s instructions. The PCR amplification was performed by using SYBR Premix Ex Taq II (TaKaRa Biotechnology). The CXCL5 primer sequences were 5ʹ-CTCAAGCTGCTCCTTTCTCG-3ʹ and 5ʹ-GCGATCATTTTGGGGTTAAT-3ʹ. The GAPDH primer sequences were 5ʹ-GAAGGTGAAGGTCGGACTC-3ʹ and 5ʹ-GAAGATGGTGATGGGATTTC-3ʹ. All PCR reactions were run in a multiplex real-time PCR machine (StepOne, Applied Biosystems, Foster City, CA, USA). Quantification of GAPDH served as an internal control for normalization. Relative differences in mRNA levels over control values were calculated using the ΔCt (cycle threshold) method according to the manufacturer’s protocol (Applied Biosystems). The PCR reactions were independently repeated at least twice.
Cell culture and transfection
Ishikawa cells, a human endometrial epithelial cell line, were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were cultured in RPMI-1640 medium (Invitrogen/Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Invitrogen/Thermo Fisher Scientific) and antibiotics at 37°C and 5% CO2. The cells were transfected with CXCL5 expression vector (1 and 2 µg) and control plasmid pcDNA3.1 using jetPRIME transfection reagent (Polyplus Transfection SA, New York, NY, USA) according to the description of the protocol. After incubation with the transfection mix for 24 hours, the culture medium was replaced, and cells were harvested from the dishes following another 24 hours of incubation. Finally, the cells were assayed by western blotting.
Western blotting
Cells and tissue samples were lysed with radioimmunoprecipitation assay (RIPA) buffer containing a protease inhibitor cocktail (B14001, Bimake, Houston, TX, USA). Protein concentrations were measured by bicinchoninic acid assay. Equal amounts of protein mixtures (30 µg) were run on sodium dodecyl sulfate-PAGE, and the proteins were transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA) and incubated in PBS-Tween 20 with 5% nonfat milk for 1 hour at room temperature. The blot was incubated with primary antibodies, including anti-CXCL5 (1:100, no. ab198505, Abcam), anti-MMP9 (1:1000, no. 3852, Cell Signaling Technology, Danvers, MA, USA), anti-p65 (1:1000, no. 8242, Cell Signaling Technology) and anti-β-actin (1:20,000, no. KC-5A08, Shanghai Kangcheng, Shanghai, China) overnight at 4°C. Secondary antibodies conjugated to horseradish peroxidase were incubated with the blot for 1 hour at room temperature. The proteins revealed by western blotting were visualized by using enhanced chemiluminescence reagents (Servicebio, Wuhan, China). The densities of bands were analyzed by ImageJ software (National Institutes of Health, Bethesda, MD) and normalized to expression of GAPDH.
Statistical analysis
All data are expressed as means ± standard deviations and were analyzed using GraphPad Prism software, version 6.0 (GraphPad Inc., San Diego, CA, USA). Comparisons between groups were analyzed using Student’s t-test. For analysis of immunohistochemistry staining, a two-way ANOVA test was used. P < 0.05 was considered significant.
Results
Morphological changes of the endometria in the IUA rat model
Previous studies have shown a correlation between chemokines and degradation of the extracellular matrix and fibrosis.25,26 To investigate the potential role of CXCL5 in the pathogenesis of IUA, we first established a rat model of endometrium adhesion using phenol mucilage. All rat uteruses were resected 14 days after the operation, and gross morphology showed the occurrence of contractures in the uterus of rats in the IUA group compared with that in the control and sham-operated groups (Figure 1a–c). Micrographs in Figure 1d–f show that the endometrial cavity surface was covered by regularly arranged columnar epithelium with abundant endometrial glands in the control and sham-operated groups. Compared with the control and sham-operated rats, the uterine endometrial glands of IUA model rats were remarkably reduced, with tissue hyperplasia. Large amounts of macrophages and plasma cells were replaced by collagen fibers, indicating that inflammation is involved in the pathogenesis of IUA.

Abnormal morphology in intrauterine adhesion (IUA) rat model. (a–c) Representative images of isolated uteruses from three groups of rats; black arrowhead points to the intrauterine adhesion. (d–l) Forty rats were randomly separated into three groups: control (10 rats with no treatment), sham-operated (10 rats with vehicle treatment), and IUA group (20 rats with phenol mucilage treatment). One side of the uterus in each rat was treated with phenol mucilage or vehicle treatment. The uteruses from the three groups were collected for hematoxylin and eosin (HE) staining 14 days after treatment. Representative photomicrographs of uterus sections obtained from rats in the three groups at original magnifications of 40× (d–f, scale bar = 400 µm), 100× (g–i, scale bar = 200 µm), and 400× (j–l, scale bar = 50 µm). The uterine cavity was smaller in IUA rats than in rats of the other two groups.
Degree of endometrial fibrosis in the IUA rat model
To evaluate the degree of endometrial fibrosis, we performed Masson staining and observed numerous blue stromal collagen fibers tightly arranged in the endometrium in the IUA model group (Figure 2). Histological examination of the IUA uterus showed the presence of intrauterine adhesions and endometrial fibrosis. The HE and Masson staining results confirmed that the IUA rat model was successfully established.

Masson staining for uteruses in rats of three groups: control (n = 10), sham-operated group (n = 10), and intrauterine adhesion (IUA) group (n = 20). Selected representative images of uteruses in each group reveal interstitial fibrosis changes. Blue staining in IUA rats demonstrated more severe adhesions compared with control and sham-operated rats. Magnification: 40× (a–c, scale bar = 400 µm), 100× (d–f, scale bar = 200 µm), and 400× (g–i, scale bar = 50 µm).
Expression of CXCL5 in the IUA rat model
Previous studies17,21,27,28 indicate that abnormal expression of CXCL5 might play an important role in the occurrence of IUA. To this end, we examined CXCL5 expression in the endometrium of IUA rats and in sham-operated and healthy control rats by immunohistochemistry. The uteruses of control and sham-operated groups showed intense positive CXCL5 staining, which was mainly expressed in the cytoplasm of glandular epithelial and stromal cells (Figure 3). Quantification of staining indicated that expression of CXCL5 in the IUA group was significantly lower (P < 0.001) than that in the sham-operated and control groups, whereas there was no obvious difference between the control and sham-operated groups (Figure 3j). These results indicated that the low CXCL5 expression was related to IUA formation during the genesis and development of IUA.

Expression of C-X-C motif chemokine ligand 5 (CXCL5) in intrauterine adhesion (IUA) rat model detected by immunohistochemistry (IHC). The localization of CXCL5 (brown stained) was observed in three rat groups by light microscopy: control (n = 10), sham-operated group (n = 10), and intrauterine adhesion (IUA) group (n = 20). Magnification: 40× (a–c, scale bar = 400 µm), 200× (d–f, scale bar = 100 µm), and 400× (g–i, scale bar = 50 µm). (j) Summary of proportion and intensity of positive staining. The staining score for CXCL5 was significantly lower in the IUA group than in the control and sham-operated groups (***P < 0.001).
Reduced expression of CXCL5 in endometria of patients with IUA
To further confirm the low expression of CXCL5 in patients with IUA, we examined the expression of CXCL5 in endometrial tissues of six patients with IUA and six patients who did not have IUA (control group). The mRNA expression of CXCL5 in the endometrial samples (Figure 4a) was significantly downregulated (P < 0.01) in patients with IUA compared with in patients in the control group. Protein expression of CXCL5 in endometrial samples of patients with IUA was remarkably lower than that in healthy endometria (Figure 4b). Figure 4c shows a significant difference in CXCL5 expression (P < 0.01) in the two groups of patients. As with the IUA rats, reduced expression of CXCL5 in patients with IUA indicated that low expression of CXCL5 is essential in forming IUA.

Expression of C-X-C motif chemokine ligand 5 (CXCL5) in intrauterine adhesion (IUA) endometrium and the correlation between CXCL5, matrix metalloproteinase 9 (MMP9), and transcription factor p65. (a) mRNA expression of CXCL5 in six patients with normal endometria (Ctrl) and in six patients with IUA was measured by real-time PCR and normalized to that of GAPDH. (b) Protein expression of CXCL5 in endometrial tissues of the Ctrl and IUA groups was detected by western blotting and normalized to that of β-actin. (c) Quantitative analysis of CXCL5 protein expression in Ctrl and IUA groups. (d) Ishikawa cells were transfected with CXCL5 (1 and 2 µg) or vector, and expression of CXCL5, MMP9, and p65 was measured by western blotting, with β-actin used as an internal control. (e) Quantitative analysis of relative protein expression is shown. The error bars in a, c, and e represent the standard deviations of mRNA and protein expression. Significance of differences between the two groups was assessed by t-test: *P < 0.05, **P < 0.01.
CXCL5 positively regulates expression of MMP9 and p65 in endometrial cells
It has been reported that low expression of MMP9 occurs in IUA and osteosarcoma tissues, and the protein level of MMP9 can be regulated by CXCL5 through PI3K/AKT signaling.20,21 Recently, Xue et al. 18 demonstrated that expression of p65 is increased in IUA, and that CXCL5 and p65 positively promote the protein expression of each other. To confirm these findings, we performed western blotting to detect the protein levels of CXCL5, MMP9, and p65 in Ishikawa cells after transfecting the cells with a CXCL5 overexpression plasmid. We observed a significant increase in MMP9 expression (P < 0.01) and a slight increase in p65 expression (P < 0.05) with overexpression of CXCL5. Representative results are shown in Figure 4d, and the relative expression of proteins was calculated in Figure 4e, indicating that overexpressed CXCL5 upregulated expression of both MMP9 and p65 in human endometrial cells.
Discussion
IUA is the most common syndrome affecting fertility and pregnancy outcomes, and it usually occurs after infection or injury-related inflammation. 3 Until recently, hysteroscopy was widely considered the gold standard to diagnose and treat IUA. Various techniques for adhesiolysis and prevention of scar formation have been applied in the clinic. 9 Hysteroscopy with lysis of scar tissue can restore normal uterine function in many cases. However, this approach is not applicable in severe cases owing to the likelihood of recurrence after surgical repair. 29 Reported statistics show that postoperative recurrent rate is very high, and postoperative endometrial recovery and clinical cure rate are much worse in patients with severe IUA than in those with mild and moderate IUA.30,31 Therefore, the present clinical treatment of IUA primarily involves restoring the shape of the uterine cavity, but repair of endometrial function has poor efficacy, 9 driving the need for additional research into the molecular mechanism underlying the pathogenesis of IUA. Different from cells with active proliferative capacity (e.g., reproductive cells, stem cells, tumor cells), endometrial cells have achieved terminal differentiation and readily undergo apoptosis. Therefore, in vitro IUA models have rarely been used in previous IUA studies. Here, we applied in vivo studies to directly detect CXCL5 expression in IUA formation in an IUA rat model and in patients with IUA, and we used an in vitro model of Ishikawa cells to investigate the possible molecular mechanisms.
Collecting a large number of human endometrium samples is difficult because of ethical issues, in addition to the substantial experimental time and cost. Compared with clinical samples, the IUA rat model is more widely used for immunohistochemical analysis of endometrium in IUA5,32 because it can closely mimic the development and pathological characteristics of IUA in human patients and its experimental conditions are easy to control. To investigate the molecular mechanism underlying IUA genesis, we established an IUA rat model using the phenol mucilage method. Different from many other model animals, the rat has a duplex uterus with two distinct cervices, which is an important advantage for studying IUA. The pathological characteristics of rats with IUA closely mimic the human disease, and the cost of feeding rats is very low compared with feeding larger animals such as rabbits. However, the rat is still large enough to facilitate surgical procedures that would be more difficult in smaller animals such as mice. Therefore, rats are commonly used for establishing models for uterine diseases, particularly IUA.
Phenol mucilage is widely used to establish the inflammation-induced adhesion animal model in IUA17,33 and other inflammation-related disorders, such as peritoneal adhesion, 34 cervicitis, 22 and pelvic inflammatory disease, 35 because it is highly effective and reliable. Phenol mucilage contains three toxic ingredients: liquefied phenol, Arabic gum, and glycerin. Liquefied phenol is a chemical burn agent; it is highly corrosive and can destroy local tissues and cause an inflammatory reaction. Arabic gum and glycerin are thickeners that reduce the damage to adjacent tissues caused by diffusion of phenol. This chemical method is easier to operate and control than a mechanical injury method, and therefore is suitable for use in the reproducible inflammation-induced animal model of IUA. Although inflammation at the site of endometrial injury is widely considered the leading cause of IUA,1–3 other factors such as mechanical damage may lead to IUA and have been used to create animal models of IUA. 32 To this end, it is necessary to include a sham-operated group to exclude mechanical damage, including the effects of surgical procedures and injection process, in IUA model rats and to confirm that the pathological characteristics in IUA rats are caused only by the administered toxic agents. To simulate the endometrial injury of IUA rats caused by surgery and injection, we established a sham-operated group that used treatment with PBS, a non-toxic agent.32,36 Our IUA model not only helps in our understanding of the IUA phenotype and molecular mechanisms of adhesion formation but is also useful for identifying effective therapeutic strategies for IUA prevention and treatment. As mentioned above, we observed a reduction in uterine volume of the IUA rats, together with endometrial thinning and atrophy, a reduction in the number of glands, and significant proliferation of collagen fibers (Figures 1 and 2). These changes are consistent with the pathological manifestation of human IUA, confirming that our model is a qualified rat model of IUA.
The immunohistochemistry results showed that the protein level of CXCL5 in the IUA rats was significantly lower than that in the control and sham-operated rats (Figure 3). Additional measurements of mRNA and protein levels of CXCL5 in human patients confirmed the low expression of CXCL5 in the endometria of patients with IUA (Figure 4a, b). These results indicate that the low expression of CXCL5 induced IUA formation after endometrial injury.
Previous studies have shown that CXCL5 is involved in PI3K/AKT signaling to regulate the expression of matrix metalloproteinases (MMPs), which play critical roles in fibrogenesis and fibrosis. 21 After binding to its receptor CXCR2, CXCL5 induces increased phosphorylation of AKT and activates the PI3K/AKT pathway. MMP9 (a hydrolase) can degrade type IV and V collagens and is essential for the breakdown of extracellular matrix in normal physiological and disease processes. 19 Hu et al. 20 demonstrated low MMP9 expression in endometrial tissues of IUA and indicated that this low expression was closely related to IUA development. Gao et al. 21 demonstrated that low MMP9 expression in bladder cancer can be attributed to low CXCL5 expression or, if CXCL5 expression is high, to the inhibition of PI3K/AKT signaling. However, the low expression MMP9 in IUA cannot be caused by the inhibition of PI3K/AKT signaling, because of the low expression of CXCL5 in both IUA rat and patient endometria compared with non-IUA samples in this study. Therefore, we suggest that low CXCL5 expression leads to reduced expression of MMP9 in IUA. This, together with our second finding that enhanced CXCL5 expression in endometrial cells led to increased expression of MMP9, revealed a positive correlation between expression of CXCL5 and MMP9. Based on the above analyses, we suggest a molecular mechanism of CXCL5 in IUA as follows (Figure 5): low expression of CXCL5 induces reduced expression of MMP9 through PI3K/AKT signaling, leading to accumulation of extracellular matrix and proliferation of collagen fibers on the damaged endometrium; then, normal growth of endometrium is inhibited and finally intrauterine adhesion is formed.
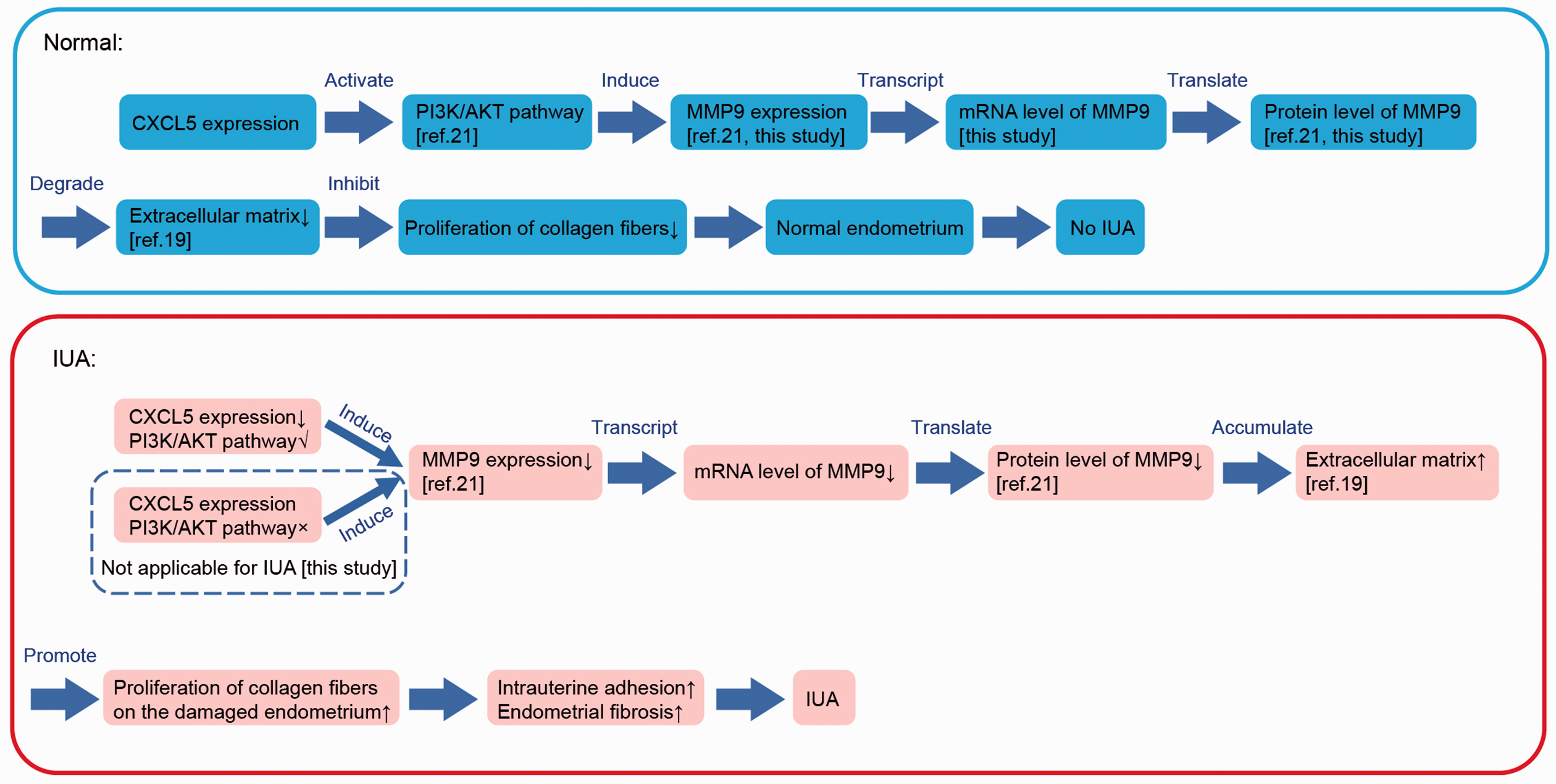
Schematic describing the molecular mechanism of C-X-C motif chemokine ligand 5 (CXCL5) in intrauterine adhesion (IUA) proposed in this study. In this proposed model, low expression of CXCL5 induces decreased expression of MMP9 (matrix metalloproteinase 9) through PI3K/AKT signaling, which leads to accumulation of extracellular matrix and proliferation of collagen fibers on the damaged endometrium; then normal growth of endometrium is inhibited, and finally intrauterine adhesion is formed.
CXCL5 can upregulate p65 expression and activate the NF-κB pathway, whereas p65 binds to the promoter of CXCL5 and induces its transcription in cancer cells.16,37 Recent evidence showed that the NF-κB pathway is associated with disorders of fibrogenesis and is upregulated in IUA genesis. 17 However, it remains unclear whether CXCL5 regulates p65 expression in the NF-κB pathway in IUA. Importantly, Xue et al. 18 found overexpression of p65 in IUA. To further investigate the correlation between p65 and low expression of CXCL5 in IUA found in this study, we detected expression of p65 in CXCL5-overexpressed endometrial cells. Surprisingly, we observed a slight increase in p65 protein level with the increase in CXCL5 expression in endometrial cells, indicating that p65 expression is not solely regulated by CXCL5 in IUA; its increased expression in IUA could be attributed to regulation by other factors. Otherwise, p65 would have a low expression in IUA, which is different from results reported in Xue et al. 18 Thus, we speculate that the NF-κB signaling is not the principal molecular mechanism underlying IUA formation involving CXCL5.
Conclusions
CXCL5 showed significantly decreased expression in IUA endometrial tissue in both rats and humans, and CXCL5 positively regulated MMP9 expression, indicating that CXCL5 plays key roles in IUA formation after endometrial injury and therefore is a potential biomarker of IUA. Based on our findings, we proposed a molecular mechanism for IUA formation, which includes downregulation of MMP9 by the low expression of CXCL5. These results open up the possibility of using CXCL5 in the prevention and targeted therapy of IUA.
Footnotes
Acknowledgments
The authors express their sincere thanks to all participants in this study and to Wenyan Wang, Yu Zhang, Enxue Song and Yijun Fan from the Department of Gynecology and Obstetrics, The Second Affiliated Hospital of Anhui Medical University (Hefei, China) for their technical assistance.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by National Natural Science Foundation of China (nos. 81801511 and 81802586). The funders had no role in the design of the study, in the collection, analysis, and interpretation of the data, or in the writing or approval of the manuscript.
Author contributions
SS proposed and supervised the project, designed the study, and analyzed data; ZF performed experiments under the supervision of SS and BW; YH and CS helped with sample collection and western blotting; LZ and GZ helped with analysis and interpretation of data; and SS and ZF wrote the manuscript. All authors read and approved the final manuscript.
