Abstract
Objective:
Endometriosis is a chronic gynecological disease associated with inflammation and severe pelvic pain in 6%–10% of women of reproductive age. Although the pathophysiology and management of endometriosis are currently understood, further research is needed to develop new diagnostic methods, particularly those involving inflammatory pathways, which could guide targeted therapies. This study aims to analyze the differences in levels of Chitinase-3-like protein 1 (CHI3L1) and Cyclooxygenase-2 (COX-2) in the walls of endometriosis cysts compared to non-endometriosis cysts (controls) and to evaluate the correlation of CHI3L1 with COX-2 and menstrual pain level using the Visual Analog Scale (VAS).
Methods:
This observational analytical study utilized a cross-sectional design and included 64 samples divided into 2 groups: 32 endometriosis cysts and 32 non-endometriosis cysts. Diagnosis is made through ultrasound and laparoscopic surgery or laparotomy, with histopathological confirmation. Samples were taken from the cyst walls and analyzed for CHI3L1 and COX-2 levels using the Enzyme-linked immunosorbent assay (ELISA) method. Statistical analysis was conducted using the Mann-Whitney test and Spearman’s Rank correlation.
Results:
The results showed that CHI3L1 and COX-2 levels in the walls of endometriosis cysts were significantly higher than in controls (
Conclusion:
This study concludes that CHI3L1 may play a role in the pathogenesis of endometriosis through an inflammatory pain pathway involving COX-2 and prostaglandins.
Introduction
Endometriosis is a chronic gynecological disorder characterized by the presence of endometrial-like tissue, including glands and stroma, outside the uterus.1,2 This condition is known to exhibit estrogen dependence and progesterone resistance, which have been linked to chronic inflammation, severe pelvic pain, and infertility. 2 Endometriosis is significant not only from medical and social perspectives but also from an economic standpoint. The endometriosis-associated pain and bodily dysfunction exacerbate the quality of life and reduce professional productivity, thereby contributing to the economic burden of a country.3,4 The prevalence of endometriosis can affect 10%–15% of women of reproductive age, with 35%–50% experiencing pelvic pain and infertility. 5 Endometriosis is also reported to occur in 70% of women with chronic pelvic pain. 6
One type of pain associated with endometriosis is inflammatory pain.4,7 In endometriosis, the inflammatory response is mediated by cytokines and prostaglandins, which signal the inflammatory process at the implantation sites, laying the foundation for new vascularization and the formation of new fibrous tissue. The most common type of endometriosis, affecting approximately 17%–44% of women, is endometrioma. Patients with endometriomas typically experience a more severe stage of endometriosis compared to the stages one or two endometriosis. 8 Therefore, it can be said that inflammatory mediators also contribute to angiogenesis and interact with sensory neurons, inducing varying pain thresholds depending on the state and location of the disease. 9
In endometriosis, inflammatory mediators include Prostaglandin E2 (PGE2), Vascular Endothelial Growth Factor (VEGF), tumor necrosis factor-α (TNF-α), Nerve Growth Factor (NGF), and interleukins (IL). 4 Additionally, Chitinase-3-like protein 1 (CHI3L1/YKL-40), a new inflammatory biomarker, is also secreted by macrophages and neutrophils in inflamed tissues, such as in endometriosis.10–12 CHI3L1 glycoprotein originates from various cells, such as macrophages, neutrophils, vascular smooth muscle cells, bone cells, endothelial cells, cancer cells, and others.13,14 The function of CHI3L1 includes regulating cell proliferation, apoptosis, differentiation, invasion, tissue remodeling, angiogenesis, and cancer metastasis, primarily in infection and inflammation conditions. Several cytokines, such as IL-1, IL-13, IL-6, and TNF, can stimulate CHI3L1 expression. Meanwhile, CHI3L1 can regulate various cytokines, including IL-6, IL-8, IL-12, IL-18, Interferon-gamma (IFN-γ), and TNF-α.12,14
Preliminary studies related to CHI3L1 in endometriosis have been conducted, but the relationship between CHI3L1 and inflammation-induced pain, hormonal factors in endometriosis, and other cytokines such as Cyclooxygenase-2 (COX-2) has not been fully explored. COX-2 expression in ectopic endometrial cells is significantly higher compared to eutopic endometrium. 15 In endometriosis, there is a notable increase in aromatase enzyme, which converts androgens into estrogens, compared to normal endometrial tissue due to autonomic production by the endometriotic cells.10,16 Under the influence of estrogen, produced by the aberrant activity of aromatase, endometriotic implants can proliferate and increase the expression and release of pro-inflammatory factors. The estrogen generated by aromatase modulates the immune system by stimulating the release of inflammatory cytokines such as TNF-α, IL-1β, IL-6, and IL-13. These cytokines, in turn, stimulate macrophages to release inflammatory proteins such as CHI3L1, which activate cytokines and chemokines, further activating COX-2.14,16,17 The activation of COX-2 converts arachidonic acid into PGE2. 18 The increased production of PGE2 induces inflammation, which in turn activates steroidogenic acute regulatory protein and aromatase, leading to further estrogen production. 17 This estrogen then provides positive feedback, stimulating local hyperestrogenism and elucidating the estrogen-dependent relationship with inflammation in the pathogenesis of the disease. 19
Currently, there is no research connecting CHI3L1 in endometriosis with COX-2 inflammatory mediators and menstrual pain graded by Visual Analog Scale (VAS) on the endometriosis cyst wall. This has led the authors to investigate differences in CHI3L1 levels in endometriosis cases and explore potential correlations between increased CHI3L1 levels and COX-2 and menstrual pain level (measured by VAS) as part of endometriosis pathogenesis. Understanding the molecular mechanisms involved in endometriosis pathogenesis comprehensively may support the future development of CHI3L1 or COX-2 targeted therapies, offering potential new strategies for disease management. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Methods
Study design
This research is an analytical observational study employing a cross-sectional design, analyzing differences and correlations between independent and dependent variables simultaneously. The study subjects were divided into two groups: those with endometriosis cysts and those with non-endometriosis cysts. The researcher conducted assessments of CHI3L1, COX-2, and menstrual pain level at a single evaluation time.
Sampling was performed using consecutive sampling, whereby patients were selected in the order of their arrival, provided they met the inclusion criteria, did not meet the exclusion criteria, and fulfilled the minimum sample size requirement. The minimum sample size was calculated using previously reported mean differences in CHI3L1 and COX-2 levels between endometriosis and non-endometriosis cysts. With a two-sided α = 0.05 and 80% power, data from Tuten et al.
10
indicated a required minimum of 8 subjects per group for CHI3L1, while estimates from Ota et al.
20
suggested 13 subjects per group for COX-2. For correlation analysis, a minimum of 29 endometriosis patients was needed to detect an expected correlation coefficient of
This study was conducted with the approval of the Padjadjaran University Ethics Committee under the following registration number DP.04.03/D.XIV.6.5/105/2024. Written informed consent was obtained from patients regarding the use of their anonymized data and tissues for research purposes. All methods involving human participants were performed in accordance with the ethical standards outlined in the Declaration of Helsinki (1975) and the relevant ethical standards of the institutional and/or national research committee. The manuscript has been reported in line with the Strengthening the Reporting of Observational Studies in Epidemiology guideline throughout the whole process. 21
Data collection
For endometriosis patients, we recruited patients with endometriosis between the ages 20–40 years old, with normal Body Mass Index (BMI) according to WHO criteria for Asia-Pacific (18.5–24.9 kg/m2), was diagnosed with endometriosis cysts based on transvaginal or abdominal USG and confirmed histopathologically, having undergone laparoscopic and laparotomy procedures, and consented to the study protocol. As for the control group, we recruited non-endometriosis (dermoid, serous, and mucinous cysts) patients with a similar age range and BMI, having undergone laparoscopic and laparotomy procedures. For both groups, we excluded patients with a history of receiving hormonal therapy within the last 3 months (progestin-only, estrogen + progesterone (E + P) combinations and their derivatives, gonadotropin-releasing hormone (GnRH) agonists) and anti-inflammatory therapy such as steroids and Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) within the last 3 months, with other infections, ovarian malignancy, or a history of immunological, endocrine, or metabolic diseases. Dysmenorrhea, or menstrual pain, was defined as pain during the menstrual cycle, typically located in the lower abdomen and potentially radiating to the inner thighs and back. The severity of dysmenorrhea was evaluated using the VAS, ranging from 0 to 10. 22
Tissue samples (1 cm³) were collected from each patient’s surgical specimen. Samples were rinsed, weighed, and homogenized in phosphate-buffered saline, followed by centrifugation to collect the supernatant. The CHI3L1 and COX-2 protein levels were measured using ELISA, the Human Chitinase 1/Chitotriosidase ELISA Kit, 96 wells (Cat. No. BZ-08120454-EB), and COX-2 levels were measured using the Human COX-2 ELISA Kit, 96 wells (Cat. No. BZ-08120870-EB). Both assays were conducted at the Molecular Genetics Laboratory, Faculty of Medicine, Universitas Padjadjaran/RSUP Dr. Hasan Sadikin, Bandung. The detection sensitivity for each kit was within the manufacturer’s specifications, with intra- and inter-assay coefficients of variation of <10% and <12%, respectively. Samples were incubated with specific antibodies, washed, and then treated with substrates. The absorbance was measured using a microplate reader at 450 nm. The results were then compared with standard curves to normalize and obtain quantitative data.
Statistical methods
Data collected were analyzed using SPSS version 27 (IBM). with a 95% confidence level and a significance threshold of
Results
A study investigating the correlation between CHI3L1 levels and COX-2, as well as the menstrual pain measured by VAS in endometriosis cysts, was conducted involving 32 patients who attended several hospitals in Bandung from April to July 2024. Additionally, a comparison was made with 32 patients with non-endometriosis cysts (8 mucinous cysts, 9 serous cysts, and 15 dermoid cysts), serving as the control group. Cyst walls were obtained from patients who underwent either laparotomy or laparoscopy. Data recorded for all subjects included demographic characteristics such as age, BMI, as well as CHI3L1 and COX-2 levels in both endometriosis and non-endometriosis cyst walls.
The study addressed differences in CHI3L1 and COX-2 levels between the endometriosis group and controls (non-endometriosis), and the correlation of CHI3L1 levels with COX-2 levels and menstrual pain level (VAS) in endometriosis cyst walls. Table 1 provides general information on the characteristics of subjects from both research groups, including age, BMI, parity, infertility, laterality, locularity, cyst diameter, and menstrual pain level as indicated by VAS scores. Most of the study subjects in the endometriosis and non-endometriosis groups are homogeneous, showing no significant differences (
Characteristics of research subjects.

BMI: Body mass index; VAS: Visual Analog Scale; SD: Standard deviation.
Mann-Whitney test. **Chi-Square.
Comparison of CHI3L1 and COX-2 levels between endometriosis and non-endometriosis cyst walls is detailed in Table 2. Significant differences were observed in CHI3L1 levels between the endometriosis cyst group and the non-endometriosis cyst group (
Comparison of CHI3L1 and COX-2 between endometriosis and non-endometriosis groups.
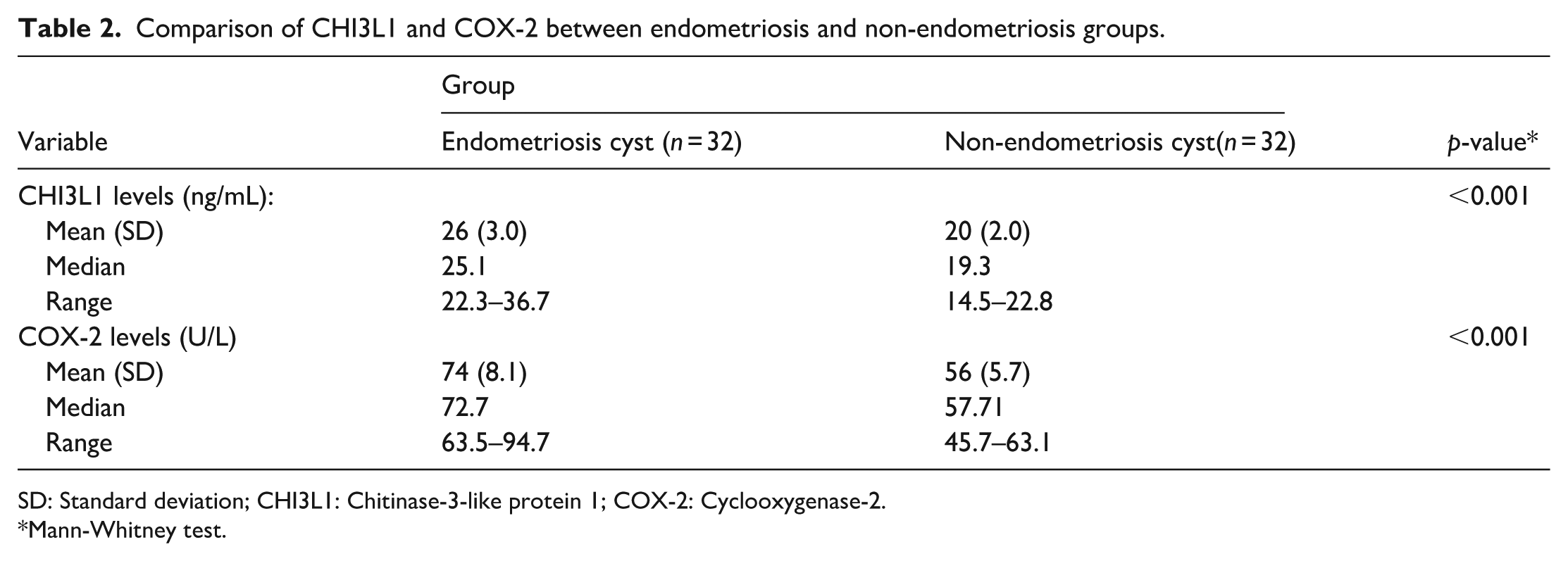
SD: Standard deviation; CHI3L1: Chitinase-3-like protein 1; COX-2: Cyclooxygenase-2.
Mann-Whitney test.
The correlations between CHI3L1 and COX-2, as well as between CHI3L1 and menstrual pain level (VAS) in endometriosis cyst walls, are illustrated in Figures 1 and 2. The results of the statistical analysis using Spearman’s rank correlation test at a 95% confidence level, which examines the correlation between CHI3L1 levels and COX-2, as well as menstrual pain level (VAS) in endometriosis cyst walls, are presented in Table 3. The correlation analysis between CHI3L1 levels and COX-2 levels yielded a correlation coefficient (rs = 0.767), indicating a significant positive correlation. This suggests that as CHI3L1 levels increase, COX-2 levels also significantly increase. The correlation between CHI3L1 levels and menstrual pain level (VAS) showed a strong and statistically significant positive correlation (rs = 0.511), meaning that higher levels of CHI3L1 are associated with greater menstrual pain (VAS).

Correlation between CHI3L1 levels and COX-2 levels in the wall tissue of endometriosis cysts.

Correlation between CHI3L1 levels in the wall tissue of endometriosis cysts and menstrual pain level (VAS).
Correlation between variables in endometriosis cyst wall.

VAS: Visual Analog Scale; CHI3L1: Chitinase-3-like protein 1; COX-2: Cyclooxygenase-2.
Rank Spearman correlation test.
Discussion
Endometriosis is a chronic gynecological condition characterized by the presence of endometrial-like tissue outside the uterus, leading to chronic inflammation. 6 A key mechanism influencing the development of endometriosis is retrograde menstruation. Retrograde menstruation, as proposed in the pathogenesis theory of endometriosis, involves the process of endometrial tissue invaginating into the ovaries and the peritoneal cavity. Retrograde menstruation involves inflammatory processes, macrophage activation, and the release of pro-inflammatory cytokines such as CHI3L1, COX-2, and PGE2 within the endometrial tissues that are shed and subsequently implanted at the ectopic sites. Chronic inflammation plays a crucial role in the pathogenesis of endometriosis due to the presence of ectopic lesions producing excessive prostaglandins, cytokines, and chemokines.23,24
Characteristics of study subjects
The study involved 64 female subjects aged 20–40 years, divided into 2 groups: 32 with endometriosis cysts and 32 with non-endometriosis cysts. All participants had a normal BMI (18.5–24.9 kg/m²), were diagnosed via transvaginal and abdominal ultrasound, and confirmed by histopathology. Age and BMI were controlled to avoid hormonal influences from puberty and premenopause.
25
The average age of women with endometriosis was 32 years (
The study also indicated that most endometriosis cysts were unilateral, with only a few being bilateral (
Menstrual pain was assessed using the VAS, showing scores of 7–10 for the endometriosis group and 0–2 for the non-endometriosis group (
Comparison of mean CHI3L1 levels between endometriosis cyst and non-endometriosis cyst groups
This study found that mean CHI3L1 levels were higher in the endometriosis cyst group (26 ± 3.0) compared to the non-endometriosis cyst group (20 ± 2.0), with statistically significant differences (
Comparison of mean COX-2 levels between endometriosis cyst and non-endometriosis cyst groups
The study showed that the mean COX-2 level was higher in the endometriosis cyst group (74 ± 8.1) compared to the non-endometriosis cyst group (56 ± 5.7), with a significant difference (
Correlation between CHI3L1 and COX-2 levels in endometriosis cyst walls
The study identified a strong positive correlation (
Correlation of CHI3L1 levels in endometriosis cyst walls with menstrual pain level
This study shows a notable positive correlation (
Conclusion
This study concludes that CHI3L1 and COX-2 levels are significantly higher in endometriosis cyst walls compared to non-endometriosis cyst walls. In addition, there is a strong positive correlation between CHI3L1 and COX-2 levels within endometriosis cyst walls. Furthermore, CHI3L1 levels in endometriosis cyst walls are positively correlated with menstrual pain level as measured by the VAS.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251409312 – Supplemental material for Elevated CHI3L1 and COX-2 levels correlate with menstrual pain severity in endometriosis
Supplemental material, sj-docx-1-smo-10.1177_20503121251409312 for Elevated CHI3L1 and COX-2 levels correlate with menstrual pain severity in endometriosis by Dewi Karlina Rusly, Wiryawan Permadi, Hartanto Bayuaji, Hanom Husni Syam, Tita Husnitawati Madjid, Mulyanusa Amarullah Ritonga and Muhammad Rusda in SAGE Open Medicine
Footnotes
Ethical considerations
Ethical approval for this study was obtained from the Health Research Ethics Committee of Hasan Sadikin General Hospital (DP.04.03/D.XIV.6.5/105/2024).
Consent to participate
Written informed consent was obtained from patients regarding the use of their anonymized data and tissues for research purposes. All methods were performed in accordance with the relevant guidelines and regulations.
Consent for publication
Written informed consent was obtained from all subjects before the study.
Author contributions
D.K.R. conceived the study under the supervision of W.P. and H.B. D.K.R. collected the data. W.P. and H.B. analyzed the data. D.K.R. and H.B. drafted the manuscript. H.H.S., T.H.M., M.A.R., and M.R. reviewed and edited the article. All authors approved this version of the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article. Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
