Abstract
Objective
To assess the clinical and laboratory predictors of venous thromboembolism (VTE) in patients with sickle cell anaemia (SCA) and its relationship to morbidity and mortality.
Methods
This retrospective case–control study analysed data from patients with SCA that experienced VTE compared with matched control patients with SCA but no VTE (2:1 ratio).
Results
A total of 102 patients with SCA were enrolled (68 cases with VTE and 34 controls). Amongst the 68 cases (median age, 29.5 years), 26 (38.2%) presented with isolated pulmonary embolism (PE). A higher prevalence of splenectomy (73.5% versus 35.3%) was observed in the cases compared with the controls. A significantly higher prevalence of central venous catheter (CVC) insertion (42.6% versus 8.8%) was observed in the cases compared with the controls. High white blood cell counts, serum lactic dehydrogenase (LDH), bilirubin and C-reactive protein (CRP) and low haemoglobin (Hb) and HbF were significant risk factors for VTE. Forty-two cases (61.8%) developed acute chest syndrome, 10 (14.7%) had a stroke and seven (10.3%) died.
Conclusions
VTE in patients with SCA has a high impact on morbidity and mortality. PE was the leading presentation of VTE, with CVC insertion, high LDH, bilirubin, CRP and white blood cell counts along with low Hb and HbF constituting other significant risk factors.
Introduction
Sickle cell anaemia (SCA) is a common and life-threatening haematological disorder that affects millions of people worldwide.1,2 In Oman, SCA is a major public concern, where 5.7% of the population carry the sickle cell gene and 0.3% Omani people have SCA.3,4 It is a monogenic recessively inherited disorder with abnormal beta-globin alleles, where glutamic acid is replaced by valine in the haemoglobin subunit beta (
Sickled erythrocytes occlude blood vessels and disrupt blood flow, causing repeated ischaemia and inflammation. This vessel occlusion is a complex process involving an interaction between the sickle cell, vessel endothelial cells, platelets and other blood components.9,10 The adherence of the sickle cell to the vessel endothelium results in activation of the endothelial cells and secretion of inflammatory mediators and biomarkers that increase platelet activation and induce blood coagulation. 10
Patients with SCA frequently develop acute pulmonary complications, including acute chest syndrome (ACS), asthma and a higher risk for thromboembolism.11–13 Several mechanisms have been suggested to explain this hypercoagulability including platelet reactivity, with elevated plasma concentrations of platelet-derived microparticles, hydroxyl radical formation, fibronectin and thrombospondin elevation, deficient proteins S and C, and elevated levels of factor VIII and tissue factor.14,15
Several studies in SCA patients have recorded a high prevalence of venous thromboembolism (VTE).16–18 These studies found that the incidence of VTE events in SCA patients was higher than in the non-SCA population.16–18 Also, several laboratory parameters were abnormal in the patients with VTE such as heterozygosity for factor V Leiden, elevated platelet counts as well as high factor VIII: C levels. 18 A previous study that investigated the prevalence of pulmonary embolism (PE) and deep vein thrombosis (DVT) in SCA patients reported that 0.44% of patients had a discharge diagnosis of PE and DVT, as compared with 0.12% of non-SCA patients, indicating a significantly high prevalence. 18 A previous study reported that the cumulative incidence of VTE was 17.1% for severe sickle cell disease (SCD) patients (hospitalized ≥3 times a year) versus 8.0% for the matched asthma controls and was associated with increased mortality. 19 The Cooperative Study of Sickle Cell Disease on 1523 SCD patients aged ≥15 years with 8862 years of follow-up reported an incidence rate for first VTE of 5.2 events/1000 person-years (95% confidence interval [CI] 3.8, 6.9) with a cumulative incidence of 11.3% (95% CI 8.3, 15.3) by age 40 years. 20 Thus, it is apparent that SCA is a risk factor for DVT and PE.
This current study aimed to assess the clinical and pathological characteristics of VTE amongst patients with SCA in order to determine the predisposing factors and determine its impact on morbidity and mortality.
Patients and methods
Patient population
This retrospective case–control study enrolled consecutive patients presenting with thromboembolic complications including unprovoked PE, upper and lower extremity DVT, portal vein thrombosis (PVT), superior vena cava thrombosis, right atrial thrombosis and cerebrovascular accident (CVA) in the Department of Haematology, Sultan Qaboos University Hospital, Muscat, Oman between January 2007 and January 2018. The inclusion criterion for the case group was patients with SCA that were admitted with venous thrombosis or thromboembolic complications. Patients that had SCA without thrombosis that attended the outpatient clinic of the Department of Haematology, Sultan Qaboos University Hospital for routine appointments and prescription medication or were admitted for pregnancy formed the control group in a ratio of 2:1 due to the consecutive enrolment during the study period. None of the patients included in the study were on a regular transfusion protocol for the prevention of SCA complications. All the patients in this study were confirmed to have SCA by high performance liquid chromatography. All patient data were collected from the hospital information system at the Sultan Qaboos University Hospital.
The Medical Research Ethics Committee of the Sultan Qaboos University Hospital approved the study (no. MREC 1491). Patient consent was not required due to the retrospective design of the study. The reporting of this study conforms to STROBE guidelines. 21
Data collection
The data collected from the hospital information system included age, sex and SCA-associated complications, such as frequency of vaso-occlusive crisis (VOC), ACS, splenic sequestration and febrile episodes. The VOC frequency was categorized as high (>6 VOC/year), moderate (3–6 VOC/year) and low frequency (1–2 VOC/year). ACS was defined as patients presenting with fever and/or respiratory symptoms, accompanied by new infiltrates on the chest radiograph. Steady state was defined as the absence of fever or VOC in the previous ≥4 weeks in the patient. Amongst the cases and controls, asplenia was defined as splenic atrophy as confirmed by ultrasound/computed tomography (CT) scan and/or past surgical splenectomy. All central venous catheter (CVC) lines used in these patients included peripherally inserted central lines, PORT-A-CATH®, regular central line in the neck and femoral lines. The type of thrombosis, CVC insertion, hydroxyurea (HU) use and pregnancy details were recorded as clinical parameters. Laboratory data were recorded at baseline and during the thromboembolic episode in the cases. Haematological parameters included haemoglobin (Hb) level, white blood cell (WBC) and platelet counts, reticulocyte %, HbS% and HbF%. Biochemical parameters included serum total bilirubin, serum lactic dehydrogenase (LDH), serum vitamin B12, serum fibrinogen, D-dimer and C-reactive protein (CRP). In addition, the radiological results of magnetic resonance imaging, CT angiography and abdominal ultrasound were recorded. The impact of VTE on morbidity and mortality in all patients was determined. VTEs were managed using a standard international protocol based on the American College of Chest Physicians guidelines. 20
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA). Normally distributed continuous data are presented as mean ± SD. Continuous data that were not normally distributed are presented as median with interquartile range (IQR). Mann–Whitney
Results
This retrospective case–control study enrolled 102 patients with SCA. Table 1 shows the demographic and clinical characteristics of the cases (
Demographic and clinical characteristics of patients with sickle cell anaemia with (case group,

Data presented as median (interquartile range) or
aχ2-test was used to compare the categorical data between the case and control groups; NS, no significant between-group difference (
ACS, acute chest syndrome; VOC, vaso-occlusive crisis.
Amongst the haematological parameters (Table 2), there were significant differences between the two groups at baseline in Hb, WBC count, reticulocytes, HbS% and HbF% (
Laboratory parameters in patients with sickle cell anaemia with (case group,

Data presented as median or
aMann–Whitney
The pattern of thromboembolism in the study population is shown in Table 3. Isolated PE and upper and lower limb DVT were the predominant presentations in the case group. Among the risk factors for thromboembolic complications, the rate of CVC insertion was significantly higher in the case group compared with the control group (
Comparison of events and outcomes in patients with sickle cell anaemia with (case group,
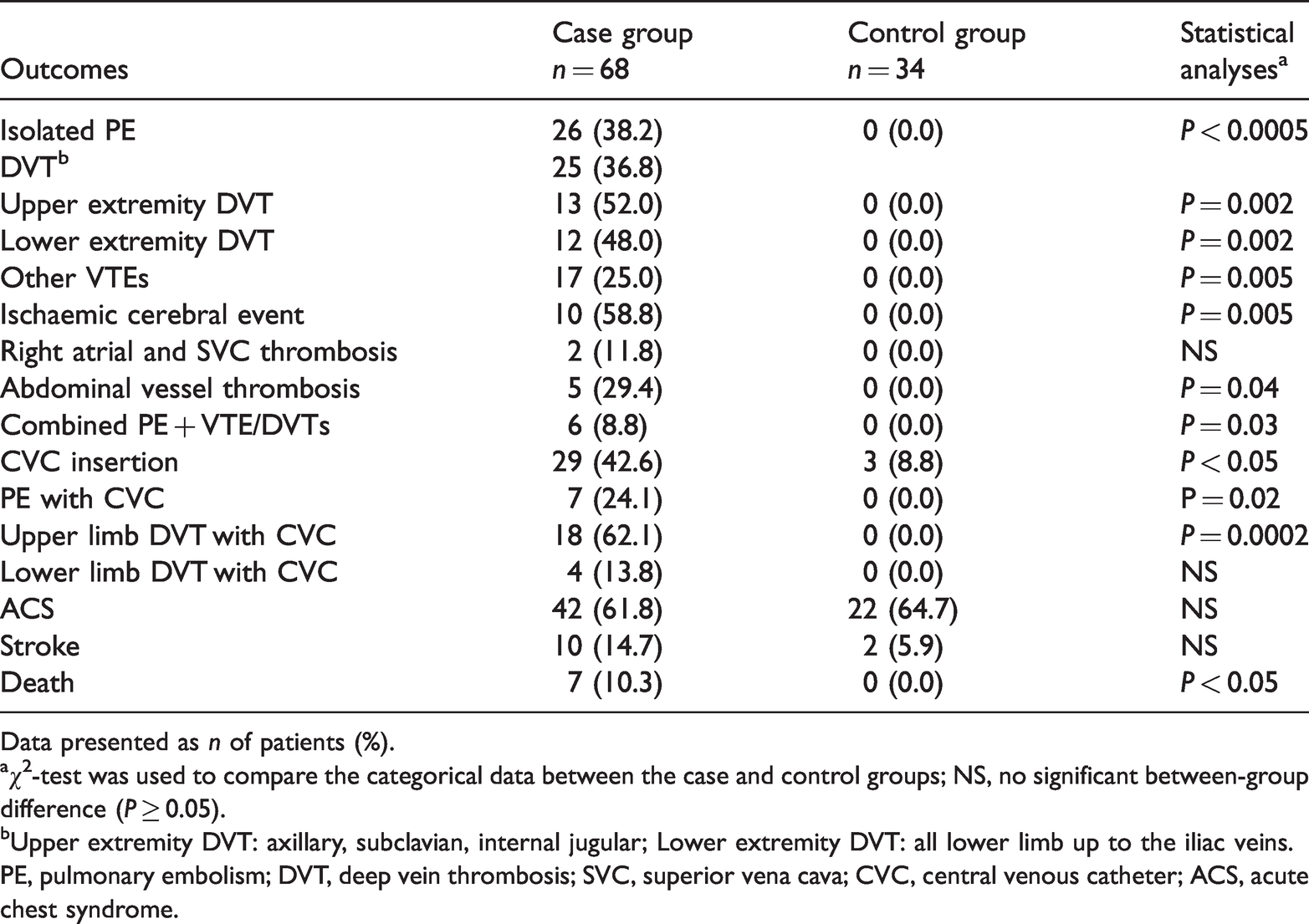
Data presented as
aχ2-test was used to compare the categorical data between the case and control groups; NS, no significant between-group difference (
bUpper extremity DVT: axillary, subclavian, internal jugular; Lower extremity DVT: all lower limb up to the iliac veins.
PE, pulmonary embolism; DVT, deep vein thrombosis; SVC, superior vena cava; CVC, central venous catheter; ACS, acute chest syndrome.
There were moderate correlations between VTE and PE and CVC insertion, with correlation coefficients of r = 0.35 and r = 0.3, respectively.
Discussion
Several studies have shown that VTE is one of the common complications of SCA with a cumulative incidence that increases with the age, with the mean age ranging from 24 to 37 years.18,19,22 Researchers have also compared SCA with inherited thrombophilia with a thrombosis prevalence around 25%, which is similar to those seen in a family with strong thrombophilic defects. 23 In this current retrospective study, the median age of SCA patients that developed VTE was 29.5 years (range, 12–80 years), which was not significantly different from the median age of the control group (33.0 years) and similar to previous research. 19 However, it was lower than the median age of 68 years for thrombosis seen in the general population. 24 In addition, the current study found that sex was not a risk factor for VTE in patients with SCA, but the prevalence data indicated a preponderance for females, probably driven by pregnancy, although it did not reach statistical significance. These data were similar to another study from the US in which females were more at risk for VTE than males, again, not in keeping with the observations in the general population. 19
The spectrum of thrombosis in the current study cohort revealed that DVT/VTEs (
One of the aims of this current study was to identify the predisposing factors that increase the risk of developing VTE in patients with SCA. There are a number of potential predisposing factors to thrombosis in SCA, including frequent hospitalizations, raised inflammation and hypercoagulable state associated with this disease, however, those factors are not well studied. Although the increased frequency of VOC is not a direct risk factor, it could be considered as a surrogate factor, since it leads to frequent and prolonged hospitalization of SCA patients, which is a risk factor for thromboembolism. This current study found that patients with more frequent VOCs had a significantly higher prevalence of VTE. This may again reflect disease severity and has been seen in other research. 19 This current study found that a high percentage of the VTE patients had a history of ACS (82.4%), implying that it did predispose to the occurrence of VTE, and such patients suffered from ACS as a complication of PE. Furthermore, previous research has demonstrated that PE is considered to significantly predispose for ACS, with PE episodes developing during ACS, 25 which is again similar to these current observations. However, it is difficult to conclude (from the current data), whether PE is a cause or a consequence of ACS. 28 It is believed that patients with SCA are more prone to infection as a result of immune impairment because of asplenia. 29 Infection, by promoting an increase in inflammatory cytokines, can provoke thrombosis, especially in asplenic patients. 30 Furthermore, asplenia in haemolytic diseases including SCA was reported to be associated with an increased risk of thrombosis, as seen in this current study. 16
Central venous catheter insertion has a significant correlation with VTE episodes. 15 CVC is generally required for many patients with SCA and it has been shown to be an independent risk factor for VTE.15,31 Furthermore, prolonged use of catheter implantation increases the risk of thrombosis,17,31 as seen in the current study cohort. Among the 68 patients with VTE in the case group, 29 patients had CVC at the time of VTE, including 18 with upper limb DVT, seven with PE and four had lower limb DVT.
Pregnancy is significantly correlated with VTE as the rate of VTE during pregnancy and puerperium was 1.5–2.5-times greater than in non-pregnant SCA women.32,33 It is reported that pregnancy-related VTE in females with SCA is 1.5–5-times greater than pregnancy-related VTE in the general population.
32
The current study demonstrated that 27.3% of cases compared with 16.7% of controls had a history of pregnancy. Interestingly, PE was reported in 16 (36.4%) of females in the case group, whereas none of the three pregnant females in the control group developed PE (
The use of HU therapy over the last 30 years has been shown to increase HbF in SCA patients leading to improved clinical efficacy. 35 It is associated with a reduction in the levels of biomarkers of hypercoagulability in SCA patients and is likely to reduce the prevalence of VTE. 36 In this current study, the median HbF levels were significantly higher in the control group compared with the case group. Furthermore, a higher percentage of control patients (61.8%) were treated with HU compared with 52.9% in the case group, which might explain the higher median HbF in the controls. Although there are contradicting results on the impact of HU on VTE prevention, these current results were in inconclusive with respect to a positive role of HU in VTE prevention, knowing that that HbF inhibits intracellular HbS polymerization.37,38
In the current study population, there were elevations in the median WBC count, LDH level, bilirubin and CRP in the case group from baseline to VTE episode; and the baseline levels were significantly different compared with the control group. Higher WBC count and elevated LDH level are reported to be risk factors for VTE in SCA. 39 Biomarkers are helpful in identifying different pathophysiological mechanisms in SCA, including hypercoagulability, haemolysis, nitric oxide depletion, inflammation, oxidative stress, vasculopathy, reperfusion injury and tissue necrosis with infarction.7–10,19,25Over half of the patients in the case group in the current study had elevated D-dimer (54.4%) at the VTE episode and the CRP levels were significantly higher at the VTE episode compared with the controls at baseline, which were presumably due to an acute phase reaction, often seen in patients with thromboembolism and in sickle cell crisis. 33
Among the clinical complications of VTE in this current group of patients, a large majority of the patients in the case group developed ACS (61.8%), which was consequential of the development of PE; and 10 patients (14.7%) developed stroke as a manifestation of venous and arterial thrombosis within cerebral vessels. In 306 autopsies of SCA patients, a previous study reported that 9.8% died due to stroke and 4.9% died due to pulmonary emboli/thrombi. 40 However, another autopsy study from Nigeria reported that the mortality rate from fatal thromboembolic complications was 37%. 41 Thus, although infections and multiorgan failure are major causes of death in SCA, SCA with VTE is still associated with a significant mortality.40,41 These data support the regular use of prophylactic low molecular weight heparin for the purpose of VTE prevention and improved VOC management.42,43
In conclusion, acknowledging the limitation of the retrospective nature of this current study, VTE was one of the common complications of SCA and it occurred at a younger age than the general population. PE showed a higher prevalence in patients with SCA. Several risk factors contributed to the development of VTE in patients with SCA such as CVC insertion and pregnancy. In addition, coagulation activity in patients with SCA was enhanced with abnormal activation of the fibrinolytic system and increased levels of WBCs, LDH, bilirubin and CRP. This current study demonstrated that thromboembolic complications in patients with SCA had a high impact on morbidity and mortality and calls for the use of prophylactic anticoagulation in all patients with SCA during their hospital stay.
Footnotes
Acknowledgements
We wish to thank the hospital administration for the use of hospital materials in this study.
Author contributions
All authors made substantial contributions. They have all seen and approved the final version of the manuscript. S.A., A.R.A., S.R.A, S.Y.A., N.F., S.K.B. and A.V.P. were fully involved in the conception and design of the study, recruitment and care of patients, acquisition of data, or analysis and interpretation of the data. S.A. and A.V.P. were instrumental in the drafting the article and critical appraisal before submission.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
