Abstract
Objective
This study aimed to quantify the effect of the dwell time deviation constraint (DTDC) on brachytherapy treatment for cervical cancer.
Methods
A retrospective study was carried out on 20 patients with radical cervical cancer. The DTDC values changed from 0.0 to 1.0 by a step size of 0.2. We adjusted the optimization objectives to ensure that all plans were optimized to a high-risk clinical target volume (HRCTV) D90 (the dose to 90% of the HRCTV) = 6 Gy, while keeping the dose to the organs at risk as low as possible. The dose–volume histogram parameters and the dwell time data were compared between plans with different DTDC values.
Results
The HRCTV volume covered by 150% of the prescription dose gradually increased with increasing DTDC values. As the DTDC value increased from 0.0 to 1.0, the effective dwell point proportion increased from 61.78% to 90.30%. The mean dwell time initially decreased with an increase in the DTDC value, reached the minimum value at DTDC = 0.8, then slightly increased at DTDC = 1.0.
Conclusions
When using inverse planning simulated annealing optimization for radical cervical cancer cases, the recommended DTDC value is approximately 0.6 if the organ dose needs to be limited.
Keywords
Introduction
Brachytherapy has the characteristics of a high dose near the source and rapid dose drop-off away from the source. Brachytherapy is widely used in treating cervical cancer, prostate cancer and head and neck cancer. 1 Currently, image guided three-dimensional (3D) brachytherapy is the mainstream for brachytherapy. Using optimization technology, 3D brachytherapy achieves an individualized patient dose distribution with improved tumor dose coverage while sparing surrounding normal tissue. 2
The inverse optimization algorithm in 3D brachytherapy usually produces a plan with a large dwell time variation. A large change in the dwell time generates high-dose and low-dose regions in the tumor along the direction of the applicator. From a radiobiological point of view, such high-dose and low-dose regions should be avoided unless the tumor requires a non-uniform dose distribution. To control variation in the dwell time, physicians can use inverse optimization algorithms, such as the inverse planning simulated annealing (IPSA), hybrid inverse planning optimization and SagiPlan, to provide a dwell time modulation factor (DTMF) for controlling variation in the dwell time. The dwell time deviation constraint (DTDC) is used for IPSA, the dwell time gradient restriction is used for hybrid inverse planning optimization and the dwell time homogeneity error weight is used for SagiPlan. The DTMF is a user-specified value. The minimum DTMF value does not impose any restriction on the dwell time variance, and the maximum DTMF value restricts all dwell times to have the most uniform value. By changing the DTMF, the planner can obtain a clinically satisfactory treatment plan.3,4
Several studies have reported the effect of the DTMF on brachytherapy treatment plans, mainly on prostate cancer.3,5,6 However, there have been a limited number of studies on the effect of the DTMF on the treatment plan quality for cervical cancer. Roy et al. studied the effect of the DTDC on intracavitary plans. 7 They showed that increasing the DTDC value not only reduced local hot spots, but also decreased the maximum dose to 2 cm3 (D2cc) of the bladder.
Another study by Mosleh-Shirazi et al., however, showed that choosing a non-zero dwell time homogeneity error weight led to a general increase in the dwell time uniformity, but increasing this parameter resulted in an increase in bladder
This study aimed to investigate the effect of the DTDC on brachytherapy treatment planning in cervical cancer. We increased DTDC values from 0.0 to 1.0 by a step size of 0.2. Dosimetric parameters, the dwell time and the volume covered by low-dose 1, 2 and 3 Gy (
Materials and methods
Patient data
We retrospectively studied 20 patients with radical cervical cancer who completed intracavitary brachytherapy in our hospital. All patients who were treated from September 2020 to November 2020 were randomly selected. All tumors were squamous cell carcinomas and consisted of International Federation of Gynecology and Obstetrics stage IIA–IIB (6 with IIA, 14 with IIB). In all patients, magnetic resonance imaging scans were acquired after 45 to 46 Gy of whole pelvic external beam radiotherapy (EBRT), and subsequently, 4 to 5 fractions of brachytherapy boost were performed. Each patient underwent a computed tomography (CT) scan after the insertion of a Fletcher applicator (Elekta part no. 189.730; Elekta AB, Stockholm, Sweden). The CT resolution was 0.1 × 0.1 × 0.3 cm. On the basis of a previous magnetic resonance imaging and gynecological examination, a physician completed delineation of the target and organs at risk (OARs) using the Oncentra Brachy V4.3 treatment planning system (Elekta AB). The delineation was generally followed by the European study on MRI-guided BRAchytherapy in locally advanced CErvical cancer (EMBRACE) recommendation (see http: //www.embracestudy.dk). The target was the HRCTV, and the OARs included the bladder, rectum and sigmoid. In the EMBRACE recommendation, the HRCTV
The study was reviewed and approved by the ethics committee of Sichuan Cancer Hospital (reference number: SCCHEC-02-2020-008). Patients participating in the study provided verbal informed consent. We have de-identified all patients’ details.
Plan simulation
The original treatment plan was performed with the Oncentra Brachy V4.3 treatment planning system. The applicator reconstruction was carried out according to the European Society for Radiotherapy and Oncology guidelines. 9 All treatment plans were optimized using the IPSA+graphic optimization approach in which treatment plans were initially optimized with IPSA, then a graphic optimization method was used to adjust the dose distribution until the target and OAR dose were satisfactory. The source step size was 2.5 mm and the dose grid resolution was 0.1 × 0.1 × 0.1 cm for all plans. The total number of dwell points in the 20 patients ranged from 28 to 47, with a median value of 43. The average number of dwell points in tandem was 21 and the average number of dwell points in two ovoids was 21.
The simulation plan only used IPSA optimization. The DTDC values changed from 0.0 to 1.0 by a step size of 0.2. Each patient had six plans corresponding to different DTDC values. We kept the prescription dose of the HRCTV (volume: 31.15–69.03 cm3, average: 49.15 cm3) unchanged. The weight of the target, the prescription dose and the weight of the OARs were adjusted according to the different anatomy of each patient to ensure that all plans were optimized to an HRCTV
Evaluation of plan quality
The quality of optimized treatment plans for the 20 patients was evaluated using different criteria. For different DTDC values, we first assessed the plan by the dosimetric parameters recommended by the European Society for Radiotherapy and Oncology, including
We used the conformal index (COIN) to evaluate how accurately the target was covered by the prescription dose, and the COIN was calculated using the following formula:
The dwell time data of all patients were collected. We calculated the mean dwell time per dwell point for each patient, then calculated the mean dwell time and standard deviation of all patients. The effective dwell point (EDP) was defined as the dwell point that had a dwell time greater than 0.1 s. We calculated the EDP proportion for each patient and documented the mean EDP proportion in all patients.
We used the plan modulation index (PMI) to interpret the change in dwell time distribution when the DTDC increased. The PMI was defined as the maximum deviation of the dwell time from the average dwell time for each catheter, which was normalized to the maximum dwell time for the treatment plan, and averaged over all catheters in the plan.
5
The PMI was calculated using the following formula:
Results
Dosimetric parameters
The patients were aged 35 to 70 years at the time of treatment. Table 1 shows the mean dosimetric parameters in the 20 patients. There was no considerable change in the COIN value and the maximum COIN difference was 0.03. HRCTV
Dosimetric parameters for the target and organs at risk.
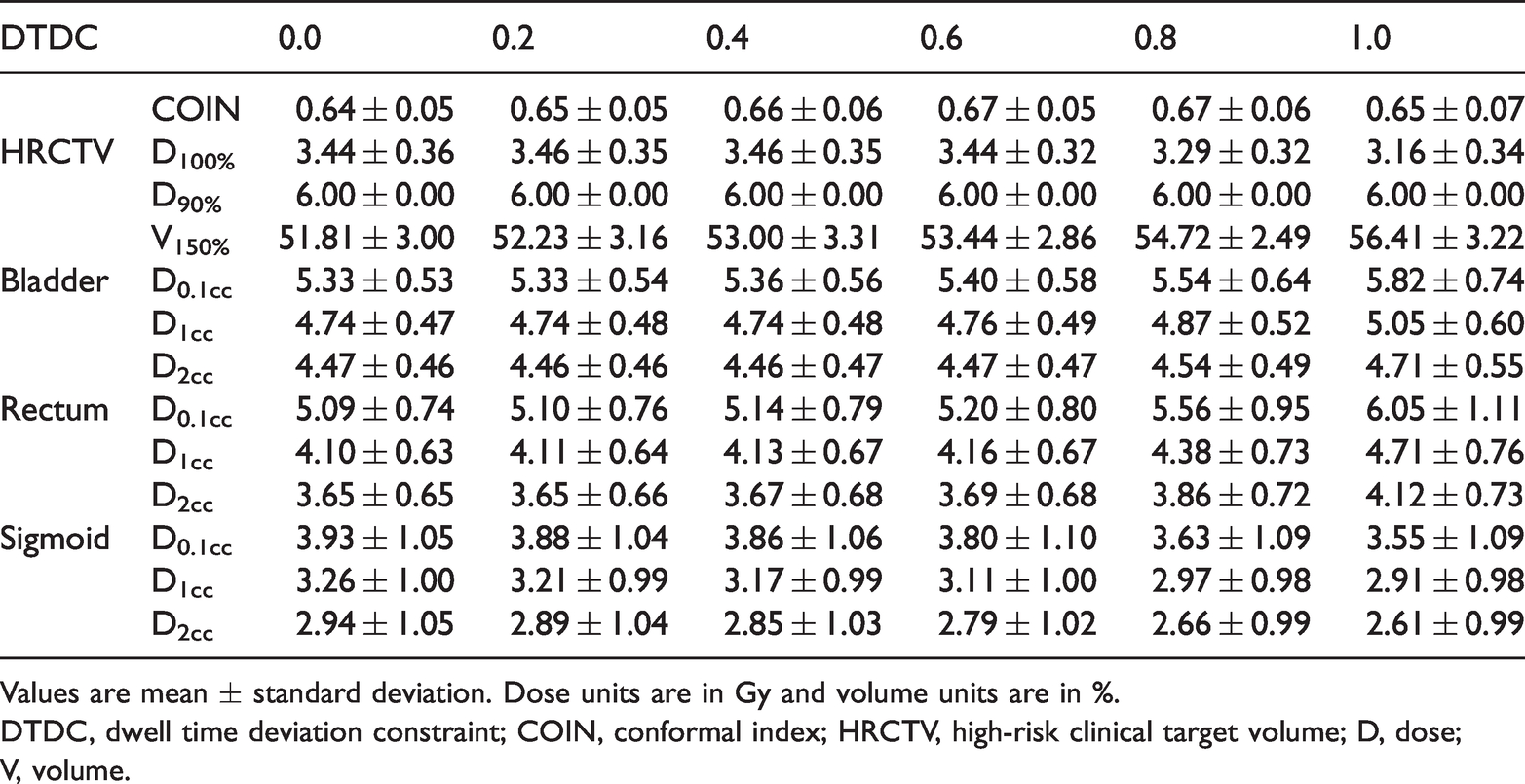
Values are mean ± standard deviation. Dose units are in Gy and volume units are in %.
DTDC, dwell time deviation constraint; COIN, conformal index; HRCTV, high-risk clinical target volume; D, dose; V, volume.
Figure 1 shows the variation in low-dose volume as a function of the DTDC for the 20 patients.

Relationship between the DTDC and low-dose volume
Dwell time
Table 2 shows the EDP proportion, mean dwell time and PMI. As the DTDC value increased from 0.0 to 1.0, the EDP proportion increased from 61.78% to 90.30%. The trend of the change in the mean dwell time with DTDC was consistent with the low-dose volume. The mean dwell time initially decreased with an increase in the DTDC value, reached a minimum value at a DTDC = 0.8, then slightly increased. With an increase in the DTDC value, the mean PMI decreased, resulting in a more homogeneous dwell time distribution within each catheter.
Effect of increasing the DTDC on the dwell time and the plan modulation index.

Values are mean ± standard deviation.
DTDC, dwell time deviation constraint; EDP, effective dwell point; PMI, plan modulation index.
Discussion
Conventional radiotherapy for locally advanced cervical cancer combines EBRT and brachytherapy. Brachytherapy treatment planning systems allow the user to apply a DTMF to reduce any large change in the dwell time. In this study, we investigated the effect of the DTDC on the brachytherapy plan for cervical cancer. We found that an increase in the DTDC value increased the mean dose in the bladder and rectum. However, an increase in the DTDC also increased the high-dose (9 Gy) volume in the target. Additionally, the mean dwell time per dwell point decreased as the DTDC value increased in a range of 0.0 to 0.8, which resulted in a decrease in the low-dose volume. After balancing the OAR dose, the high-dose volume in the tumor, and the low-dose (1, 2 and 3 Gy) volume, the recommended DTDC value for cervical cancer is approximately 0.6 if the OAR dose is below the upper limit.
The effect of the DTDC value on the COIN can be ignored. We found that the difference between the best COIN and the worst COIN was 0.03. However, an increase in the DTDC increased the dosimetric parameters of the bladder and rectum. When the DTDC value is 0, the dwell points near the OARs have a short or even zero dwell time, thus the bladder and rectum can be protected. The dwell point in the target and away from the OARs has a long dwell time to ensure dose coverage of the target volume. The EDP increases with an increase in the DTDC. The dwell points near the bladder and rectum are activated, the dose distribution becomes more uniform, and the dose in the bladder and rectum increases. When the DTDC value increases to approximately 0.8, the dose distribution is close to the traditional pear-shaped dose distribution. Studies have reported that the bladder and rectum dose optimized on the basis of the IPSA is lower than the traditional pear-shaped dose distribution.11,12 Our results are different from those in a previous study by Roy et al.
7
who showed that the DTDC value was negatively correlated with
In the treatment of locally advanced cervical cancer, brachytherapy plays an irreplaceable role.
14
A main reason for this important role is that brachytherapy provides a higher central radiation dose to the tumor than that with EBRT. A high dose in the center of the tumor is more effective in tumor control than a homogeneous dose distribution.
15
Therefore, from the point of view of tumor control, local low-dose and high-dose regions should be avoided in a tumor unless there is a special radiobiological consideration. We observed that the number of EDPs increased with an increase in the DTDC value. In this study, we defined the high dose as 150% of the prescription dose (9 Gy), and the homogeneity of the high dose inside the target as
In clinical practice, we rarely evaluate the low-dose volume in brachytherapy. This may have clinical implications that we are not aware of at present. The clinical effect of an increased low-dose volume is not known in detail, but previous studies have shown that a large volume receiving more than 60 Gy (EBRT+brachytherapy) is correlated with side effects.
16
Therefore, we support the notion proposed by Trnková et al. that taking these non-defined anatomical structures into consideration is clinically more appropriate.
17
Increasing the DTDC value reduced the mean dwell time per dwell point for DTDC values of 0.0 to 0.8, thus reducing the volume of low-dose (
We acknowledge that this study has some limitations. One limitation is that we did not consider the robustness of the plan against variations that could be caused by applicator displacement or anatomical changes. 5 However, previous studies have shown that small changes in the DTDC (e.g., from 0.2–0.4) do not significantly affect the robustness of the plans for catheter displacement. 6 Another limitation is that only one applicator type was included in this study. There are other applicators (e.g., tandem and ring applicators) that can be used in intracavitary brachytherapy for cervical cancer. Different applicators have different dosimetric properties. The determination of DTDC values in other applicators and other sites requires further studies.
In conclusion, this study shows that an increase in the DTDC value increases the radiation dose in the bladder and rectum. Additionally, an increase in the DTDC also increases the high-dose (9 Gy) volume in the tumor. An increase in the DTDC value in the range of 0.0 to 0.8 also reduces the average dwell time per dwell point, resulting in a lower low-dose volume. For radical cervical cancer cases, our study suggests that the DTDC value should be approximately 0.6 if the OAR dose is limited. DTDC values in other applicators and other sites need to be determined in further studies.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chengdu Science and Technology Program (2019-YF09-00095-SN, 2019-YF05-00425-SN) and the Sichuan Science and Technology Program (2019YJ0581, 2021YFG0320).


