Abstract
Objective
This study was performed to identify predictive factors for bacteremia among patients with pyelonephritis using a chi-square automatic interaction detector (CHAID) decision tree analysis model.
Methods
This retrospective cross-sectional survey was performed at Juntendo University Nerima Hospital, Tokyo, Japan and included all patients with pyelonephritis from whom blood cultures were taken. At the time of blood culture sample collection, clinical information was extracted from the patients’ medical charts, including vital signs, symptoms, laboratory data, and culture results. Factors potentially predictive of bacteremia among patients with pyelonephritis were analyzed using Student’s t-test or the chi-square test and the CHAID decision tree analysis model.
Results
In total, 198 patients (60 (30.3%) men, 138 (69.7%) women; mean age, 74.69 ± 15.27 years) were included in this study, of whom 92 (46.4%) had positive blood culture results. The CHAID decision tree analysis revealed that patients with a white blood cell count of >21,000/μL had a very high risk (89.5%) of developing bacteremia. Patients with a white blood cell count of ≤21,000/μL plus chills plus an aspartate aminotransferase concentration of >19 IU/L constituted the high-risk group (69.0%).
Conclusion
The present results are extremely useful for predicting the results of bacteremia among patients with pyelonephritis.
Keywords
Introduction
Pyelonephritis is a common disease encountered by many physicians. For example, in the United States, approximately 250,000 cases of acute pyelonephritis are diagnosed each year, resulting in more than 100,000 hospitalizations. 1 The management guidelines for urinary tract infections in the United States and Japan recommend that patients with mild pyelonephritis be treated in an outpatient clinic,2,3 whereas patients with severe disease should be considered for hospitalization and initial administration of intravenous antibiotics. 4 Hospitalization is preferable for patients with pyelonephritis accompanied by bacteremia because severe pyelonephritis accompanied by bacteremia has a mortality rate of 10% to 20%.5,6 Typically, however, the results of blood cultures are not available on the same day that a patient is diagnosed with pyelonephritis; because results are expected on a later day, patients with pyelonephritis may be hospitalized as a precautionary measure before blood culture results are obtained. The ability to predict positive results of blood cultures on the same day that patients are diagnosed with pyelonephritis would be clinically useful. Based on our preliminary research, blood culture-positive causative diseases are often diagnosed as pyelonephritis in patients visiting departments of general medicine. 7 Furthermore, other preliminary research has indicated that older age, positive urinary nitrite test results, and chills tend to be seen in patients with pyelonephritis accompanied by bacteremia. 8 However, the quick Sequential Organ Failure Assessment (qSOFA), which is now widely used, was not incorporated as part of this preliminary research. 9 Furthermore, a score classification system for recommendation of hospitalization does not exist for patients with pyelonephritis, in contrast to our experience with other diseases (e.g., CURB-65 for patients with pneumonia).10–12 As described above, among patients with pyelonephritis, those with bacteremia typically require hospitalization; there would be a great advantage to a classification system that identifies the need to hospitalize patients with pyelonephritis who are at increased risk of developing bacteremia.
To identify predictive factors for bacteremia among patients diagnosed with pyelonephritis, we compared clinical parameters between patients with and without bacteremia among those diagnosed with pyelonephritis. The goal of this process was to determine which patients with pyelonephritis should be hospitalized.
Methods
Study design and study population
This retrospective, cross-sectional study was performed at Juntendo University Nerima Hospital, a 400-bed university-affiliated hospital in Tokyo, Japan. We recruited patients who were diagnosed with pyelonephritis from 1 January 2010 to 31 July 2020. We excluded patients from whom blood cultures had not been taken and patients who had already (as part of the same admission) received an antimicrobial agent prior to blood culture submission (given possible effects on blood culture results).
We also excluded patients with obstructive pyelonephritis due to a calculus or tumor and those who had undergone urgent stenting because these individuals were hospitalized regardless of the blood culture results. Pyelonephritis was diagnosed in conformity with the infection guidelines of Japan (Japanese Association for Infectious Disease Guide to Clinical Management of Infectious Disease). However, the clinical diagnosis of pyelonephritis is generally consistent among countries, reflecting the clear criteria of blood culture results and specific standard symptoms (e.g., chills, vomiting, and malaise). Additionally, given that pyelonephritis is diagnosed based on multiple clinical findings, several physicians are involved in each such diagnosis. In our hospital, more than three doctors (representing all our hospital doctors, including infectious disease specialists) were involved in each diagnosis. Bacteremia in patients with pyelonephritis was defined as the detection of identical causative bacteria from both blood and urine cultures. Clinical information extracted by chart review included (but was not limited to) the following: vital signs including the qSOFA score, subjective symptoms, objective physical findings, laboratory findings, and results of blood and urine cultures.
Statistical analysis
Patients’ characteristics were compared between those with and without bacteremia using the t-test for continuous data or the chi-square test for categorical data. We then applied chi-square automatic interaction detector (CHAID) decision tree analysis to identify risk factors for bacteremia. CHAID decision tree analysis is a data-mining technique13,14 with a salient advantage of advanced graphic presentation for interpretation. 15 CHAID enables the consideration of whole variables, effective partitioning of continuous data, and construction of decision trees by use of a forward-stopping or -pruning rule. 16 Moreover, CHAID is the only model that permits the formulation of multiple nodes. 15 Unlike other techniques, CHAID permits the significance level to be adjusted for the number of comparisons. CHAID has been previously applied in the medical field17,18 and has been shown to be superior to logistic analysis. 19 In addition, prediction rules with the CHAID model are visibly intuitive and easy to interpret in clinical settings. In the present study, mother and daughter nodes were set as 25 and 15, respectively. To compare the predictive ability of qSOFA and CHAID, the model’s goodness of fit was examined by the receiver operating characteristic (ROC) curve and area under the curve (AUC). All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp., Armonk, NY, USA).
This study was approved by the Ethics Committee of Juntendo University Nerima Hospital (Approval Number 2020054). This was an observational study, and the requirement for written informed consent was waived because of a public health outbreak investigation of the ethics committee.
Results
In total, 261 patients were diagnosed with pyelonephritis during the study period. Within this population, 27 (10.3%) patients were excluded because of urgent stenting for obstructive pyelonephritis, and 36 (13.8%) patients were excluded because of precedent administration of an antimicrobial agent before blood culturing. As shown in Table 1, 198 patients (60 (30.3%) men, 138 (69.7%) women; mean age, 74.69 ± 15.27 years) were included in this study, and 92 (46.4%) patients had bacteremia. Table 1 also shows the patient characteristics in the two groups (with and without bacteremia) and the results of the bivariable analysis. The factors related to bacteremia were chills, the neutrophil count, the band-form neutrophil count, the lymphocyte count, the blood urea nitrogen concentration, the total protein concentration, the albumin concentration, the lactate dehydrogenase concentration, the C-reactive protein (CRP) concentration, and the hemoglobin A1c concentration.
Patient characteristics.

Data are presented as mean ± standard deviation or n (%).
*p < 0.05.
The algorithm for the prediction of bacteremia derived from CHAID is shown in Figure 1. Based on the observed results, the patients were categorized into three risk groups: low risk (≤30%), moderate risk (30%–60%), and high risk (≥60%). The white blood cell (WBC) count (≤21,000/μL, >21,000/μL), chills, the aspartate aminotransferase (AST) concentration (≤19 IU/L, >19 IU/L), and the albumin concentration (≤3.60 g/dL, >3.60 g/dL) were included in the decision tree, and five terminal nodes were employed. According to the incidences, we divided these nodes into the following categories: low risk, 16.3% incidence of blood culture-positive pyelonephritis; moderate risk 1, 34.5%; moderate risk 2, 44.6%; high risk, 69.0%; and very high risk, 89.5%. We evaluated the quality of this model using the ROC curve, which yielded an AUC of 0.750 and a 95% confidence interval of 0.682 to 0.817 (Figure 2).
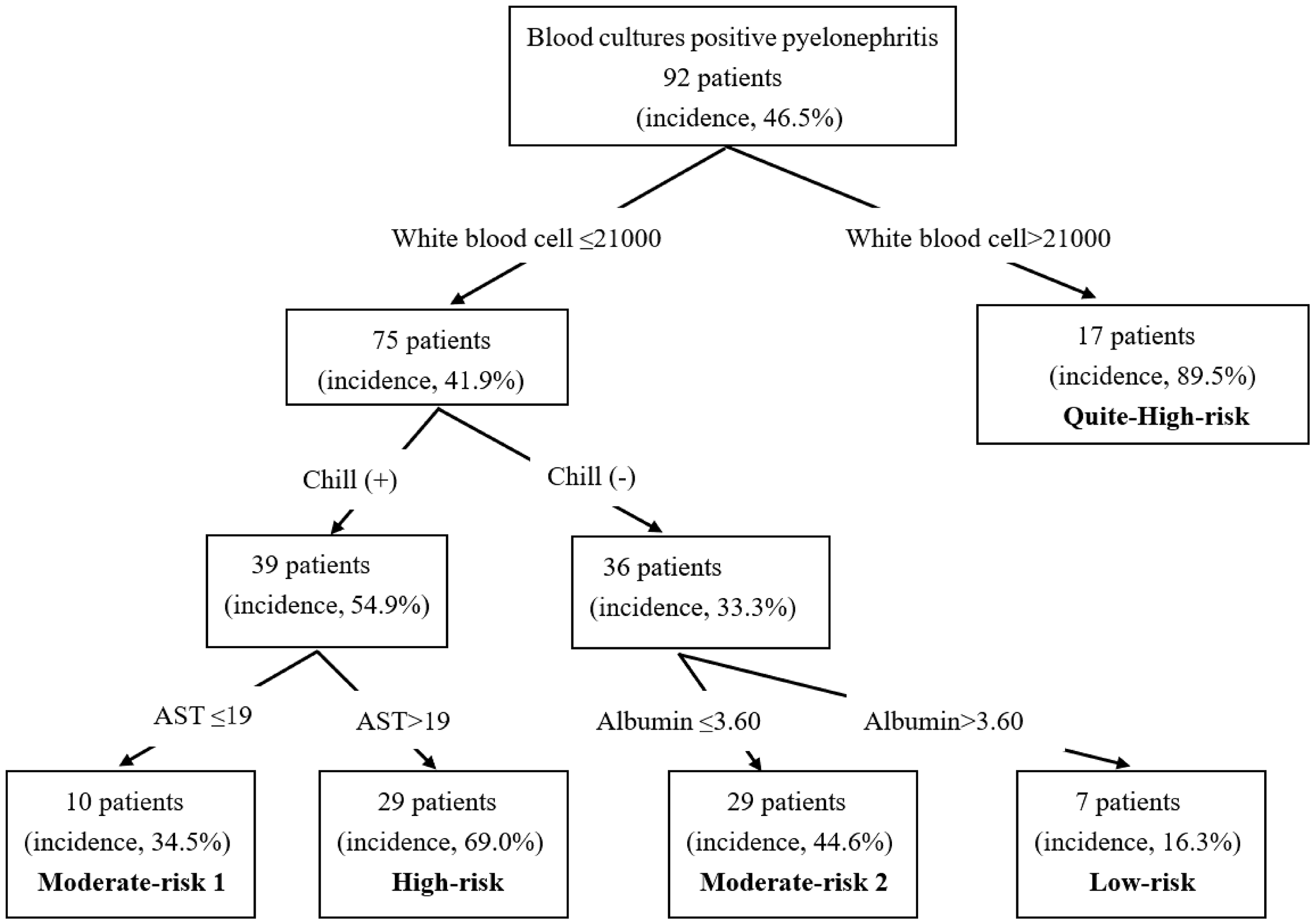
Algorithm for the prediction of bacteremia derived from CHAID. Categories were defined based on the incidence of bacteremia as follows: low-risk group, ≤30%; moderate-risk group, 40%–60%; high-risk group, ≥60%.

Receiver operating characteristic curve of CHAID-formulated decision tree for positive risk factors for bacteremia among patients with pyelonephritis.
The results of the chi-square test of the qSOFA score are shown in Table 2. Patients with pyelonephritis with a qSOFA score of 2 had a significantly higher percentage of bacteremia (p = 0.04). We evaluated the quality of qSOFA using the ROC curve, which yielded an AUC of 0.537 and a 95% confidence interval of 0.456 to 0.617 (Figure 3). Comparison between the AUCs in Figures 2 and 3 indicated that the model obtained by CHAID decision tree analysis in the present study had greater power than that obtained by qSOFA. 20
Results of the chi-square test of the qSOFA score.

Data are presented as n (%).
qSOFA, quick Sequential Organ Failure Assessment.
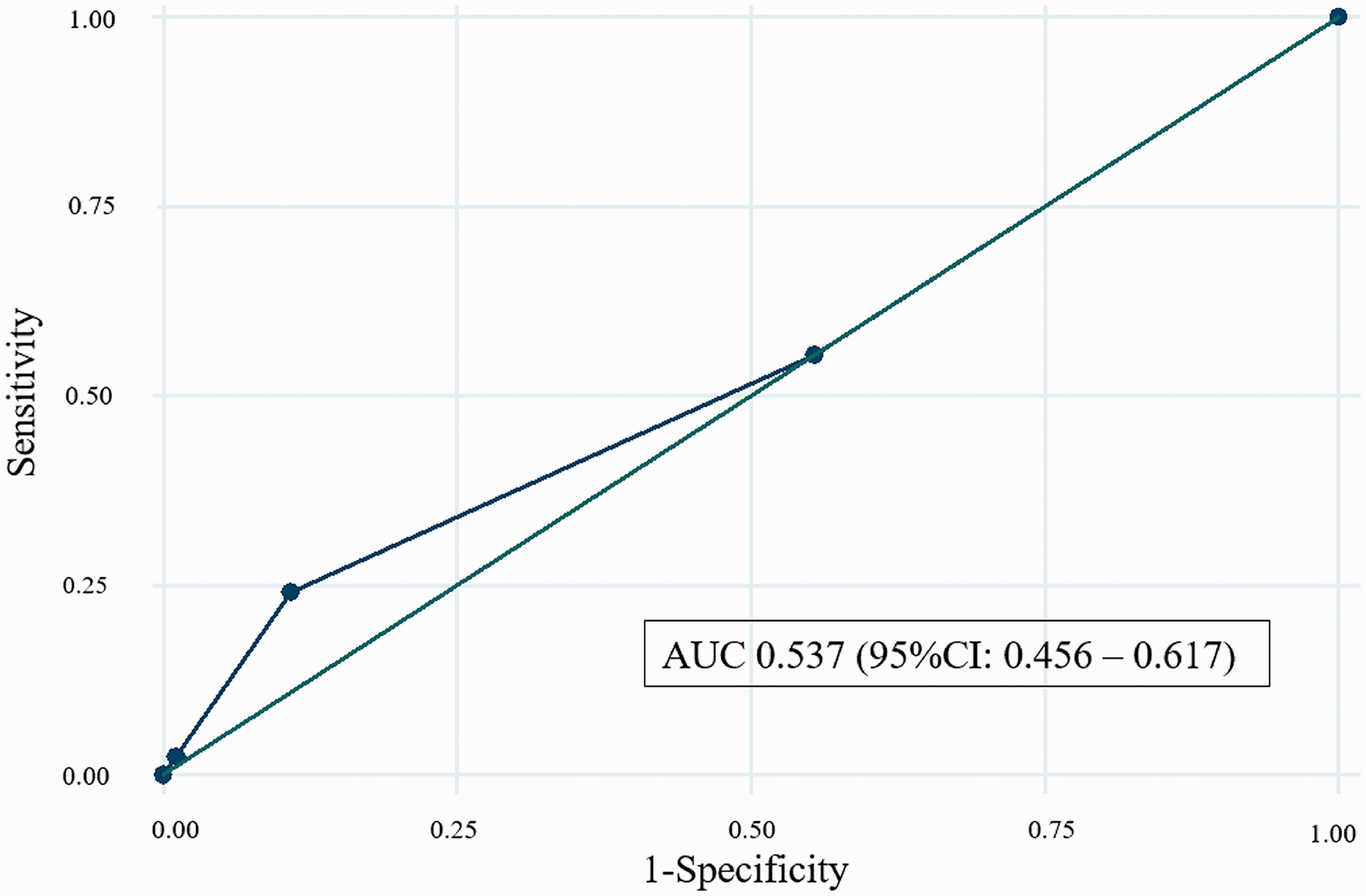
Receiver operating characteristic curve of the quick Sequential Organ Failure Assessment for positive risk factors for bacteremia among patients with pyelonephritis.
Table 3 shows the results of the blood and urine cultures. Escherichia coli was the most frequent causative microorganism.
Results of blood and urine cultures.

Data are presented as n (%).
ESBL, extended-spectrum beta-lactamase.
*Polymicrobial infections included the following:
• Escherichia coli + Escherichia coli (ESBL-producing)
• Escherichia coli (ESBL-producing) + Pseudomonas aeruginosa
• Escherichia coli (ESBL-producing) + Enterococcus faecalis
• Escherichia coli (ESBL-producing) + Klebsiella pneumoniae
• Escherichia coli + Klebsiella pneumoniae
• Escherichia coli + Morganella morganii
• Escherichia coli + Staphylococcus aureus
• Escherichia coli + Pseudomonas aeruginosa
• Proteus vulgaris + Serratia marcescens
• Enterococcus faecalis + Pseudomonas aeruginosa
• Klebsiella oxytoca + Morganella morganii
Discussion
To our knowledge, this is the first study to use CHAID decision tree analysis for direct comparison between patients with and without bacteremia among those with pyelonephritis. Patients with a WBC count of >21,000/μL constituted the very-high-risk group. Patients with a WBC count of ≤21,000/μL plus chills plus an AST concentration of >19 IU/L constituted the high-risk group. In contrast, patients with a WBC of ≤21,000/μL plus no chills plus an albumin concentration of >3.60 g/dL constituted the low-risk group. The AUC demonstrated acceptable accuracy.
The WBC count, CRP concentration, and procalcitonin concentration are frequently used as markers of a systemic inflammatory reaction, but the CRP and procalcitonin concentrations were not incorporated into the model for bacteremia among patients with pyelonephritis in our CHAID decision tree analysis. Several studies have shown the usefulness of the CRP concentration for estimating the risk of bacteremia in patients with neutropenia who have cancer 21 or are being treated in the intensive care unit 22 ; in contrast, another study concluded that the CRP concentration was not a sensitive or specific marker for bacteremia in patients with signs of sepsis. 23 Indeed, the CRP concentration was not detected as a bacteremia-related factor in our preliminary research. 7 Similarly, another study demonstrated the usefulness of procalcitonin in judging the severity of sepsis and revealed its high prognostic value. 24 In contrast, a meta-analysis concluded that procalcitonin was not a sensitive or specific marker for bacteremia in patients with signs of sepsis. 25 Our study results suggest that an increase in the WBC count is a timely and reliable response to bacteremia. It is important to consider obtaining blood cultures when patients have a high WBC count, even when the CRP and procalcitonin concentrations are not elevated. Furthermore, hospitalization should be strongly considered for patients with a WBC count of >21,000/μL because these patients constituted the very-high-risk group in the present study.
Several studies have shown that the presence of chills is a powerful single predictor of bacteremia.26–30 In Japanese emergency rooms, the severity of chills reportedly correlates with the risk of bacteremia.31,32 Separately, increases in the AST concentration are postulated to reflect rhabdomyolysis associated with fever and early circulatory impairment. Even if the WBC count remains below 21000/μL, hospitalization is recommended when patients have chills and an AST concentration of >19 IU/L because these patients constituted the high-risk group in the present study.
Another study formulated a predictive model for the mortality of patients with Staphylococcus aureus bacteremia, in which a low serum albumin concentration was one of the predictors. 33 Furthermore, hypoalbuminemia is an acute-phase inflammation biomarker.34–36 In contrast, our results suggest that hyperalbuminemia is relevant marker because a serum albumin concentration of >3.60 g/dL was a predictor for the low-risk group.
Patients with pyelonephritis who exhibited a WBC count of ≤21,000/μL plus no chills plus an albumin concentration of >3.60 g/dL constituted the low-risk group in the present study; thus, it is possible that such patients with low-risk pyelonephritis can be treated on an outpatient basis. However, the CHAID analysis still provides only an index; it remains important to carefully ascertain the state of each patient and decide on treatments in an informed manner. If a patient’s overall clinical status is poor, hospitalization should be considered.
The qSOFA is now widely used. In out-of-hospital, emergency department, or general hospital ward settings, adult patients with suspected infections can be rapidly identified as being more likely to have poor outcomes typical of sepsis if these patients meet at least two of the following three clinical criteria: respiratory rate of ≥22 breaths/minute, altered mentation, or systolic blood pressure of ≤100 mmHg. 9 In the present study, patients with pyelonephritis who presented with a qSOFA score of 2 had a significantly higher percentage of bacteremia (p = 0.04) (Table 2). However, based on the AUCs shown in Figures 2 and 3, the model based on the CHAID decision tree analysis exhibited greater power than qSOFA. Thus, CHAID decision analysis was superior to qSOFA in identifying bacteremia-predictive factors among patients with pyelonephritis. In fact, other studies providing comparisons with qSOFA have already been reported,37,38 and further such studies are expected in the future.
Based on the literature, E. coli is the most frequent cause of pyelonephritis. 39 In this study, E. coli was also the most frequent causative microorganism. Among the possible virulence factors of this species is the ability to adhere to and colonize the urinary tract, an important initiating factor in all urinary tract infections. 39 Furthermore, in the present study, 13.5% of E. coli isolates (10 of the total 74 E. coli cases) were extended-spectrum beta-lactamase (ESBL) producers. A previous study also showed that ESBL-producing E. coli is being increasingly recognized among community-acquired infections. 40 Given that the fraction of ESBL-producing E. coli may further increase in future, there is an increasing need to exercise care in the choice of medical treatment for pyelonephritis.
This study had some limitations. Our hospital is a university hospital, and our medical staff typically examines a large number of patients referred from general practitioners within the Japanese medical system. Thus, our patient population may include patients with more serious pyelonephritis. Furthermore, the submission of blood cultures may vary among countries (for example, only inpatient samples may be submitted); thus, our results may not be directly applicable to all countries.
The present study used a CHAID model, but the risk of bacteremia may also be evaluated by other techniques. Furthermore, we note that some values are missing in Table 1. We suspect that the missing values may have affected our CHAID analysis, given that CHAID treats all systems and user-missing values for each independent variable as a single category. For scale and ordinal independent variables, a given category may or may not be subsequently merged with other categories of that independent variable, depending on the growing number of criteria. Thus, we believe that the effect of missing values on our results was minimized.
The patient population enrolled in this study was limited to a single hospital. In addition, this study was retrospective in nature. As a next step, a multicenter prospective study should be conducted with a larger number of patients.
Conclusions
Our results emphasize the importance of hospitalization among patients with pyelonephritis who are classified in the very-high-risk group (WBC count of >21,000/μL) or the high-risk group (WBC count of ≤21,000/μL plus chills plus AST concentration of >19 IU/L). Furthermore, it is possible that patients with pyelonephritis who are classified in the low-risk group (WBC count of ≤21,000/μL plus no chills plus albumin concentration of >3.60 g/dL) can be treated in outpatient clinics.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
