Abstract
Objective
This study compared the continuity equation-based effective orifice area (EOA) of prosthetic mitral valves between two-dimensional (2D) and 3D transesophageal echocardiography (TEE).
Methods
Thirty-four patients without major aortic valve abnormalities underwent mitral valve replacement surgery. The EOAs of prosthetic mitral valves were calculated using the continuity equation with 2D and 3D TEE. For 18/34 patients using a biological valve prosthesis, the EOA of the prosthesis was obtained from commercial records.
Results
The EOA of prosthetic mitral valves significantly varied between the 2D and 3D methods (2.22 ± 0.71 vs 2.35 ± 0.70 cm2, n = 34). The area of the diameter of the left ventricular outflow tract as determined by the 3D method was significantly higher than that by the 2D method (mean difference: −0.14 ± 0.20 cm2), with 95% coherence boundaries of −0.53 and 0.25 cm2. The regression equation for the EOA by 3D and 2D TEE was y = 0.27 + 0.94x, with a good correlation.
Conclusions
The EOA of prosthetic mitral valves is underestimated using the 2D TEE method compared with the 3D TEE method. The 3D-TEE method has the advantage of higher precision over the 2D TEE method, and it may be helpful for better assessment of prosthetic mitral valves intraoperatively.
Keywords
Introduction
Estimation of the effective orifice area (EOA) of a prosthesis is challenging, especially when the prosthesis is damaged. The continuity equation is frequently used to assist such judgment. In the clinical situation and experimental studies, calculations of the continuity equation valve area have been conclusively validated.1,2 Furthermore, calculating the continuity equation valve area is considered to be a credible parameter when making predictions of clinical outcome and clinical decisions.
According to the principle of continuous equations, in the absence of valvular reflux and intra-cardiac shunts, the blood flow rate passing through the normal aortic valve and that through the mitral valve should be equal. 2 Measurement of the diameter of the left ventricular outflow tract (LVOT) needs to be accurate because it is essential for calculation of the continuity equation. This equation is calculated according to the assumption that the shape of the LVOT is circular. Therefore, a single diameter can be used to obtain a precise assessment of the LVOT area. However, the LVOT is currently recognized as not circular, but elliptical, with major and minor axes in a large percentage of patients.3–5 The cross-sectional area of the left ventricular outflow tract (CSALVOT) originating from a two-dimensional (2D)-based diameter of the LVOT may result in different outcomes of the EOA of a prosthetic mitral valve with calculation of the continuity equation compared with the 3D-planimetered LVOT area with real-time 3D transesophageal echocardiography (TEE).
We conducted this study to measure and compare the EOA of prosthetic mitral valves between two techniques. This area was calculated with the CSALVOT originating from a 2D-based diameter of the LVOT and with a 3D-planimetered LVOT area using real-time 3D TEE in patients undergoing a mitral valve replacement operation. This study aimed to determine a more accurate and reliable method for determining the EOA of a prosthesis.
Materials and methods
The study was part of a current protocol for collection of intraoperative echocardiographic data. This study was approved by the ethics committee of Nanjing First Hospital, Nanjing Medical University (approval number: KY20170811-03). All patients were informed that they would participate in the study and provided written consent before the operation. They were also informed before the operation that they would receive a TEE examination, and informed of the relevant risks and precautions. We studied 2D and 3D data that were routinely collected from patients who underwent elective mitral valve replacement surgery in Nanjing First Hospital, Nanjing Medical University.
We collected echocardiographic data from patients who received mitral valve replacement surgery with intraoperative 3D TEE during March 2019 to August 2019. We excluded patients who had undergone emergency procedures, those with previous valvular heart surgery, those who had more than moderate regurgitation of the aortic valve, those with moderate regurgitation of the mitral valve after surgery, those with combined procedures (e.g., ascending aortic surgery or aortic valve replacement), and those with suboptimal 3D images from this study. Two more patients were excluded because of suboptimal 3D images.
All operations were performed by two senior attending physicians with more than 5 years of experience in TEE examinations and a TEE advanced qualification certification. The examination methods and programs were consistent. The 3D images were transferred to a computer from the ultrasonic equipment after the operation and an investigator analyzed acquired 3D data. The intraoperative values were blinded for the investigator. For the archived 3D data, the investigator measured the results again 1 week later, and the results were averaged with the first measurement.
Using a Philips CX50 ultrasound system (Philips Healthcare, Andover, MA, USA) and its X7-2t probe, we conducted intraoperative TEE examinations after closing the chest and before transferring the patient. According to the European and American Echocardiographic Societies Recommendations for Image Acquisition and Display Using Three-dimensional Echocardiography, we selected the 2D mode at a frame rate above 45 HZ and the 3D mode at a frame rate of 15 to 25 HZ. Comprehensive 2D exams were performed according to these guidelines. Stroke volume (SV) was calculated as CSALVOT × velocity time integral (VTI) through the LVOT (VTILVOT). CSALVOT was calculated using 2D measurement results and data derived from 3D images when using the 3D method. The EOA of the prosthetic mitral valve was calculated with the continuity equation as follows: EOA = SV/VTIpmv, where VTIpmv is velocity–time through the mitral valve prosthesis. Therefore, EOA = CSALVOT ×VTILVOT/VTIpmv. For calculation of the EOA, we obtained VTIpmv, as measured by continuous-wave Doppler below the mitral valve prosthesis. We also collected the VTILVOT using pulse-wave Doppler in the deep transgastric window (Figure 1). The same values of VTILVOT and VTIpmv were used in the 2D and 3D methods.

The + LVOT VTI in the deep transgastric window was obtained. SV was calculated as + LVOT VTI×CSALVOT.
In the 2D method, from the mid-esophageal position, we obtained LVOT images in the long-axis view (120°–150°). Using the zoom function, we measured the LVOT diameter approximately 5 mm from insertion of the aortic leaflets in the mid-systolic phase (Figure 2). The CSALVOT was calculated as π×(LVOT diameter/2)2 and was automatically derived by the instrument’s software. In the 3D method, measurement and 2D TEE were performed simultaneously with the same system. We acquired 3D data of the LVOT and aortic valve in the center of the screen. When we aligned the multi-planar reformatting planes, including the sagittal, coronal, and transverse planes, in the mid-systolic position (Figure 3), we displayed three geometrically orthogonal views of the LVOT and the aortic valve. To achieve the highest spatial and temporal resolution, we avoided absence of motion or electrical interference.

The LVOT diameter was obtained from LVOT images in the long-axis view. In this case, we took measurements three times with +LVOT Diam, ×LVOT Diam, and ▪ LVOT Diam, and used the average value of these results.

Alignment of orthogonal multi-planar reformatting planes to each other. An accurate en face view of the left ventricular outflow tract was obtained from three-dimensional images. An elliptical shape of the cross-sectional area through the left ventricular outflow tract was displayed.
After surgery, the 3D data were transferred to a computer with Q-Lab Version 8.1.2 Advanced Ultrasound Quantification Software (Philips Healthcare). Using this software, an investigator performed geometrical reestablishment and analyzed the LVOT. The shape of the 3D-planimetered LVOT area (CSAplanimetered) was traced and the result was obtained from the software mentioned above. SV was calculated as CSAplanimetered × VTILVOT.
The EOAs of 18 patients using a biological valve prosthesis were obtained from commercial records (R-EOAs). We did not find EOA data from commercial records for 16 patients with mechanical valves. The indexed EOA was defined as the EOA of a prosthetic mitral valve divided by the patients’ body surface area. We defined prosthesis–patient mismatch (PPM) as an indexed EOA ≤1.20 cm2/m2, with severe PPM defined as an indexed EOA <0.9 cm2/m2. 6
Continuous data are expressed as mean ± standard deviation. Categorical data are expressed as the number and percentage. For comparison of two continuous variables, the two-sided paired t test or Mann–Whitney U test was used, with or without a normal distribution. The relationship of EOAs was assessed by applying Pearson’s correlation coefficient analysis and simple regression analysis. We also used Bland–Altman analysis to define the biases and limits of agreement between the two methods. All of the data were analyzed using IBM SPSS version 20.0 (IBM Corp, Armonk, NY, USA).
Results
We examined 34 patients and their baseline clinicopathological characteristics are shown in Table 1. The mean age was 57.26 ± 9.87 years, and 34% (n = 12) were men and 66% (n = 22) were women.
Characteristics of the patients (n = 34).

Values are mean ± standard deviation or n (%).
The mean EOA of the prosthetic mitral valve as estimated by the 3D method was significantly larger than that estimated by the 2D method (2.35 ± 0.70 vs 2.22 ± 0.71 cm2, P < 0.001). The CSALVOT as estimated by the 3D method was also significantly larger than that obtained by the 2D method (3.12 ± 0.83 vs 2.95 ± 0.87 cm2, P < 0.001). SV as estimated by the 3D method was significantly higher than that estimated by the 2D method (52.39 ± 3.42 vs 49.34 ± 3.36 cm2, P < 0.01).
Bland–Altman analysis (Figure 4) was used to test the coherence of the EOA calculated by the two techniques. We found good agreement of the EOA calculated by the two methods, which showed a fixed bias (mean difference was 0.14 ± 0.20 cm2, P < 0.001), and the 95% coherence boundaries were −0.53 and 0.25 cm2. We also found a good correlation of the EOA of the mitral valve between the 2D and 3D methods (r = 0.94, P < 0.001) (Figure 5). The observed mean difference in the EOA between the 2D method and R-EOA was 0.37 cm2 (2.22 ± 0.71 vs 2.59 ± 0.37 cm2). The observed mean difference in the EOA between the 3D method and R-EOA was 0.24 cm2 (2.35 ± 0.70 vs 2.59 ± 0.37 cm2). These mean differences between the 2D and 3D methods and R-EOA were not significant.
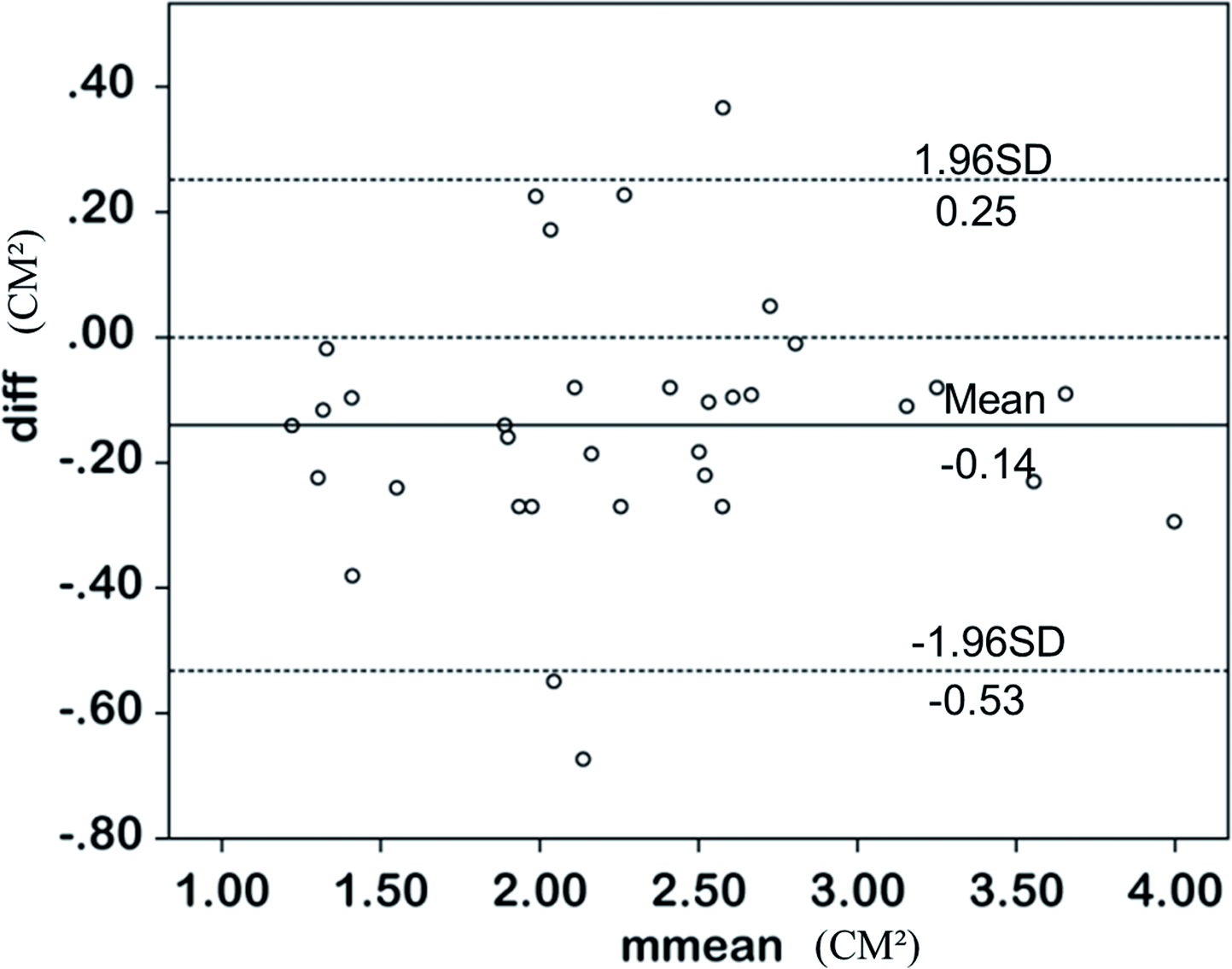
Comparison of the effective orifice areas of the two methods with Bland–Altman analysis.

Correlation of the EOA of the mitral value between 2D and 3D measurements (R2 = 0.919, P < 0.001).
In the 2D method, mitral PPM was found in 32.3% (11/34) of the patients. Four of the 11 patients with mitral PPM had severe PPM. Using the 3D method, mitral PPM was found in 26.4% (9/34) of patients. Three of nine patients with mitral PPM had severe PPM. The probability of occurrence of mitral PPM was significantly higher using the 2D method than the 3D method (P < 0.05).
Discussion
The current study suggested that use of the continuity equation to calculate the EOA of prosthetic mitral valves may have been affected by estimation of the CSALVOT with 2D echocardiography. The 2D method is based on the diameter of a single axis. However, the LVOT is not circular, but elliptical instead, with major and minor axes in a large percentage of patients.3–5 Therefore, the CSALVOT with 2D echocardiography may be either under- or overestimated depending on which single diameter (i.e., minor or major or axis 7 ) is used. With calculation of the continuity equation, a difference in SV leads to a difference in calculation of the EOA of the mitral valve.8,9
In our study, calculation using the 2D-based CSALVOT of SV led to underestimation of SV by approximately 6% compared with the 3D-planimetered LVOT area. This finding is in agreement with previous reports on the effect of the elliptical shape of the LVOT by observation and calculation of the aortic valve area.10–14 Our study showed a slightly smaller underestimation of SV than that reported previously (10%). 15 The reason for this difference between studies may be due to differences in different types of surgery and different time points of observation. Two-dimensional-derived calculations may have limitations compared with 3D imaging.16,17 In the mid-esophageal long-axis view of 2D, there may be a good opportunity to visualize the minor axis of the LVOT. Therefore, the area may be underestimated using this measurement. However, in the 3D method, acquisition of an en face view of the LVOT might be more precise because it may exclude the assumption of circularity. 18
We recorded the R-EOA of 18 patients using bioprosthetic valves from commercial records. We found that the difference between EOA values measured by the 3D method and R-EOA was smaller than that between EOA values measured by the 2D method and R-EOA. Bioprosthetic valves are biologically closer to human valves compared with mechanical valves, and the valid valve area recorded by commercial products is easier to obtain. However, after implantation of a valve, the EOA of the value is also affected by other factors. The R-EOA can only be used as a reference. 9
The shape of the LVOT may be slightly distorted after mitral valve replacement because of the presence of a prosthesis. Additionally, continuing to use the 2D method to measure the CSALVOT after implantation of a prosthesis may lead to inaccurate results. However, when the CSALVOT is measured by the 3D method, the resulting error caused by distortion can be corrected because the shape can be adjusted by tracing according to the actual situation. This situation also requires attention especially after mitral valve replacement surgery.
Mitral PPM may adversely affect long-term survival of patients with regurgitation of the mitral valve. 18 We found that the incidence rate of mitral PPM using the 3D method was lower than that using the 2D method. Therefore, the 3D method may be useful in early identification of PPM perioperatively.
A limitation of our study is that a relatively small number of patients were included. A further study on continuity equation-based estimation of the EOA of prosthetic mitral valves using 2D and 3D TEE with a larger population is required in the future. Additionally, SV as measured by pulmonary artery catheterization may need to be added for providing vital information in future studies. We performed analysis of data using software in a computer and routine analysis of 3D data of geometrically orthogonal views (sagittal, coronal, and transverse planes). However, because of resource constraints, this analysis cannot be achieved on a routine basis. Calcification of the elliptical shape of the LVOT also needs to be further investigated. However, the possibility that the LVOT area as measured with the 2D method may be more reliable in certain situations cannot be excluded.19,20 Additionally, because of the low frame rate in early studies of 3D ultrasound, frame loss may have severely affected the effectiveness of 3D results. However, in our study, the 3D mode at a frame rate of 15 to 25 HZ may have overcome this weakness of frame loss. The 3D mode also has a limitation that if 3D data in multiple cardiac cycles need to be collected, the present frame rate is insufficient.
Conclusions
When the continuity equation is used, the EOA of a prosthetic mitral valve may be underestimated using the 2D TEE method based on the diameter of a single axis compared with the 3D-planimetered area. The advantage of the 3D method versus the 2D method is higher precision, which leads to better assessment of a prosthetic mitral valve intraoperatively.
