Abstract
Objective
The Pain Sensitivity Questionnaire (PSQ) is a clinically beneficial instrument that has been proven to be correlated with various experimental pain sensitivity assessments in healthy people and in patients with chronic pain. In this study, we aimed to translate the PSQ into Turkish (PSQ-T) and validate it for the measurement of pain sensitivity among Turkish people.
Methods
Seventy-three patients with chronic back pain who were planning to undergo an interventional procedure completed the Brief Pain Inventory-Short Form (BPI-SF), Beck Depression Inventory (BDI), Beck Anxiety Inventory, Pain Catastrophizing Scale, and PSQ prior to their procedure. Subcutaneous infiltration of lidocaine was used as a standardized experimental pain stimulus. Pain was evaluated using a visual analog scale (VAS 1: infiltration in the hand, and VAS 2: infiltration in the procedure area)
Results
Scores on the PSQ-T were significantly correlated with those on the BPI-SF. A significant positive relationship was observed between VAS 1 and VAS 2 values and the PSQ-T score, BPI pain score, and BPI interference score.
Conclusions
The PSQ-T can be used as a valid and reliable tool for the assessment of pain sensitivity in the Turkish population.
Introduction
Patients with chronic pain frequently undergo interventional procedures for the purpose of pain control or as a part of treatment for an underlying illness. The perception of the pain intensity occurring during and after such procedures may differ from person to person.1,2 Use of the same amount of analgesia for every patient may result in either insufficient or excessive treatment because pain intensity varies from patient to patient. The ability to estimate pain could be beneficial in planning appropriate treatment. Innate pain sensitivity is an important factor that influences the perception of procedure-related pain, and the determination of this sensitivity may be helpful for the prediction and control of procedural and postoperative pain. 3 Innate pain sensitivity may affect the need for sedatives and analgesics during an intervention, as well as the response to pain treatment. 4 Pain sensitivity not only affects symptoms but also the course of treatment. It has been shown that patients with chronic pain display a general increase in localized pain as well as in pain perception.5–7 High pain sensitivity is a risk factor for a patient’s pain becoming chronic, and pain sensitivity can be used to predict treatment success rates.8–11 For these reasons, the assessment of general pain sensitivity is important in patients with chronic pain.
Different types of stimuli (heat, mechanical, ischemia, or electricity) are used in experimental studies to assess innate pain sensitivity. 12 However, the application of these methods is hampered by the need for specialized equipment, a highly labor-intensive and time-consuming nature, cost, and the negative effects on individuals involved in such experiments. 13 As an instrument that is free of such restrictions, the Pain Sensitivity Questionnaire (PSQ) was developed by Ruscheweyh et al. 13 The PSQ comprises 17 items in which patients imagine painful situations in daily life and rate their pain intensity levels from 0 to 10. The PSQ is a clinically valid tool that has been proven to be correlated with experimental pain sensitivity assessment in healthy people and in patients with chronic pain.13–15 Compared with other methods, the PSQ is easier to use and it has been used to identify pain sensitivity in clinical practice.16–22
Validation studies of the PSQ have been conducted in different languages.4,23–26 However, the PSQ has not yet been studied in a Turkish-speaking population and has not been applied in a pain management clinic. In this study, we aimed to apply the Turkish version of the PSQ (PSQ-T), by translating the original German version into Turkish and applying the instrument in an adult Turkish population with chronic pain to ensure its validity.
Methods
Study participants and ethical considerations
This was an observational study with a prospective, single-arm design. The study is registered at Clinicaltrials.gov (identifier: NCT03609788). Ethical approval was received from the Medical Faculty of Bezmialem Vakif University (IRB: 71306642-050.01.04). Written informed consent was obtained from all patients before their inclusion in the study.
This study was conducted among adult patients with American Society of Anesthesiologists (ASA) physical status classification system grade I–II.27,28 Included patients were aged 18 to 90 years; had chronic back pain for longer than 3 months; and planned to receive lumbar epidural steroid injection (LESI), lumbar facet joint injection (LFJI), or sacroiliac joint injection (SIJI). Patients who had failed back surgery syndrome, were morbidly obese (body mass index [BMI] > 40), could not speak Turkish, had diabetic neuropathy, were substance users, had major psychiatric disorders (severe depression, schizophrenia, bipolar disorder), and those who were allergic to the drugs planned for use were excluded from the study. Patient data, including age, sex, height, weight, pain duration, and use of analgesics (opioid/non-opioid) were recorded.
Measurement instruments
Before the study procedures were carried out, all patients completed the Brief Pain Inventory-Short Form (BPI-SF), Beck Depression Inventory (BDI), Beck Anxiety Inventory (BAI), Pain Catastrophizing Scale (PCS), and PSQ.
The visual analog scale (VAS) is commonly used in pain management and clinical studies to measure pain intensity.29,30 Patients state the intensity of their pain on a horizontal line labeled from “no pain” (pain score 0) and “the worst pain” (pain score 10).
The BPI-SF is a questionnaire used to assess pain intensity and its impact on daily functions with a numeric scale of 11 points. 31 On the BPI-SF, pain intensity is rated from “no pain” (pain score 0) to “the worst pain you could ever dream of” (pain score 10). The instrument also measures how the pain limits seven daily activities or states (general activity, walking, work, mood, enjoyment of life, relationships with others, and sleep) during the previous 24 hours. 31 Patients rank each activity on an 11-point scale from 0 to 10 (from “does not interfere” to “completely interferes”). The BPI-SF interference value is scored as the average of seven daily activity items. The Turkish version of the BPI-SF has been validated. 22
The BAI and BDI consist of 21 questions each. Each item is rated between 0 (never) and 3 (serious), and high scores indicate an increase in complaints of anxiety and increased intensity of depressive complaints, respectively. The total points on these scales range between 0 and 63.32–35 The Turkish versions of the BAI and BDI have been validated.34,35
The PCS is a self-report questionnaire comprising 13 items that is used to assess relationships between the patient’s emotions, thoughts, and feelings and their pain and pain catastrophizing. The PCS has three sub-dimensions: helplessness, magnification, and rumination. A 5-point scale (0: never, 4: always) is used to rate each item. The total PCS score ranges from 0 to 52, and higher scores indicate more pain catastrophizing. 36 The Turkish version of the PCS has been validated. 37
The PSQ is a self-report questionnaire used to measure pain sensitivity. The PSQ yields results similar to experimental methods for assessing pain sensitivity. 13 The PSQ involves rating the intensity of 17 different painful states in daily life on an 11-point scale (0; no pain, 10; the most intense pain). The survey can be completed within 5 to 10 minutes.
Three items address states that are not assessed as painful by healthy individuals (for instance, having a hot shower), and these are not added to the total score. Two additional scores have been developed, the PSQ-minor score and PSQ-moderate score, as well as the PSQ-total score. These sub-scores have been defined using factor analysis in a previous study. 13 The PSQ-minor includes seven conditions that cause low levels of pain (average score <4 on an 11-point scale). The PSQ-moderate comprises seven conditions that cause moderate levels of pain (average score 4–6 on an 11-point scale). The PSQ-total score is calculated by taking the average values of item numbers 1–4, 6–8, 10–12, and 14–17 (except for the three painless states). The PSQ-minor score is calculated as the average score for items 3, 6, 7, 10–12, and 14. The PSQ-moderate score is calculated as the average score for items 1, 2, 4, 8, and 15–17.
The original German version of the PSQ (PSQ-G) is significantly correlated with experimentally produced pain intensity degrees in both healthy individuals and patients with chronic pain.13–15 The PSQ has been translated into Turkish from German, in accordance with international standards. 38 First, the PSQ was translated from German into Turkish by two bilingual Turkish medical translators independently of each other. The two versions were compared and the Turkish version was then back-translated into German by two bilingual German translators independently of each other; the translators were unaware of the purpose of translation. The back-translated German version was compared with the original, corrections were made, and this retranslated German version was approved by a professional translator. A pilot study was conducted with 10 healthy volunteers; to assess cultural appropriateness, respondents were asked whether they had any uncertainties, difficulties, or linguistic problems with respect to the questionnaire. No corrections were needed following the pilot study. The Turkish version of the PSQ is shown in Appendix 1.
Experimental pain stimulus
In this study, controlled subcutaneous injection was used as a noxious stimulus, in accordance with clinical protocols, similar to previous clinical studies.39–43 Subcutaneous infiltration of lidocaine was used as the standardized experimental pain stimulus to assess hyperalgesia, as well as verbal and behavioral pain responses in patients receiving interventional procedures to treat chronic back pain.44–46
Study protocol
Patients who signed the written informed consent form and completed the questionnaires were enrolled in the study. As a subcutaneous local anesthetic injection, 1 mL of 1% lidocaine (Aritmal 2%; Osel, Istanbul, Turkey) was injected using a 25-gauge needle intò the dorsum of the dominant hand before insertion of an intravenous cannula. Patients were then asked to assess the pain related to this injection, and their responses were recorded as VAS 1. Next, with the patient lying in a prone position on a surgical table, an appropriate injection site for fluoroscopy was determined. A 1-mL subcutaneous injection of 1% lidocaine was given using a 25-gauge needle. The patient was asked to assess the pain related to the second injection, and their response was recorded as VAS 2. After that, deeper local anesthesia was applied to the area in which the interventional procedure was to be carried out, with 8 mL of 1% lidocaine injected using a 22-gauge needle under sedation with 2 mg midazolam (Dormicum; Deva, Istanbul, Turkey) and 50 µg fentanyl (Fentanyl; Johnson and Johnson, Istanbul, Turkey) given intravenously. For the LESI procedure, an 18-gauge, 3.5-inch, Tuohy needle was inserted in an anteroposterior (AP) and lateral manner using the loss of resistance under fluoroscopy. To verify the epidural expansion and decrease the likelihood of intravascular injection, 1 mL of contrast medium was injected in the lateral and AP positions. A local anesthetic (lidocaine) and steroid (triamcinolone or methylprednisolone) (KenaCort-A; Deva and Depomedrol; Zentiva, Istanbul, Turkey, respectively) mixture was injected. A 22-gauge, 3.5-inch spinal needle was inserted in an AP, lateral, and oblique manner under fluoroscopy in the LFJI and SIJI. When the needle tip could be seen within the joint space, 0.5 mL contrast medium was injected to verify in-joint and periarticular expansion and decrease the likelihood of vascular injection. A local anesthetic (bupivacaine) (Marcaine; AstraZeneca, Istanbul, Turkey) and steroid (triamcinolone or methylprednisolone) mixture was injected without force. All patients were taken to the recovery unit after their procedure, and all were given standard clinical care.
Sample size determination
The degree of correlation between the English version of the PSQ-minor (PSQ-E-minor) and VAS 1 in previous studies was calculated as 0.26, and the correlation coefficient with VAS 2 as 0.34; the correlation between the English version of the PSQ-total (PSQ-E-total) and BPI interference score was calculated as 0.40. 4 A sample size of 70 individuals was calculated when the impact size attained in this study was taken into consideration; the power of the test was set to 80% and the type I error rate (alpha coefficient) was 5%.
Statistical analysis
We used IBM SPSS version 22.0 (IBM Corp., Armonk, NY, USA) for the statistical analysis. The one-sample Kolmogorov–Smirnov Test was used to test whether numerical variables had a normal distribution. An independent-samples t-test was used for comparisons by sex for normally distributed variables, and the Mann–Whitney U test was used for non-normally distributed variables. The relationships among categorical variables were assessed using Pearson’s correlation analysis. The absolute values of r were as follows: 0.00–0.19 very weak, 0.20–0.39 weak, 0.40–0.59 moderate, 0.60–0.79 strong, and 0.80–1.0 very strong. 47 p < 0.05 was set as the threshold to indicate statistical significance.
Results
Although we initially aimed to enroll 120 individuals, only 73 patients were finally enrolled in this study in accordance with the inclusion/exclusion criteria. The demographic characteristics of the included patients are presented in Table 1. The mean patient age was 57.37 (standard deviation [SD] = 15.37) years; 65.8% of patients were women and 34.2% were men.
Patients’ demographic characteristics.
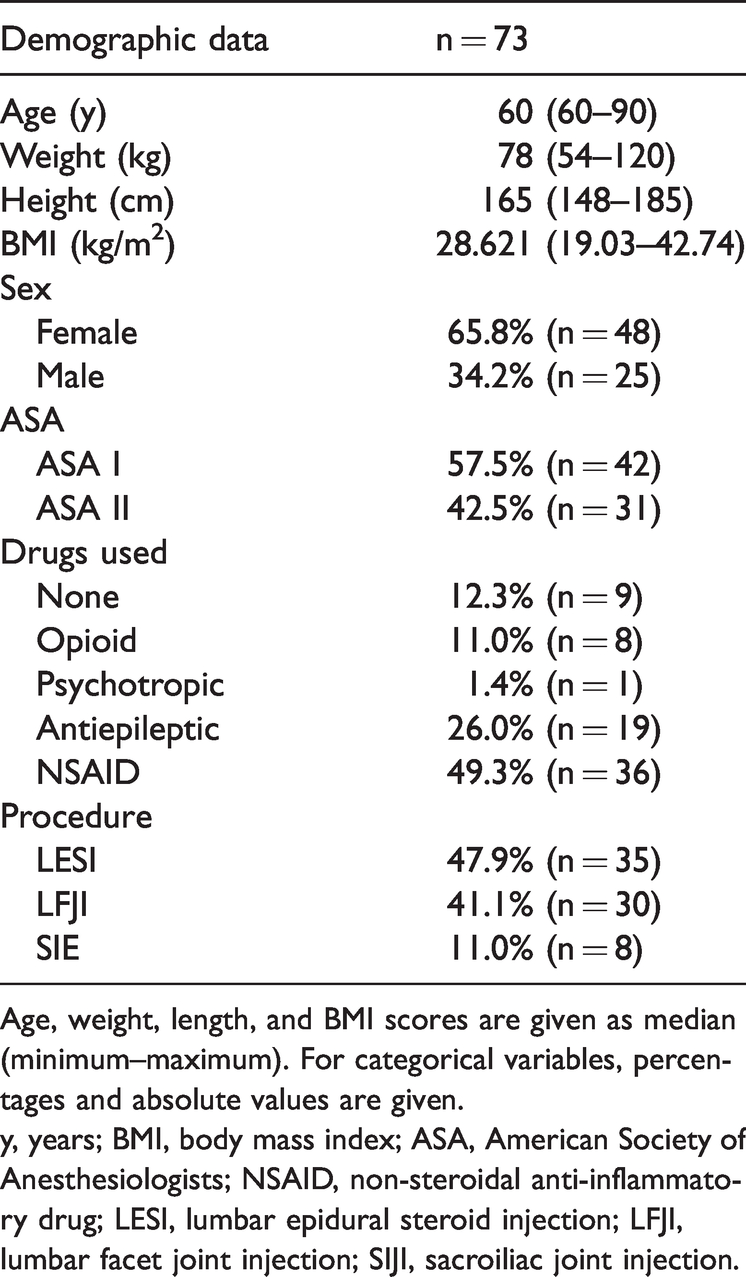
Age, weight, length, and BMI scores are given as median (minimum–maximum). For categorical variables, percentages and absolute values are given.
y, years; BMI, body mass index; ASA, American Society of Anesthesiologists; NSAID, non-steroidal anti-inflammatory drug; LESI, lumbar epidural steroid injection; LFJI, lumbar facet joint injection; SIJI, sacroiliac joint injection.
Patients’ scores on the PSQ-T, VAS, BPI, BDI, BAI, and PCS are given in Table 2. PSQ scores were not correlated with psychological cofactors, depression, anxiety, or pain catastrophizing (BDI, BAI, and PCS scores, respectively; Table 3). Similarly, PSQ scores were not correlated with age, BMI, and pain duration (Table 3). However, we observed a significant positive correlation of Turkish PSQ-T-total, PSQ-T-minor, PSQ-T-moderate with BPI pain scores (p < 0.05; r = 0.28; r = 0.24; r = 0.31, respectively) (Table 3). We observed a significant positive correlation between the BPI interference score and PSQ-T-total and PSQ-T-minor scores (p < 0.05; r = 0.31; r = 0.34, respectively) (Table 3).
Patient characteristics.

PSQ-T, Pain Sensitivity Questionnaire-Turkish; VAS, visual analog scale; BPI, Brief Pain Inventory; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory; PCS, Pain Catastrophizing Scale.
Correlations of PSQ-T-total, PSQ-T-minor, and PSQ-T-moderate with parameters.

Pearson correlation analysis was used.
*p < 0.05.
PSQ-T, Pain Sensitivity Questionnaire-Turkish; VAS, visual analog scale; BPI, Brief Pain Inventory; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory; PCS, Pain Catastrophizing Scale; BMI, body mass index.
A significantly positive relationship was observed between VAS 1 and VAS 2 values of the PSQ-T, BPI, and BPI interference scores (p < 0.05) (Table 4). The correlation between VAS 1 and VAS 2 of PSQ-T scores was highly significant (r > 0.60, strong correlation) (Table 4). VAS 1 and VAS 2 values were not associated with psychological cofactors of pain (BDI, BAI, and PCS scores, respectively; Table 4). When parameters were compared in terms of sex, only the average BAI score was found to be significantly higher in women (p = 0.028) (Table 5).
Correlation of VAS 1 and VAS 2 values with other parameters.

PSQ-T, Pain Sensitivity Questionnaire-Turkish; VAS, visual analog scale; BPI, Brief Pain Inventory; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory; PCS, Pain Catastrophizing Scale.
Pearson correlation analysis was used.
*p < 0.05.
Correlations of parameters according to sex.

aIndependent-samples t-test.
bMann–Whitney test.
*p < 0.05.
mo, months; PSQ-T, Pain Sensitivity Questionnaire-Turkish; VAS, visual analog scale; BPI, Brief Pain Inventory; BDI, Beck Depression Inventory; BAI, Beck Anxiety Inventory; PCS, Pain Catastrophizing Scale; SD, standard deviation.
Discussion
In this study, the PSG-G was translated into Turkish language. The values obtained in this study were in close agreement with those obtained using the PSQ-G. The results of this study prove the validity of the PSQ-T (4.7) as a measure of pain sensitivity that is equivalent to the original PSQ-G (3.6). The adult population in this study included all age groups, and no difference was detected by age or sex in PSQ-T scores. The absence of a relationship between PSQ-T scores and sex was similar to findings obtained using the German, English, and Chinese versions of the PSQ. 4,15,24 This shows that the PSQ can be applied to adult patients of any age or sex. Because the use of drugs does not affect pain sensitivity, patients who were taking different medications were included in our study. The PSQ-T minor and moderate values attained in this study were in close agreement with the findings reported by Ruscheweyh et al. 15 using the original German version, but were not in accordance with scores using the English version reported by Sellers et al. 4 Differences between our findings and the PSQ-E may be related to the homogeneity of our group, which did not include patients of different ethnicities. It has been reported in previous studies that there is no correlation between the duration of chronic pain and PSQ values.4,15 Although there was a correlation between the PSQ-T and pain in this study, no relationship was detected between the PSQ-T and pain duration. This may indicate that innate sensitivity and not the duration of pain is an important factor in pain perception.
Psychiatric conditions such as anxiety and depression can be expected to affect pain perception. However, similar to the original German and the English versions, PSQ values did not show any correlation with scores for anxiety and depression in our study. This may be because we excluded patients with major psychiatric disorders. The absence of a correlation between PSQ-T and PCS, in contrast to other studies, may be owing to cultural differences in pain catastrophizing in general. There are a variety of factors affecting PCS in the Turkish population. 48 Among cultural factors, the inability to determine education level among our participants is a limitation of our study. The detection of a correlation between the PSQ-T and BPI increases the reliability of the questionnaire and shows that patients can conduct questionnaire assessments independently of their psychological state. A significant correlation was observed between BPI scores and the PSQ in the English version, similar to the original German version, in both healthy patients and those with chronic pain. VAS 1 and VAS 2 scores obtained using standard subcutaneous infiltration methods with lidocaine as a pain stimulus (infiltration of the hand and infiltration in the procedure area, respectively) were found to be correlated with PSQ-T scores in our study, independently of psychological cofactors, which is similar to the report by Sellers et al. 4 The correlation with BPI pain score and BPI interference score also showed that the questionnaire results were not affected by the patients’ psychological status. The higher correlation between PSQ-T and VAS 1 and VAS 2 scores may indicate that the PSQ-T is an independent and important predictor of pain sensitivity and may be used for the interpretation of pain perception prior to intervention.
This study has some limitations. First, the educational levels of participants were not recorded. It would be beneficial to examine the correlation of educational level with understanding of the PSQ among Turkish people, although this has not been examined using the original PSQ-G version. Second, our study included a large number of women, which might have resulted in higher PSQ scores. Because patients with chronic pain were chosen as the study population, women are epidemiologically more represented than men. 49
Conclusion
The correlations between pain values and the Turkish version of the PSQ obtained in this study, together with other parameters, were found to be similar to those obtained with the PSQ-G. The present findings provide evidence of the validity of the PSQ-T in the assessment of patients with chronic pain.
Disclosure
This study was orally presented at the 15th National Pain Congress, Antalya, Turkey.
Footnotes
Acknowledgement
The authors would like to acknowledge R. Ruscheweyh for developing the original German version of the PSQ.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
