Abstract
Mesenteric teratoma is a rare extragonadal teratoma. Gliomatosis peritonei (GP) is mature glial tissue implanted into the peritoneum's surface and is mainly accompanied by ovarian teratoma. Only a few cases of gliomatosis have occurred in the extraperitoneum. We present a rare case of a 3-year-old boy who presented with extratesticular GP after excision of an immature mesenteric teratoma at 2 months old. After the extratesticular mass was excised, we found ductile tissue on the surface of the terminal spermatic cord and epididymis. Some ductile tissue of the epididymis was removed and sent to a laboratory for a pathological examination. The mass and the ductile tissue of the epididymis had a hard consistency. The pathological diagnosis was extratesticular gliomatosis. Complete surgical resection of the teratoma and GP is helpful for identifying the presence of malignant lesions and for preventing malignant transformation. However, characteristics of GP lesions are extensive and they are difficult to completely remove. Moreover, GP is usually benign. Therefore, the residual GP tissue was not completely removed in our case. The child is still in good health, but requires lifelong follow-up. In conclusion, we report our experience of a rare case of extraperitoneal GP from an extragonadal teratoma.
Introduction
Gliomatosis peritonei (GP) is a rare disease, which involves extensive implantation of mature miliary glial tissue on the surface of the peritoneum, mainly accompanied by ovarian teratoma.1–3 Only a few cases of GP were associated with extragonadal teratomas. 1 , 4 , 5 Mesenteric teratoma, which is a type of extragonadal teratoma, is a rare tumor, especially in children. 6 Moreover, only a few cases of GP have occurred in the extraperitoneum. 1 , 7 , 8 We present a rare case of extratesticular GP after excision of an immature mesenteric teratoma.
Case report
A physical examination showed severe abdominal distention in a 2-month-old boy. A computed tomography (CT) scan showed an extremely large mixed density mass in the abdominal cavity. The mass was 12.9 × 11.3 × 7.7 cm in size, with mainly cystic components, multiple nodular calcification, and fat density, which suggested a teratoma (Figure 1). Tumor markers showed that the serum alpha-fetoprotein concentration was 2842 ng/mL and the β-human chorionic gonadotropin concentration was normal. Exploratory laparotomy showed a massive mass arising from the mesentery with a capsule, but it was adhered to the root of the mesentery. The mass had an irregular surface and alternating cysts and solid areas (Figure 2a and 2b). The histological diagnosis was an immature teratoma (grade III) (Figure 2c and 2d). Immunohistochemistry showed the following: B-cell lymphoma 2 (+), CD56 (+), CD99 (+), protein gene product 9.5 (−), synaptophysin (−), neuron-specific enolase (−), glial fibrillary acidic protein (GFAP) (−), neuronal nuclei (−), S-100 (focal, +), and Ki67 (95%+). Chemotherapy was not provided because these tumors have a good prognosis in the neonatal age group. The serum alpha-fetoprotein concentration was 389 ng/mL after surgery.

Abdominal computed tomography scan. Images show a large cystic, solid mass with punctate calcifications.
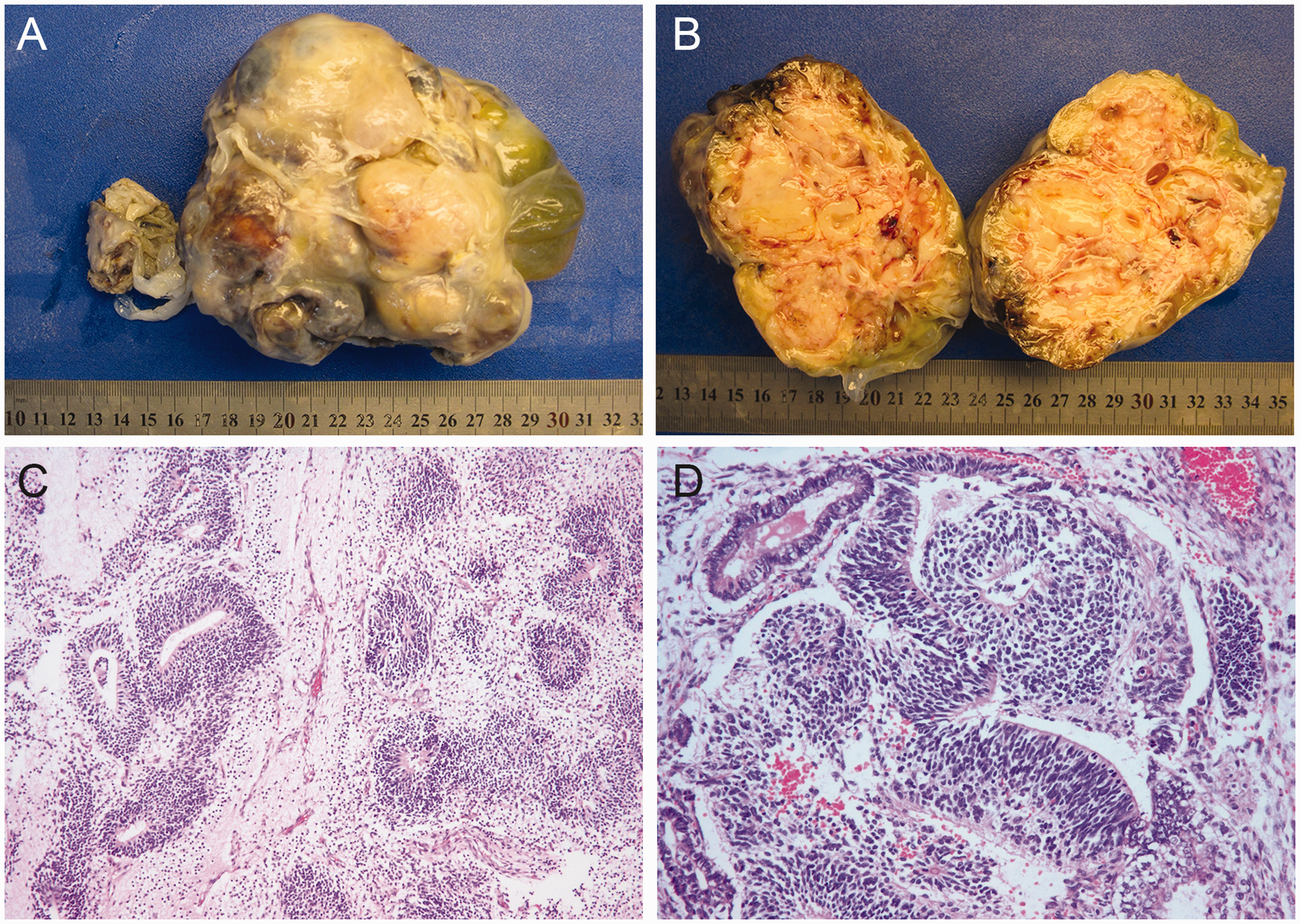
(a, b) Gross specimen of the mesenteric immature teratoma. (c, d) Microscopic findings of the immature teratoma show primitive neuroectodermal tubes. Sections were stained by hematoxylin and eosin.
A painless right scrotal mass was found at 3 years and 2 months. Magnetic resonance imaging (MRI) showed a mass in the right scrotum behind the testis with a clear boundary. The size of the mass was 10.2 × 10.4 × 10.0 mm, and the right spermatic cord was slightly thicker than the contralateral cord (Figure 3). An MRI scan of the abdomen showed no evidence of recurrence in the abdominal compartments. Serum alpha-fetoprotein and β-human chorionic gonadotropin concentrations were normal. Exploratory surgery showed that the mass was located in the posterior and lower part of the testis, and the boundary was not clear. After the mass was excised (Figure 4a), we also found that the terminal spermatic cord and epididymis did not touch the ductile tissue. Some ductile tissue of the epididymis was removed and sent for a pathological examination (Figure 4b). The mass and the ductile tissue of the epididymis had a hard consistency. A histopathological examination showed a scattered, nest-like, glial component (Figure 5a and 5b). The pathological diagnosis was extratesticular gliomatosis. An immunohistochemical examination showed the following: GFAP (+) (Figure 5c), S-100 (+) (Figure 5d), inhibin-a (−), vimentin (+), neurofilament-H (+), neuronal nuclei (+), Oct3/4 (−), and Ki67 (<3%+). No recurrence was observed at follow-up for 6 months.

Magnetic resonance imaging of the scrotum. Images show well-defined masses (red arrows) separate from the testis (red asterisk).

(a) Gross specimen of the excised extratesticular mass. (b) Gross specimen of tough white excised tissue on the surface of the epididymis.

(a) Microscopy of the gliomatosis shows a diffuse glial component (×50). (b) The glial component has a fibrillary background (×200). Sections were stained by hematoxylin and eosin. (c) The glial tissue (brown) shows positive immunostaining for glial fibrillary acidic protein (×100). (d) S-100-stained section showing positivity for glial components (×100).
Discussion
GP is a condition in which there is implantation of mature miliary glial tissue on the surface of the peritoneal membrane in patients with a mature or immature teratoma. Rare cases of extraperitoneal gliomatosis associated with extragonadal teratomas have been reported (Tables 1 and 2). 1 , 7 , 8 A case of GP was discovered in inguinal hernia sacs following excision of the gastric teratoma in 1990. 1 Yeo et al. reported an unusual case of gliomatosis of bilateral scrotal sacs arising from an immature gastric teratoma. 8 Another report described gliomatosis involving the tunica vaginalis after removal of a retroperitoneal extragonadal immature teratoma. 7 In our case, GP presented with an extratesticular mass, which developed from an immature mesenteric teratoma.
Clinical and pathological features of immature teratoma with extratesticular gliomatosis peritonei.

FIGO: International Federation of Gynecology and Obstetrics; IT: immature teratoma.
Summary of extratesticular GP cases.
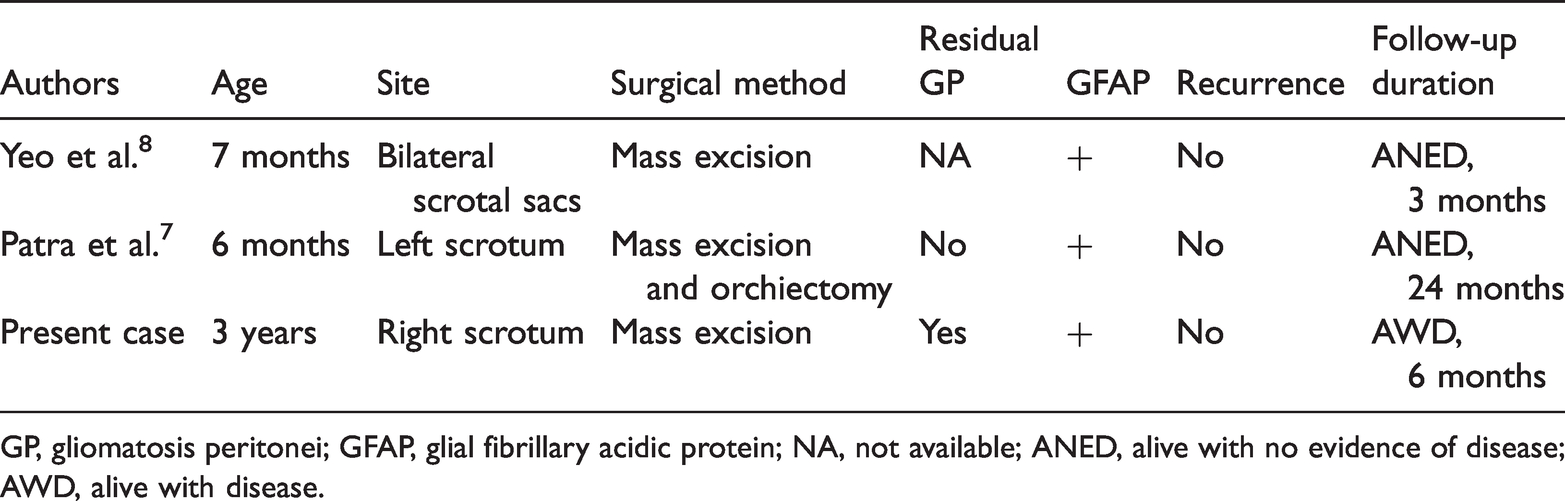
GP, gliomatosis peritonei; GFAP, glial fibrillary acidic protein; NA, not available; ANED, alive with no evidence of disease; AWD, alive with disease.
While the pathogenesis of GP remains unclear, there are several theories regarding the formation and development of GP. According to the lymphatic dissemination theory, mature glial tissue occurs in the lymph nodes, which indicates the feasibility of lymphatic transport of neural cells. 5 Another theory is that GP is derived from nonmalignant stem cells, which differentiate into glial cells with a change in the environment, and its genetic background is different from that of a teratoma. 9 , 10
One widely accepted theory is that GP results from implantation of immature neural or mature glial tissue into the peritoneum, and this occurs after surgical or spontaneous rupture of the teratoma capsule. 11 Some reports showed that most GPs were discovered simultaneously with surgery of the teratoma. 5 , 12 , 13 However, some cases of GPs occurred after surgery. 5 ,12–14 Teratomas that are adhered to the omentum or the capsule of teratomas, and tear during an operation may be the main reason for GP formation. 14 In this case, the capsule of the immature teratoma was incomplete intraoperatively, and it was closely adhered to the mesentery. We speculate that the most likely cause of GP formation in our case is that, during surgery, there was rupture of the capsule, which allowed glial components to spill and implant into the tunica vaginalis through the inguinal canal. Although ultrasound and an MRI scan of the abdomen did not reveal peritoneal nodules or a recurrent tumor, these techniques might not have been able to detect minor lesions.
Histological confirmation of the extratesticular mass in our case is consistent with the description of GP in other reports. 7 , 8 This tissue is composed of mature glial tissue, and shows strong staining of GFAP and S100, which reflects the mature nature of glial tissue.
With regard to treatment of teratomas with GP, the therapy should depend on the grade of the primary teratoma and not on GP. Because patients with an immature teratoma have a good prognosis in the neonatal age group, all patients, including our patient, underwent surgery without any further adjuvant therapy, and no recurrence was found (Table 1). Even though the majority of GP cases are usually benign, cases of malignant transformation have been reported. 15 , 16 Complete surgical resection of the teratoma and GP is useful for identifying the presence of malignant lesions and for preventing malignant transformation. However, lesions of GP are extensive, and they are difficult to completely remove. 17 Therefore, whether GP needs to be completely removed is a controversial issue. Yeo et al. 8 performed excision of the mass from GP of bilateral scrotal sacs without orchiectomy, while Patra et al. 7 performed orchiectomy along with mass excision (Table 2). In our case, biopsies showed that GP was not only an extratesticular mass, but also involved ductile tissue on the surface of the epididymis. There would have been a serious risk in destroying the testicular blood supply and causing testicular necrosis if we had removed the GP tissue completely. GP is usually benign. Therefore, after sufficient communication with the patient’s parents, the residual GP tissue was not completely removed. Consequently, the child still has this disease.
We created a lifelong follow-up plan for the child. We conducted three follow-up visits at 1, 3, and 6 months after partial excision of GP. Tumor markers were within the normal range and ultrasound showed no obvious intrascrotal nodules or a recurrent tumor. Subsequently, we will continue to follow-up the child every 6 months unless there is recurrence of a mass in the scrotum. Our patient is still in good health.
Conclusions
Rare cases of extra-peritoneal gliomatosis associated with extragonadal teratomas have been reported. To the best of our knowledge, this is the first case of extratesticular GP following an immature mesenteric teratoma. GP is usually benign, and extensive lesions of GP are difficult to completely remove. The residual GP implants in our patient were not completely removed. Even though the child still has GP, he is in good health, but requires lifelong follow-up.
Footnotes
Ethics statement
The Ethics Committee of Shenzhen Children’s Hospital waived the requirement for approval. The patient’s parents signed a consent from for publication of this case.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Sanming Project of Medicine in Shenzhen (No. SZSM201612013), Shenzhen Medical Key Discipline of Pediatric Surgery (2020-2024) (No. SZXK035), and Shenzhen Health System Research Projects (Nos. SZXJ2018045, SZFZ2018055).
Author contributions
All authors conceived and designed the study. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used during the current study are available from the corresponding author on reasonable request.
