Abstract
Objective
To estimate anti-seizure medication (ASM) treatment burden and its effects on health-related quality of life (HRQOL) in new-onset childhood epilepsy with centrotemporal spikes (CECTS) using different treatment approaches in Kazakhstan.
Methods
Forty-three patients were followed prospectively during 2015 to 2020 for at least 2 years. Patients were divided into three groups: (1) history of ≤3 seizures (n = 32); (2) ≥4 seizures (n = 6); (3) cerebral palsy coexisting with CECTS (n = 5). The first group was subdivided into treated (n = 8) and observed (n = 24) subgroups. The shortened Quality of Life in Childhood Epilepsy Questionnaire (QOLCE-55) was completed by parents after 6 months of follow-up.
Results
At the end of the study, all children had a sustained remission from seizures for at least 2 years. Differences were identified in emotional, social, and physical subscales between patients in the low seizure frequency group. Signs of low self-esteem, anxiety, depression, limited social interaction owing to pharmacotherapy, painful medical procedures, and stigma were reasons for decreased HRQOL in the treated subgroup. Overall HRQOL in treated (89.2 ± 5.2) patients was significantly decreased compared with observed children with low seizure frequency (98.0 ± 3.0).
Conclusion
ASM therapy does not necessarily improve and may decrease HRQOL in children with low seizure frequency CECTS.
Keywords
Introduction
Commonly known as rolandic epilepsy, childhood epilepsy with centrotemporal spikes (CECTS) is the most common epilepsy syndrome in children. 1 This is a self-limited type of epilepsy that usually begins between the ages of 3 and 14 years and peaks around 7 to 10 years. The incidence of CECTS is 10 to 20 per 100,000 children up to the age of 15 years. CECTS accounts for approximately 15% of all childhood epilepsies. 2
Certain patients are treated with anti-seizure medication (ASM) whereas others are only observed.3,4 Ideally, treatment of CECTS should suppress seizures and prevent evolution to atypical forms.5–7 However, nearly all ASMs cause adverse effects that range in severity from sleep disturbance, irritability, mood changes, and stomach upset to progressive impairment like aplastic anemia or hepatic failure.8–10 Therefore, side effects and familial views should be considered before initiation of pharmacotherapy.2,11
Classically, patients with CECTS are developmentally normal with infrequent seizures that mostly occur during sleep. 12 Nevertheless, there is a relationship between epilepsy stigma and impaired health-related quality of life (HRQOL).11,13 Additionally, recent sources of literature emphasize that age-dependent focal epilepsies can coexist with an underlying brain lesion, causing motor impairments such as cerebral palsy (CP).14,15 Thus, some children with CP should not be excluded from CECTS treatment approaches and directed instead to clinical pathways with more aggressive and prolonged therapy for “symptomatic epilepsy”.
In the present study, we sought to explore the outpatient management of CECTS, which may be approached differently even within a single community. Because the quality of pediatric epilepsy management depends not only on cessation of seizures but also on satisfactory levels of physical, mental, and social well-being, we used HRQOL to measure the success of the clinical treatment pathway (ASM versus observation). A relatively brief, validated 55-item version of the Quality of Life in Childhood Epilepsy Questionnaire (QOLCE-55) has been used at international level. 16 The questionnaire was recently shortened, but with its validity retained, making this approach even more practical. 17 The present study results can provide novel insight into estimation of the ASM treatment burden affecting quality of life in children with CECTS.
Methods
In this prospective observational cohort study, we followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline to ensure clear presentation of the results.18,19 Patients were diagnosed with CECTS according to the criteria set by the International League Against Epilepsy Classification of the Epilepsies (2017). 20
The present survey was conducted in a public–private outpatient clinic in Almaty, Kazakhstan, under the national health care insurance system. The clinic has the capacity to perform neurologic screening examinations and video-electroencephalography (EEG) monitoring. The clinic’s centralized outpatient services include all necessary diagnostic and therapeutic facilities: 1.5 Tesla magnetic resonance imaging (MRI), nocturnal video-EEG, as well as clinical laboratory testing and examination departments. The neuropsychological status of children is assessed by the public psychological commission upon request of a pediatric neurologist.
Patients’ well-being profiles were evaluated using the QOLCE-55, 16 which has been validated in new-onset epilepsy. 21 All patients were followed from their initial interaction with a pediatric neurologist and were therefore considered to have new-onset epilepsy. The QOLCE-55 includes 22 items covering cognitive functioning, 17 items on emotional functioning, 7 items addressing social functioning, and 9 items on physical functioning.
Parents were asked to report the frequency of occurrence for each statement describing their child’s social, physical, behavioral, and cognitive impairments during the past month. These answers were recoded as scores of 0, 25, 50, 75, and 100. Final assessment of the mean values for items on each subscale were calculated in such a way that higher scores indicated higher levels of well-being. The total score was calculated by taking the unweighted mean of the four subscales.
Research design
In the first stage of this longitudinal study, we consecutively selected 38 children who met the study criteria of CECTS: onset of epilepsy from ages 3 to 14 years with infrequent sleep-related brief hemifacial and/or hemibody sensory/motor seizures that may evolve to a focal to bilateral tonic–clonic seizure, with normal antecedent birth and neonatal history, standard development and cognition prior to clinical manifestation, unremarkable brain imaging studies, and stereotyped high-amplitude centrotemporal spikes without background abnormalities on EEG, which are activated with drowsiness and sleep. In light of recent publications indicating that the coexistence of CECTS with an underlying brain lesion might be able to be diagnosed,14,15 we also included patients with CP whose age at onset, seizure semiology, and EEG features were similar to those of children with CECTS. The included patients were followed up for at least 2 years after the initial evaluations.
In the second stage of the study, we divided patients into the following groups: group 1 included patients with CECTS and low seizure frequency (3 or less) (n = 32); group 2 comprised those with CECTS and high seizure frequency (4 or more) (n = 6); and group 3 included patients with CP coexisting with possible CECTS (n = 5).
Each participant was assigned to one of these three groups at their first appointment with a pediatric neurologist at our clinic during the ongoing longitudinal recruitment period from 2015 to 2018. The group with low seizure frequency was subdivided into two subgroups to obtain comparative assessment data regarding cognitive, emotional, social, and physical aspects of HRQOL in children with and without ASM therapy. Children in whom the decision was made to initiate anticonvulsant therapy with a history of fewer than three seizures were defined as the treated subgroup (n = 8). The observation subgroup (n = 24) comprised children whose parents or guardians chose to refrain from ASM initially. From the beginning of the study, specific treatment was not recommended in patients with a history of three seizures or less. Agreement or refusal to take anticonvulsants remained up to each child’s parents or guardians after they had been provided with detailed information on the prognosis of CECTS. All parents were informed of the most likely favorable outcome of the disease.1,22
Children with a history of more than three seizures at presentation were prescribed ASM therapy and were assigned to the second (high seizure frequency) group. Participants in the second and third groups were mandated by our clinical pathway to be prescribed ASMs. The distribution of patients by group and subgroup is presented in Table 1.
Distribution of patients by group and subgroup.

CECTS, childhood epilepsy with centrotemporal spikes; CP, cerebral palsy; ASM, anti-seizure medication.
Families of the included children consented to participate in a relatively brief interview (15 minutes) to collect HRQOL data. HRQOL was estimated prospectively using the QOLCE-55, which was completed by parents at the end of a 6-month period after group assignment. Consequently, participants in the treated subgroup were surveyed no earlier than 6 months after initiating ASM treatment. Discontinuation of ASM was encouraged in patients who had been seizure-free for at least 2 years. The treated subgroup completed the questionnaire while still taking medication, prior to ASM discontinuation.
Final assessment of the results of this study was carried out in 2020, 2 years after registration of the last patient included in the follow-up study. Anonymous patient data were additionally evaluated by the senior author to clarify the diagnosis of children who might have self-limited childhood epilepsy in the setting of additional brain lesions.
This study was approved by the Human Research and Ethics Committees of the Kazakhstan’s Medical University, School of Public Health (KSPH), Almaty. Written informed consent was obtained from the parents of all study participants.
Statistical analysis
A descriptive analysis and nonparametric statistical analysis were carried out. Overall well-being and subscale scores are presented as mean ± standard deviation (SD). The nonparametric Mann–Whitney U test was used to compare differences between the treated and observed subgroups, with statistical significance set to p < 0.01.
Owing to the high prevalence of QOLCE-55 values equivalent to 100 in the first group, a binomial distribution approach was implemented to recheck the accuracy of the calculations for small samples. The numeric values of mean subscale scores equivalent to 100 were encoded as 1 and those below 100 were encoded as 0. Differences were later assessed using Fisher’s exact test, a nonparametric test for small samples (fewer than 5), when appropriate. The results were considered statistically significant at a level of p < 0.05.
Results
Over half of the 43 patients (60.5%) presented with clinical manifestations of CECTS at age 6 to 9 (range 3–12) years. There was a slight predominance of male children (62.8%).
Patients with a high frequency of seizures (>3) were prescribed ASM owing to concerns regarding a high risk of injury and possible atypical evolution of CECTS. 6 In most patients with secondary generalized seizures, valproic acid was administered according to local treatment protocols. Two patients were taking sulthiame, an anticonvulsant with proven efficacy in CECTS. 23
Five patients with CP and possible CECTS (in group 3) presented to the outpatient pediatric neurology department several months after experiencing 1 to 3 seizures; by the end of the study, their total number of seizures did not exceed 4.
Relapse of seizures was registered in one patient in the treated low-frequency subgroup, and recurrence was detected in 11 observed patients (45.83%). Ten of the eleven relapsed children from the observation subgroup who were treated later (after their third or fourth seizure) were originally assigned to the untreated group. HRQOL evaluation in these 10 children was carried out when they were not being treated, prior to being prescribed ASM. By the end of the study (2020), all children had remission of at least 2 years, regardless of whether they were taking ASM.
Quality of life and school functioning
On the whole, the mean cognitive scores were slightly reduced in all groups of patients with CECTS, which coincides with numerous publications.24–26 In the first group (n = 32), 31% of parents (n = 10) reported that their child occasionally had trouble concentrating on a task, finding the correct words, doing one thing at a time, remembering things, making plans or decisions, and these children reacted slowly to things that were being said or done. These manifestations may be owing to seizures, comorbidities including attention deficit hyperactivity disorder (ADHD), or other factors. At the time of presentation, 28% of children with low seizure frequency had ADHD and 22% showed a slight change in thinking and memory skills.
Items defining reading and writing were excluded in preschool children (3–5 years of age), which is permissible according to the QOLCE-55 instructions. There were no significant differences in the cognitive data between observed (96.4 ± 5.7) and treated (93.0 ± 6.0) subgroups.
Decreased emotional functioning reached statistically significant values (p < 0.001) in treated patients (90.1 ± 4.7) compared with observed children who had low seizure frequency (98.6 ± 3.4). Parents described their children as worrying a lot and feeling frustrated or depressed, but parents did not mention socially inappropriate behavior, such as attacking people or swearing in public.
The social indicators showed a significant difference between the observed (98.4 ± 3.8) and treated (88.0 ± 6.6) subgroups (p < 0.001). Parents noted the presence of stigma (signs of anxiety, loss of confidence, and low self-esteem) that limited their child’s social interactions owing to the need to take ASM, mandatory trips to medical institutions, and painful procedures (blood sampling to monitor adverse reactions of ASM). The same reasons may underlie the lower subscale scores in groups 2 and 3.
The physical functioning subscale index was reduced in all groups of patients, which can be explained by the need to observe their nighttime sleep regime and some restrictions on sports activities, particularly swimming. There was a significant difference on the physical subscale between patients with low seizure frequency who were treated (85.8 ± 8.1) and observed patients (98.7 ± 2.9) (p < 0.001).
The final assessment showed that overall HRQOL scores in the treated subgroup (89.2 ± 5.2) appeared to be significantly decreased (p < 0.001) compared with observed patients with low seizure frequency (98.0 ± 3.0). Figure 1.

Final assessment of overall health-related quality of life scores in treated and observed patients with low seizure frequency.
Binomial distribution approach confirmed statistically significant decrease in HRQOL-indicators in all subscales (except cognitive) in the treated subgroup (Table 2).
Binomial distribution approach in the group with low seizure frequency.

*Significant using Fisher’s exact test.
The mean HRQOL scores in all patient groups, in comparison with the maximum score of 100, are presented in Table 3 and Figure 2.
Mean health-related quality of life scores in all study groups.
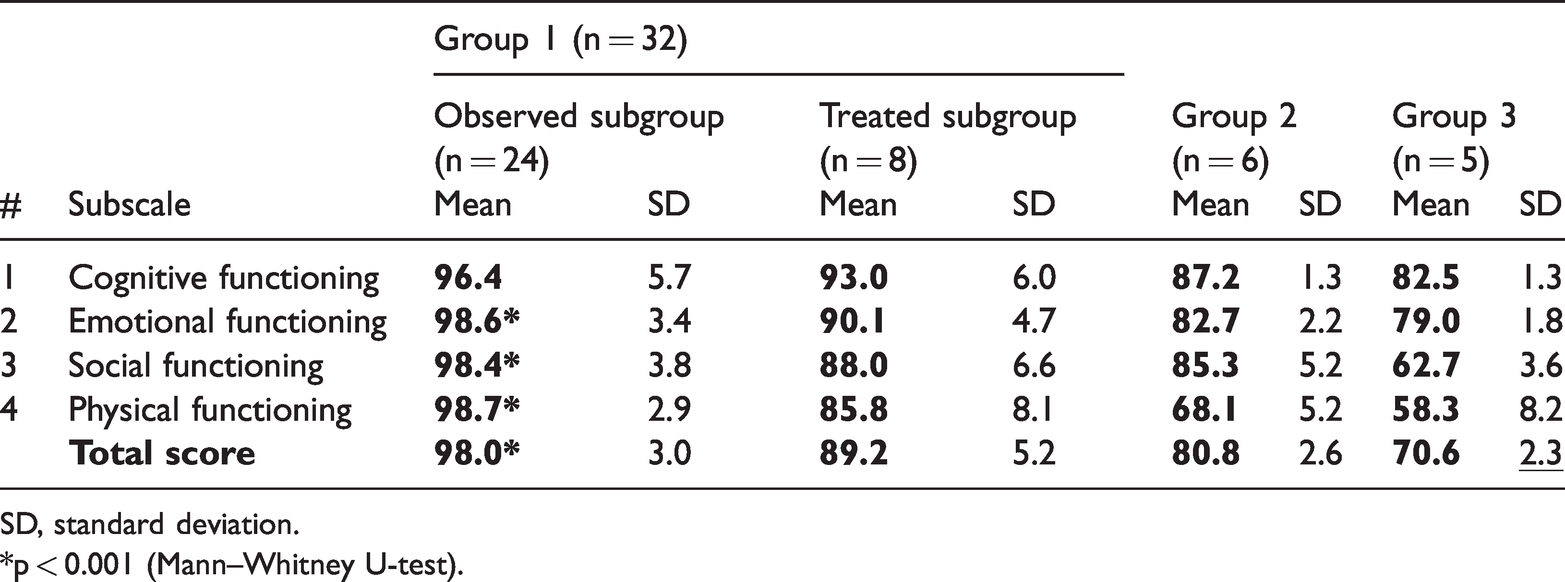
SD, standard deviation.
*p < 0.001 (Mann–Whitney U-test).

Mean health-related quality of life scores in all study groups in comparison with maximum score 100.
In all patients with CP, seizures were rare and well controlled with ASM, although there is a possibility that the seizures would have resolved in the absence of specific treatment. At the same time, indicators of social (62.7 ± 3.6) and physical functioning (58.3 ± 8.2) were significantly decreased in this group of children because of a greater degree of motor impairments causing restrictions in physical activities. The cognitive subscale index was notably reduced (82.5 ± 1.3) owing to some difficulties in writing, which is also the result of impaired coordination and refined motor skills of the hands (Table 3).
Comparative analysis of the average values of HRQOL subscales between the group with CECTS who had high seizure frequency and the group with CP coexisting with possible CECTS (the second and third groups, respectively), as well as among patients taking different ASMs, was not possible because of a small number of patients. Nevertheless, HRQOL in the second and third groups would not be expected to be improved over treated patients with low seizure frequency.
Discussion
The epidemiological data of our study correlate with global statistics regarding the onset of CECTS as well as the age and sex ratio of patients.1,2 CECTS predominantly presents between age 6 to 9 years, with a slight male predominance. Children are classically developmentally normal with infrequent seizures mostly occurring during periods of drowsiness, sleep, or on awakening (greater than 70%). Remission, regardless of whether the patient is taking ASM therapy, usually occurs 2 to 4 years after seizure onset and before age 15 years. However, as in our study population, retention of centrotemporal spikes on EEG is observed for a much longer period (more than 3 years after the last seizure, on average). 4
There is recent literature documenting the coexistence of age-dependent epilepsies and CP with an underlying brain lesion not affecting the cerebral cortex.14,15 This implies a good prognosis in children with CP associated with seizures. Because there is currently no clinical pathway in Kazakhstan for children with CP together with self-limited seizure disorders, the five patients in group 3 of the present study were considered to have “symptomatic epilepsy (by reason of CP).” These patients are prevented from discontinuing ASM after 2 seizure-free years owing to the presence of epileptiform spikes on EEG and abnormal MRI despite prolonged remission and rare seizure occurrence. Nevertheless, their age at onset, seizure semiology, normal cognitive development, and brain MRI with lesions confined to white matter without significant cortical injury and electroclinical features are similar to children with self-limited childhood-specific seizure disorders.
Defining who should be involved in a care map for CEСTS is also important and we would argue that properly screened patients without significant cortical injury (i.e., patients with CP and exclusively white matter lesions) should be able to follow this clinical pathway and not be subjected to a prolonged course of ASM.
Until recently, the effectiveness of epilepsy treatment was mainly assessed using such indicators as the response to ASM (complete remission or reduction in seizure frequency). It was assumed that if clinical and functional indicators improved during specific treatment, then HRQOL was also improved.
Modi et al. reported the negative impact of even a single seizure on pediatric HRQOL. Their retrospective study included 109 children, and the Pediatric Quality of Life Inventory (PedsQL) was used to analyze participants’ well-being profiles. The authors emphasized that evaluation of HRQOL is important for timely psychosocial intervention. 27
HRQOL in 30 Australian children with CECTS was determined in the original validation study by Connoly et al. 28 The Child Health Questionnaire, Child Behavior Checklist, and Quality of Life in Childhood Epilepsy Questionnaire were completed by parents. The children’s average psychosocial scores were significantly lower than normative data. An important point was that clinical variables had a minimal impact; cognitive and emotional impacts predominated.
In the present prospective study, differences were found in emotional, social, and physical aspects of HRQOL in treated versus observed patients in the subgroups with low seizure frequency. According to participants’ parents, epilepsy stigma was present, with signs of low self-esteem, anxiety, and depression in connection with the child’s diagnosis and the need to follow systematic therapeutic procedures. Low self-esteem is possibly the result of limited social and physical activities, such as restrictions on hobbies and sports activities in affected children. These children must also follow a strict daily routine to ensure a good night's sleep as well as a diet that focuses on the timing of eating, which are measures proposed to reduce the risk of seizures and side effects of ASM.
The findings regarding increased likelihood of depression and anxiety in our study correlates with those of Liu et al. 29 who found elevated rates of affective disturbance in 89 children with CECTS, in comparison with 75 healthy matched controls, using the Depression Self-Rating Scale for Children and the Screen for Child Anxiety-Related Emotional Disorders.
Unfortunately, we did not have a sufficient number of study participants to detect a difference in HRQOL scores among various ASMs. Because CECTS accounts for approximately 15% of childhood-onset epilepsy and presents during a limited period, 2 it should be possible to systematically compare alternative clinical pathways to optimize outcomes.
Considering the above findings, we conclude that ASM therapy does not necessarily improve and may decrease HRQOL, although this remains controversial and is approached differently in different parts of the world, which may be partly owing to cultural differences. Certain ethnic groups may be less likely to treat with medications whereas some populations are more likely to approve of pharmacotherapy.
It is difficult to measure whether preventing seizures improves HRQOL in comparison to the side effects of ASM via therapeutic drug monitoring and regular follow-up visits to a clinic; however, measuring HRQOL scores seems to be a reasonable start. Recently, an even shorter QOLCE-16 has been proposed as being equivalent to the QOLCE-55 used in our study, which would decrease the burden of HRQOL assessment on providers and patients. 17
Our study findings also suggest that ASM treatment in CECTS does not affect the ultimate prognosis, as previously known.1,6 ASM therapy should be evaluated in terms of its role of reducing seizures, preventing the evolution to atypical forms, or reducing possible negative cognitive consequences associated with epileptiform activity 11 versus the risk of side effects and impact on quality of life.
Along with traditional criteria such as the type, severity, and frequency of seizures, HRQOL is an integral indicator of the entire complex of organizational, diagnostic, and therapeutic measures. We would argue that the QOLСE-55 has been validated in patients with new-onset epilepsy, which would fit this population with CECTS. This instrument can serve as a measurable proxy to improve the clinical pathway in CECTS. At present, this is likely the optimal method to estimate the overall burden of epilepsy30,31 and ASM therapy on patients and their family.
Limitations and further objectives
In the present study, patients were recruited and followed at a single pediatric neurology center. The number of patients was later reduced to form a homogeneous sample with identical traits for further evaluation of those treated with ASM versus those who were only observed. These factors yielded a relatively small number of children included in the cohort, thereby limiting the conclusions.
Another limitation of the study is that although half of the untreated patients had additional seizures, this was not accounted for in our HRQOL assessment because these seizures occurred after the 6-month time point at which we administered the QOLCE-55. Patients’ HRQOL scores may have changed after having additional seizures. These patients were subsequently treated but only after our assessment was completed at the 6-month follow up.
In our geographic area, many parents have a negative perception of the prolonged use of medications, which might affect the study results. The implemented survey should be replicated in a larger group of patients and under other cultural and ethnic conditions.
Conclusions
The final assessment of our study findings revealed that ASM treatment does not necessarily improve and may decrease HRQOL in children who have CECTS with low seizure frequency. Except for cognitive aspects, indicators of well-being were slightly but significantly lower in patients treated for CECTS than in observed patients without ASM. A larger study with more participants may help to determine those factors that affect HRQOL in CECTS and optimize the clinical pathway implemented in Kazakhstan.
Footnotes
Data availability
All materials and data used and analyzed in the current study are available from the corresponding author on reasonable request.
Author contributions
Conceptualization: An O, Aringazina A
Data curation: An O, Nagae LM, Aringazina A, Winesett SP
Formal analysis: Aringazina A, Winesett SP
Methodology: Aringazina A
Project administration: Aringazina A, An O
Visualization: Nagae LM, Winesett SP
Writing – original draft: An O, Winesett SP
Writing – review & editing: An O, Winesett SP
All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
