Abstract
We report two suicidal cases of acute methyl ethyl ketone peroxide (MEKP) poisoning. A woman in her late 60s suffered from oral mucosal erosion, functional impairment of the heart, liver and other organs, pulmonary inflammation, elevated inflammatory markers, pleural effusion, hypoproteinemia and metabolic acidosis after oral administration of approximately 50 mL of MEKP. After admission, the patient was administered hemoperfusion four times, 8 mg of betamethasone for 6 days and symptomatic support. Hemoperfusion had an obvious effect on the treatment of oral MEKP poisoning. After discharge, the patient developed progressive dysphagia and secondary esophageal stenosis. Supplementary feeding was administered with a gastrostomy tube after the patient was completely unable to eat. A man in his mid-40s developed oropharyngeal mucosal erosion, bronchitis and esophageal wall thickening after oral administration of 40 ml MEKP. After receiving total gastrointestinal dispersal, 80 mg of methylprednisolone was administered for 7 days, and symptomatic supportive treatment was provided. Slight dysphagia was observed after discharge, and there was no major effect on the quality of life. Patients with acute oral MEKP poisoning should be followed up regularly to observe its long-term effects on digestive tract corrosion and stenosis.
Keywords
Introduction
Methyl ethyl ketone peroxide (MEKP) is widely used for industrial applications. 1 MEKP is used as a curing agent of unsaturated polyester resin at room temperature, initiator of organic synthesis, bleaching agent, fungicide, catalyst, and hardener. In recent years, the use of MEKP has increased yearly with the MEKP industry continuing to develop. The toxic effects of occupational and non-occupational exposure of MEKP are of increasing concern. Occupational poisoning is mainly chronic inhalation, skin or mucous membrane contact and explosion. Non-occupational exposure is mainly acute poisoning caused by accidental ingestion or suicide. MEKP is listed as a highly toxic substance and is categorized as United Nations Hazard class 5.2. MEKP is an unstable organic peroxide with strong oxidizing and corrosive properties. MEKP ingestion results in proximal airway edema and obstruction, corrosive burns leading to stricture formation, inhalational pneumonitis or acute respiratory distress syndrome, acidosis, liver failure and renal failure.2,3 Respiratory complications of MEKP are frequently (38%) observed and are also a direct cause for documented fatality. 4 The National Institute for Occupational Safety and Health and the American Conference of Governmental Industrial Hygienists set a recommended upper limit of 1.5 mg/m3 (0.2 PPM) for MEKP. 5 In cases in which the dose is known, toxicity occurs after ingestion of 50 to 100 mL in adults. 6 Studies have postulated that the mechanism of toxicity of MEKP is free radical formation, which leads to lipid peroxidation, corrosive damage to the mucosa of the respiratory and digestive tract, and liver damage. 2 MEKP is a colorless liquid, which cannot be distinguished from drinking water in appearance, and is easy to be taken by mistake in daily life. 7 In the process of production and living, safety marking should be present, and MEKP should be placed in a safe place to avoid poisoning caused by improper contact. Although suicidal poisoning from oral MEKP is rare, we report two suicidal cases of MEKP poisoning.
Case report
Case 1
A woman in her late 60s was transferred to our hospital from a local hospital on 9 November 2020 because of oral administration of MEKP for more than 7 hours. The patient took approximately 50 mL MEKP orally after drinking alcohol. She immediately developed nausea and vomiting. She then fell into coma without limb convulsions, accompanied by foaming at the mouth. An hour and a half later, she was sent to the local hospital for gastric lavage, catharsis, fluid replenishment, protection of the gastric mucosa, nutrition of brain cells, and symptomatic and supportive treatment. The patient then developed respiratory distress and was provided endotracheal intubation, ventilator-assisted breathing and blood purification treatment. For further treatment, she was transferred to our hospital. The patient had a history of hyperglycemia without systematic diagnosis or treatment.
On admission, the patient was confused, and a physical examination was uninformative. Endotracheal intubation with ventilator-assisted breathing was performed. The patient’s vital signs were normal, with a body temperature of 36.7°C, respiratory rate of 19 breaths/minute, blood pressure of 127/74 mmHg, blood oxygen saturation of 99% and heart rate of 97 beats/minute. The patient had swollen lips, congestion and erosion in the mouth. No other positive signs were found. Arterial blood gas analysis showed metabolic acidosis. A blood examination on admission showed an elevated white blood cell count, heart and liver dysfunction and an elevated D-dimer concentration. The patient’s family brought MEKP to the hospital to be tested and it showed a pH of 5. The patient’s diagnosis on admission was acute oral MEKP poisoning.
After admission, the patient continued to be provided ventilator-assisted respiration (synchronized intermittent mandatory ventilation mode, tidal volume: 450 mL, respiratory rate: 16 breaths/minute, initial inspiratory positive airway pressure: 12 cmH2O, positive end-expiratory pressure: 6 cmH2O, oxygen concentration: 100%). Dark green urine (Figure 1) appeared at the early stage of poisoning, which was caused by MEKP metabolism in the body. With the consent of the patient and her family, hemoperfusion, a low-dose hormone, liver protection (e.g., promoting the repair and regeneration of liver cells, improving the activity of enzymes in the liver and stabilizing bile), pantoprazole 40 mg by intravenous drip twice a day to protect the gastric mucosa, fluid replenishment, a diuretic, a laxative, nutritional support, anti-infection, sedation and other comprehensive treatments were administered. Hemoperfusion was provided twice on the first day after admission, 2 hours each time, and once for 12 hours. Arterial blood gas analysis showed that the partial pressure of oxygen was 233 cmH2O, and the oxygen concentration on the ventilator was adjusted to 60%.

(a) Photograph showing dark green urine from a urine bag brought from another hospital when the patient was admitted. (b) Photograph showing dark green urine in the patient’s urine bag on the first day of admission. (c) On day 3 of admission, the patient’s urine had become lighter in color.
On the second day after the admission, hemoperfusion was performed once for 2 hours. Reexamination of venous blood showed improved heart and liver function, hypoproteinemia and normal arterial blood gases. The patient received hemoperfusion once for 2 hours on the third day, the respiratory rate of the ventilator was adjusted for 10 times/minute and the oxygen concentration was 40%. The sedative was stopped on the fourth day, and the patient was conscious. The breathing rate of the ventilator was adjusted to six times/minute with a positive end-expiratory pressure of 4 cmH2O on the fifth day. A blood reexamination on the seventh day showed high inflammatory markers and hypoproteinemia, but restored organ function. Ventilator-assisted respiration was discontinued. Tracheal intubation was removed on the seventh day. The deep vein catheterization was removed on the eighth day. A plain computed tomography (CT) scan of the brain, chest and abdomen on the ninth day showed suspicious low-density lesions on the lateral frontal angle of the left lateral ventricle and micronodules of the right lung, bilateral pulmonary fiber foci and inflammation. Additionally, CT showed a slightly thickened tracheobronchial wall, left pleural effusion, a slightly thick pericardium, a thickened esophagus, a slightly thick gastric wall, intestinal gas, and part of the intestinal border was unclear. Inflammation of the gallbladder and cholestasis were also observed (Figure 2). On the 11th day, the patient was generally in a good condition, lucid and mentally sound. A physical examination showed coarse respiratory sounds in both lungs, and no obvious abnormalities were found in other areas of the patient’s body. Her condition had improved and she was discharged from the hospital.
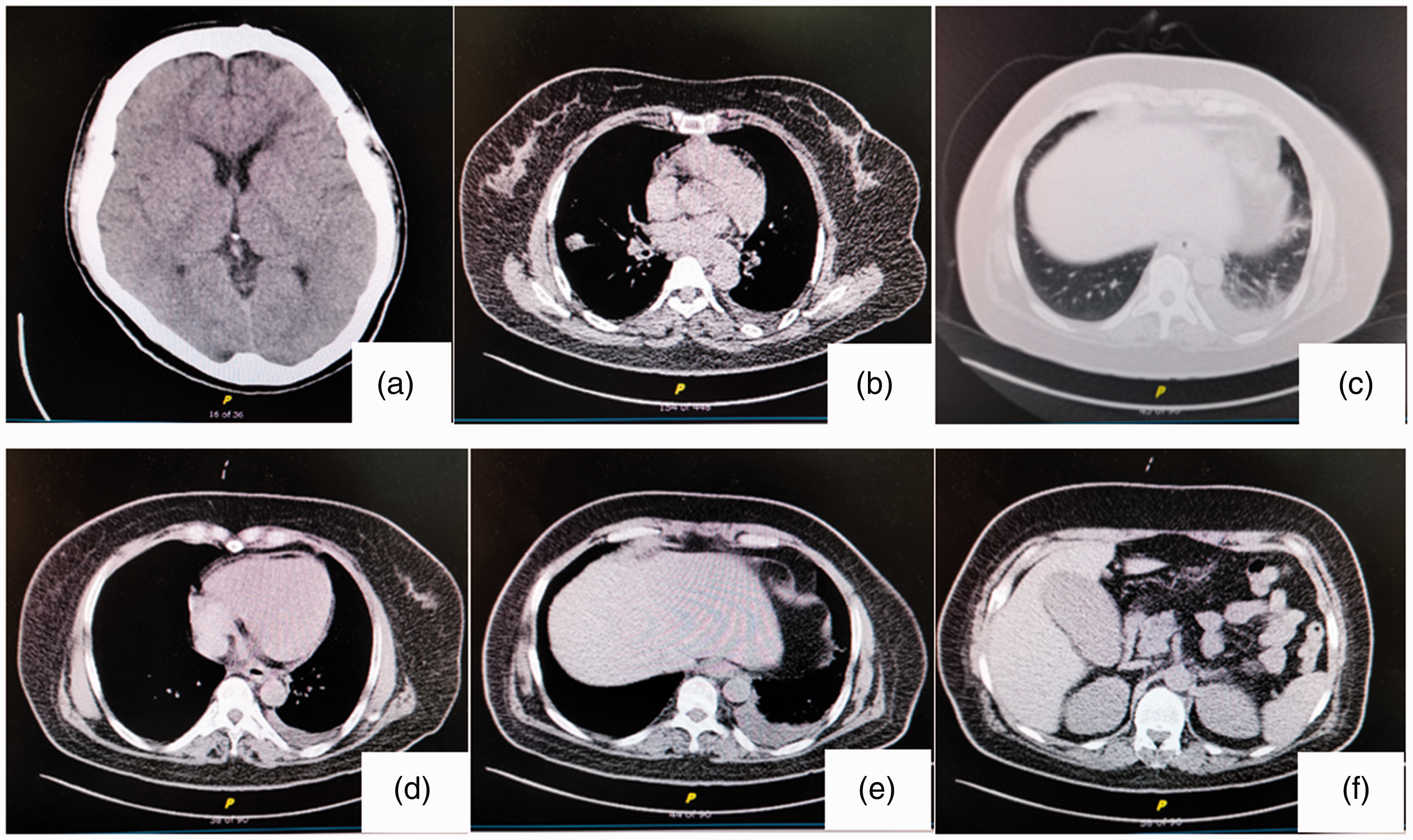
Plain computed tomography scan on 18 November 2020. (a) Suspicious low-density lesions on the lateral frontal angle of the left lateral ventricle can be seen. (b) Micronodules of the right lungs are present. (c–e) Bilateral pulmonary fiber foci and inflammation, a slightly thickened tracheobronchial wall, left pleural effusion, a slightly thick pericardium, a thickened esophagus and a slightly think gastric wall can be seen. (f) Intestinal gas can be seen, part of the intestinal border is unclear, and there is inflammation of the gallbladder and cholestasis.
The patient visited the hospital for a reexamination on 1 December 2020. A physical examination and reexamination of blood showed no obvious abnormalities, and a chest CT scan showed considerable improvement. In combination with the CT findings, esophageal stenosis was considered because the patient developed progressive dysphagia and choking from drinking water after poisoning. A gastroscopic examination showed esophageal wall stenosis at 16 cm from the incisors and the narrow lens barrel of the gastroscope was still passable. Additionally, esophageal wall stenosis and mucosal congestive edema were found at 21 cm from the incisors and the narrow lens barrel was not passable (Figure 3). Digestive tract angiography showed that the middle and lower part of the esophageal lumen extended to the cardia with different degrees of stenosis, there was serrated stenosis, and the length of stenosis was approximately 15 cm (Figure 4). After discharge, the patient transitioned from a semi-liquid diet, to a liquid diet and then to no food. The patient’s family members consulted staff at several hospitals and they believed that surgical thoracotomy and endoscopic esophageal dilation were not effective. Two months after discharge, the patient underwent gastrostomy at a local hospital, where a plastic tube was inserted through the abdominal wall to assist feeding. Eleven months after discharge, the patient was still able to inject food into her stomach through a plastic tube, but had lost more than 10 kg.
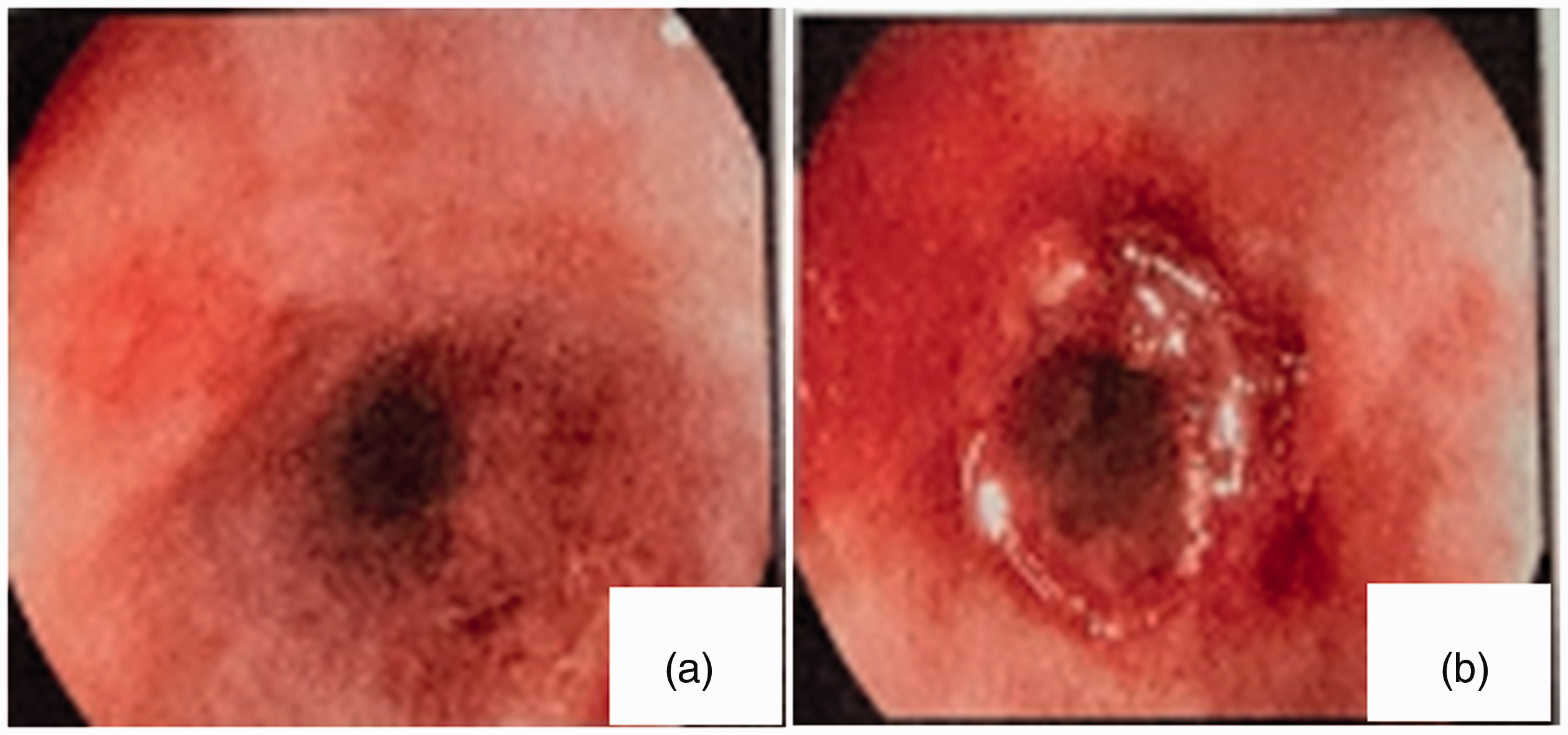
Photographs of gastroscopy. (a) Esophageal wall stenosis was found at 16 cm from the incisors. (b) Esophageal wall stenosis and mucosal congestive edema were found at 21 cm from the incisors.

Digestive tract angiography shows that the middle and lower parts of the esophageal lumen extend to the cardia with different degrees of stenosis, there is serrated stenosis and the length of the stenosis is approximately 15 cm.
Blood gas analysis during hospitalization in case 1.

PCO2, partial pressure of carbon dioxide; PO2, partial pressure of oxygen; cHCO3−, actual bicarbonate; BE, base excess; SO2, oxygen saturation; Lac, lactic acid.
Blood test results during hospitalization in case 1.

NEU, neutrophil ratio; RBC, red blood cells; Hb, hemoglobin; PLT, platelets; ALT, alanine aminotransferase; AST, aspartate aminotransferase; LDH, aspartate aminotransferase; TBIL, total bilirubin; DBIL, direct bilirubin; IBIL, indirect bilirubin; BUN, blood urea nitrogen; Cr, creatinine; Glu, glucose; CK, creatine kinase; CK-MB, creatine kinase isoenzyme.
Case 2
A man in his mid-40s was admitted to hospital because of oral administration of approximately 40 mL of MEKP for 3 days. The patient presented with perioral skin burn (Figure 5a), no nausea or vomiting, no chest tightness or palpitation and no abdominal pain or diarrhea. No special treatment had been provided. The patient then visited our hospital for further treatment. A physical examination on admission showed a body temperature of 36.8°C, heart rate of 90 beats/minute, blood pressure of 145/79 mmHg, and oxygen saturation of 99%. The patient’s condition was as follows: a clear mind, lethargy, sensitivity to light reflex, no abnormal secretions, thick breathing sounds, no dry or wet rales, a normal heart rhythm, no murmurs, a soft abdomen, no tenderness or rebound tenderness, and a normal liver and spleen. Physiological reflexes were present and pathological signs were not observed. A laboratory examination showed elevated inflammatory markers, hemoglobin value, and total bilirubin and urobilinogen concentrations, and no obvious abnormalities were found in liver or kidney function. The diagnosis at admission was acute MEKP poisoning.

(a) Corrosion damage of the mouth and lip. (b) Plain chest computed tomography scan shows esophageal thickening.
After admission, with the consent of the patient and his family, he immediately received symptomatic and supportive treatment of whole-stomach gastric lavage, hormones, liver protection, stomach protection (i.e., use of omeprazole to inhibit gastric acid secretion and protect the gastric mucosa), fluid rehydration, and diuresis. A blood reexamination on the fourth day after admission showed that inflammatory markers were still high, but they had improved compared with values at admission, an elevated hemoglobin value and no other obvious abnormalities were found. A CT scan showed a few fibrous foci in both lungs, pericardial thickening, and esophageal and gastric wall thickening (Figure 5b). No obvious abnormalities were found on brain magnetic resonance imaging. On the fifth day after admission, the patient complained of cough and sputum, and received treatment to relieve the coughing and reduce the sputum. The patient was discharged after 10 days of treatment. He was followed up every 6 months after discharge and reported slight dysphagia with little effect on his quality of life.
Discussion
The main symptoms of oral MEKP poisoning include three aspects. The first symptom is corrosion of the digestive tract due to the corrosive effects of MEKP. The second symptom is that MEKP causes abnormal heart, liver and kidney function because of metabolic abnormalities in the body. Oxidative stress may be the mechanism of multiorgan injuries. The third symptom is secondary acute complications, such as rhabdomyolysis, pneumonia and respiratory failure. Case 1 presented with oral mucosal erosion at the acute stage of poisoning, followed by progressive dysphagia and choking while drinking water. A CT scan showed esophageal wall thickening, and gastrointestinal endoscopy confirmed esophageal stenosis. Gastrointestinal angiography showed serrated stenosis of approximately 15 cm at the lower end of the esophagus, which greatly affected the patient’s quality of life. MEKP poisoning requires regular follow-up, and endoscopic dilation or surgical treatment if necessary. In this case, because of the excessively long stenosis of the esophagus, the effect of thoracotomy and endoscopic esophageal dilation was poor. Therefore, gastrostomy with tube placement was finally performed to assist with feeding. Case 2 also presented with oral mucosal erosion in the acute phase. A CT scan showed a thick esophagus and stomach wall, and then the patient presented with slight dysphagia. After long-term follow-up, there appeared to be little effect on the patient’s life.
In case 1, dark green urine (Figure 1) appeared at the early stage of poisoning, which was caused by MEKP metabolism in the body. In case 1, kidney function was normal, despite an elevation in blood urea nitrogen concentrations and the presence of dark urine. Organ dysfunction caused by metabolism is mainly myocardial injury. Peroxidation causes respiratory distress, metabolic acidosis and other symptoms. After timely administration of tracheal intubation, ventilator-assisted ventilation, hemoperfusion and other comprehensive symptomatic supportive treatment, the patient’s condition was effectively controlled. Although the two patients received similar doses of MEKP orally, the effect of MEKP was relatively mild in case 2 and did not cause organ dysfunction, which may be related to individual differences. The main clinical manifestations of case 2 were corrosion damage of the digestive tract mucosa and mild esophageal stenosis.
Van Enckevort et al. 4 found that treatment with a combination of N-acetylcysteine and hemodialysis may be a promising therapy for this severe and potentially life-threatening intoxication. This treatment should be administered within the first 24 hours of ingestion. However, further evidence is required to confirm the effectiveness of this treatment. According to clinical experience, the treatment of patients with oral MEKP poisoning is mainly blood purification, and treatment of the gastrointestinal tract and blood perfusion are important. The glucocorticoid dosage is selected according to the degree of MEKP, and comprehensive treatment, such as liver and kidney protection therapy, can be applied. In case 1, hemoperfusion effectively removed toxins in the blood and avoided further aggravation of organ injury. Using a similar treatment to that used in case 1, patients who are administered glucocorticoids have a shortened recovery period and hospitalization time. Glucocorticoids have anti-inflammatory and anti-toxic effects. In the clinical treatment of poisoning, the use of glucocorticoids can relieve symptoms, reduce the damage of the poisoning reaction to the body and help patients to survive the crisis.
According to Farrow et al, 8 the mouth should be rinsed with water after ingestion of MEKP. Drinking large volumes of milk or water to dilute the gastric contents can also be performed. Continuation of oral feeding is recommended unless perforation is suspected. Chang et al. 9 believed that oral administration of fluids should be avoided after MEKP ingestion because the gastrointestinal tract may be perforated. In case 1, the acute stage of poisoning was treated by gastric lavage. Both patients underwent catharsis, and no gastrointestinal perforation or gastric flatulence was observed. There are currently no guidelines on whether fluids should be taken orally after MEKP poisoning. We believe that a comprehensive evaluation should be conducted for MEKP poisoning. If there are no symptoms of gastrointestinal perforation, complete gastrointestinal cleansing should be performed to eliminate the remaining gastrointestinal toxins in time to prevent further absorption. If there is severe abdominal pain, tenderness, rebound pain and muscle tension in the whole abdomen, a diet should be banned and conservative treatment should be performed.
Conclusion
Oral administration of MEKP causes corrosion of the oral cavity and digestive tract, damage to multiple organs, such as the heart and liver, and even affects respiration. In the later stage, MEKP leads to gastrointestinal stenosis. In clinical practice, hemoperfusion combined with glucocorticoids and other comprehensive treatment should be adopted, and complications should be actively prevented. Additionally, patients should be followed up in the long term, and the next step of diagnosis and treatment should be performed in a timely manner if symptoms of esophageal stenosis occur.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
The reporting of this study conforms to the CARE guidelines. 10 Ethics approval was obtained from the Qilu Hospital Ethics Committee for Human Research (approval no. KYLL-2019-296). Written informed consent was obtained from the patient for publication of the case details and accompanying images.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
