Abstract
A 57-year-old woman was diagnosed with IDH-wildtype (IDHwt) astrocytoma (World Health Organization grade II) with the molecular characteristics of glioblastoma. She underwent concurrent radiotherapy and chemotherapy according to the Stupp protocol in combination with a multi-target antiangiogenic drug and additional intrathecal chemotherapy using methotrexate. During treatment, the patient’s tumor showed rapid progression. The chemotherapy with temozolomide was stopped and replaced with radiotherapy combined with tumor treating fields (TTF), the poly (adenosine diphosphate-ribose) polymerase (PARP) inhibitor niraparib, and anlotinib. After the radiotherapy was completed, the symptoms of increased intracranial pressure and epilepsy were well controlled. Considering the patient’s tolerance to the treatment, the combined therapy of TTF and anlotinib was continued, and osimertinib, a third-generation epidermal growth factor receptor–tyrosine kinase inhibitor with good permeability of the blood–brain barrier, was added. The patient was regularly followed up and had no obvious adverse drug reactions. Head magnetic resonance imaging (plain scan + enhanced scan) suggested that the lesions were stable. For rapidly progressing glioblastomas or histological grade II/III IDHwt astrocytomas, the combination of TTF and a PARP inhibitor during radiotherapy may have a synergistic effect on tumor control and is well tolerated by patients.
Keywords
Introduction
With the continuous exploration of the molecular pathology of glioma, it is currently believed that IDH-wildtype (IDHwt) astrocytoma has growth characteristics and a prognosis similar to those of glioblastoma multiforme. 1 , 2 This study was performed to present a case of IDHwt astrocytoma with multiple intracranial and central nervous system disseminations confirmed by biopsy and pathology. The patient developed continuous, rapid tumor progression during treatment with concurrent radiotherapy and chemotherapy according to the Stupp protocol. However, concurrent tumor treating fields (TTF) combined with a poly (adenosine diphosphate-ribose) polymerase (PARP) inhibitor during subsequent radiotherapy effectively blocked rapid progression of the tumor and controlled the disease.
Case description
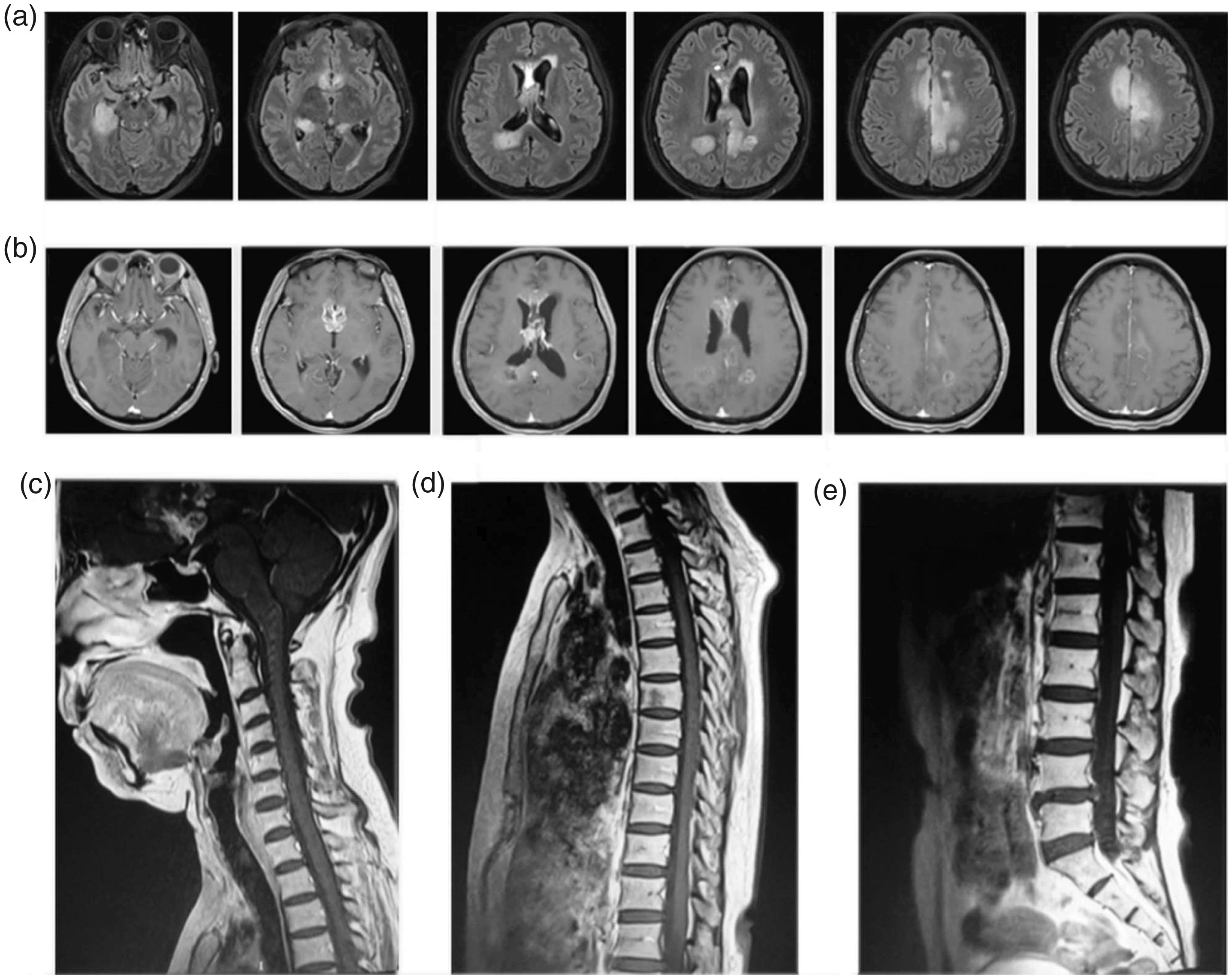
The reporting of this study conforms to the CARE guidelines. 3 A 57-year-old woman developed sudden-onset dizziness on 18 July 2019. The initial plain computed tomography scan indicated no obvious abnormalities. However, the patient’s dizziness gradually worsened and was accompanied by bilateral lower limb weakness and headache. On 28 November 2019, the patient underwent head magnetic resonance imaging (MRI) based on suspicion of multiple intracranial space-occupying lesions, with particular suspicion of glioma (Figure 1). On 12 December 2019, a stereotactic biopsy was performed on enhanced lesions in the suprasellar region, and the postoperative pathological examination revealed an astrocytoma (World Health Organization grade II). Furthermore, immunohistochemistry examination revealed the following results: GFAP(+), IDH1(−), Ki-67(10%–20%), Olig-2(+), CD20(−). The molecular pathological examination showed IDHwt, non-co-deletion of 1p/19q, unmethylated MGMT, C228T mutation in the TERT promoter, epidermal growth factor receptor (EGFR) amplification, and CDK4 amplification. The final integrated diagnosis was IDHwt astrocytoma with the molecular characteristics of glioblastoma. On 31 December 2019, an enhanced MRI examination of the head and spinal cord performed before radiotherapy showed abnormal signals in the right deep temporal region, right thalamus, bilateral paraventricular regions, bilateral frontoparietal regions, septum pellucidum, and corpus callosum, and these signals were more progressive than those during the biopsy.

(
Tumor dissemination was considered because of linear enhancement in the horizontal leptomeningeal and spinal meninges at the pons and C3 centrum as well as the horizontal spinal meninges and cauda equina at the T2–L1 centrum (Figure 2). The patient also had obvious symptoms of increased intracranial pressure and limb weakness. The Karnofsky performance scale (KPS) score was 50 points. Furthermore, lumbar puncture showed increased intracranial pressure (300 mm H2O), and biochemical analysis of the cerebrospinal fluid indicated a decreased glucose level and increased protein and lactic acid levels. Leptomeningeal dissemination and tumor metastasis were considered in combination with the clinical manifestations and imaging findings. After a multidisciplinary team (MDT) discussion, the patient underwent concurrent radiotherapy and chemotherapy according to the Stupp protocol (whole-brain radiotherapy, 36 Gy/20 fractions, five fractions/week; temozolomide, 75 mg/m2 daily) in combination with a multi-target antiangiogenic drug (oral anlotinib 8 mg every other day). During the treatment, additional intrathecal chemotherapy of methotrexate 10 mg was given once a week.
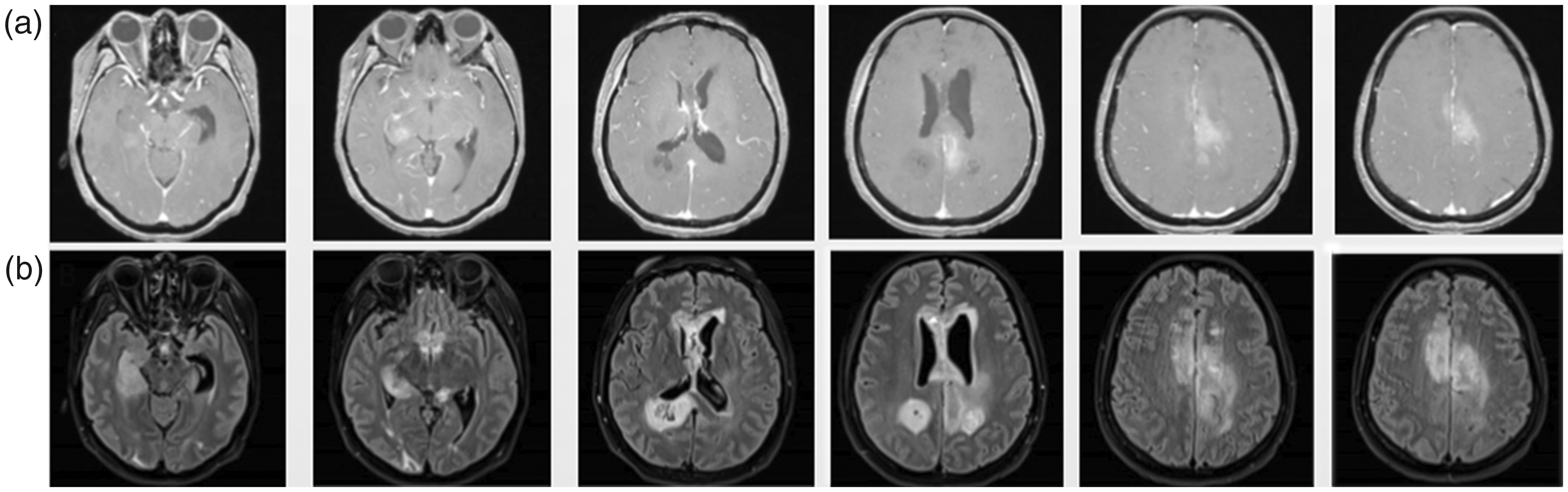
(
After starting the treatment, the patient’s symptoms persisted and she developed an increase in intracranial pressure and intermittent seizures with a further decrease in her limb muscle strength (level IV in left limb and level II in right limb). The patient received concurrent whole-brain radiotherapy (18 Gy/10 fractions) and TTF (the patch was removed during radiotherapy). Head MRI re-examination performed on 20 January 2020, while the patient was receiving whole-brain radiotherapy at 23.4 Gy/13 fractions, indicated that some lesions were in a progressive state, and new abnormal FLAIR signals were observed in several cerebral sulci (Figure 3). The KPS score decreased to 20 points. After another MDT discussion and obtaining the patient’s consent for treatment, the chemotherapy with temozolomide was stopped and replaced with radiotherapy combined with TTF, the PARP inhibitor niraparib (200 mg once nightly), and anlotinib. The symptoms of increased intracranial pressure and epileptic seizures were gradually alleviated. After the end of the whole-brain radiotherapy, local intensity-modulated radiotherapy (18 Gy/10 fractions) was continued in the area with an abnormal FLAIR signal observed by head MRI on 20 January 2020.
(
After the radiotherapy was completed on 13 February 2020, the symptoms of increased intracranial pressure and epilepsy were well controlled, and the KPS score rose to 50 points. However, a routine blood test showed a decrease in the neutrophil and platelet counts, suggesting grade III myelosuppression. After stopping the niraparib and symptomatic treatment for 2 weeks, these indexes returned to the normal ranges. A head MRI re-examination was again performed on 24 February 2020, indicating no tumor progression compared with the images obtained on 13 February 2020, and the abnormal FLAIR signals in the sulci had disappeared (Figure 4). Moreover, another lumbar puncture and monitoring of the cerebrospinal fluid indicated that the glucose had returned to the normal range. Considering the patient’s tolerance to treatment, the combined therapy of TTF and anlotinib was continued, and osimertinib, a third-generation EGFR–tyrosine kinase inhibitor (EGFR-TKI) with good permeability of the blood–brain barrier, was added; intrathecal chemotherapy with methotrexate was concurrently performed once a month. The patient continued regular follow-up and had no obvious adverse drug reactions. Head MRI performed on 25 April 2021 suggested that the lesions were stable.
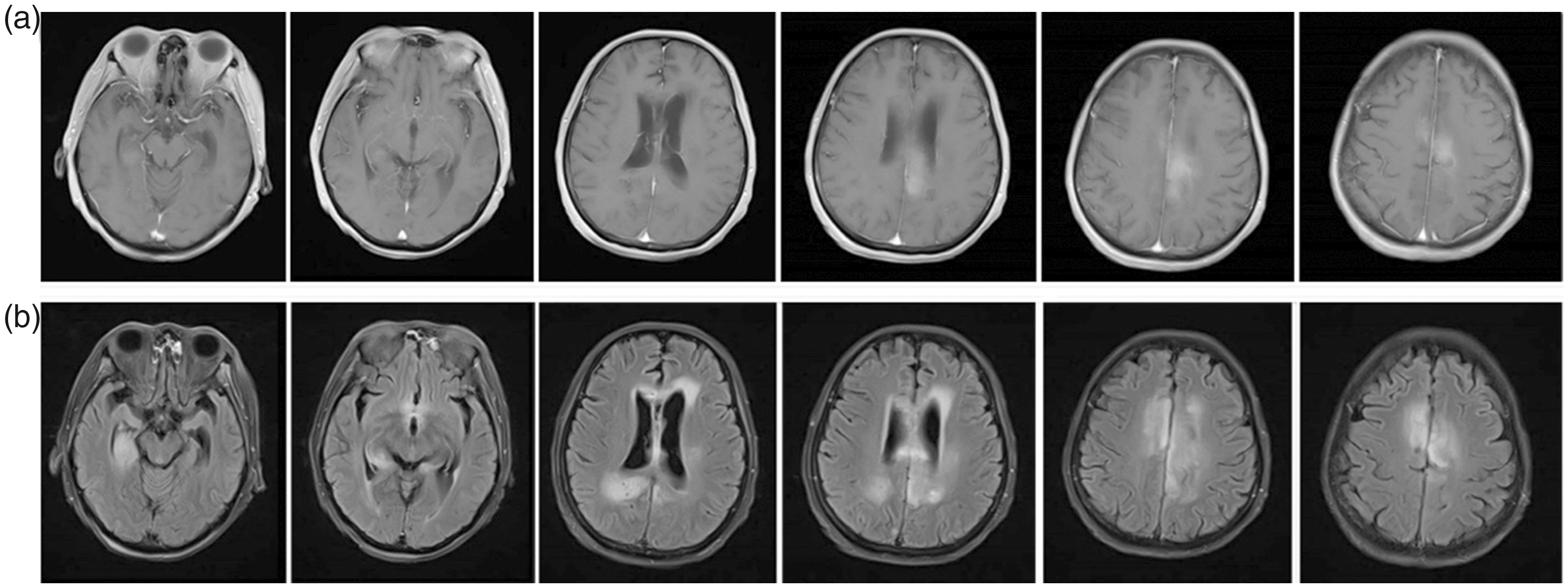
(
Discussion
The biopsy and molecular pathology findings confirmed that the patient had IDHwt diffuse astrocytoma with EGFR amplification and TERT promoter mutation, which is consistent with the molecular characteristics of glioblastoma and has a poor prognosis. 2 In this case, the patient’s clinical symptoms continued to be aggravated during concurrent radiotherapy and chemotherapy according to the Stupp protocol, and the MRI re-examination indicated tumor progression. The EF-14 experiment fully confirmed that TTF can improve the prognosis of newly diagnosed glioblastoma. 4 Furthermore, preclinical studies have confirmed that TTF can synergize with radiotherapy by inhibiting the repair mechanism of DNA double-strand damage, strengthening the inhibition of cell mitosis, and reducing the survival of glioma cells. 5 This synergistic effect was also confirmed in subsequent small-scale clinical trials. 6 Therefore, TTF was applied concurrently in the subsequent radiotherapy treatment of our patient, and the TTF patch was removed during radiotherapy to avoid the influence of the patch on the radiation dose to the greatest extent possible.
Previous studies have shown that the efficacy of TTF is closely related to patient compliance. 4 , 7 Our patient’s average daily patch-wearing time reached 96%, which was the basis for ensuring efficacy. During TTF treatment, no adverse effects were observed other than scalp pruritus, and the safety of this combined therapy was basically consistent with that described in the literature. 5 In MDT discussions, the following possible reasons were considered for the worsening of the patient’s symptoms during the initial treatment according to the Stupp protocol and for the progression of the lesions in the posterior occipital horn of the right lateral ventricle, as revealed by the MRI re-examination. (1) The tumor was highly malignant and in a state of rapid progression, while the cumulative dose of early radiotherapy was low and the therapeutic dose was not reached, or the tumor itself was resistant to radiotherapy. (2) The molecular pathological report showed a low degree of methylation of the MGMT promoter and minimal benefit of concurrent chemotherapy with temozolomide. (3) The patient had multiple intracranial lesions in a scattered focus range, and some parts might not have been covered at a higher electric field intensity. (4) The special treatment mechanism of TTF slowed down its efficacy. Previous studies have shown that the survival advantage rendered by TTF usually takes 3 months.4,8
Because our patient developed tumor progression during the combined therapy, a new therapy had to be developed. PARP inhibitors have been found to have a synergistic effect on current treatments. Karanam et al. 6 performed an in vitro experiment showing that TTF combined with radiotherapy and a PARP inhibitor increased the lethality against tumor cells. PARP is a family of proteins related to the repair of DNA damage. When DNA damage occurs, PARP1 is recruited to the lesion site and mediates the addition of PAR, thus forming a molecular scaffold for the repair process and mobilizing other DNA repair factors. PARP inhibitors inhibit the phosphorylation of PARP1 and increase the affinity of PARP1 to sites of DNA damage. 9
In a small-scale preclinical trial on children with high-grade astrocytoma and diffuse endogenic pontine glioma, a PARP inhibitor was observed to have radiosensitizing properties that induced DNA damage. 10 After MDT discussions in the present case, niraparib was used at a reduced dose of 200 mg once nightly. Nevertheless, the patient developed a dry cough during treatment, and grade III myelosuppression was observed at the end of the radiotherapy. Considering the patient’s treatment tolerance and other factors, niraparib was not continued during the subsequent treatment but was instead replaced with osimertinib, a third-generation EGFR-TKI drug with a better tolerance, for maintenance treatment based on the patient’s genetic test results. Despite controversies regarding the treatment of glioma with EGFR-TKIs, a progression-free survival period of up to 14 months was observed after radiotherapy. However, the efficacy must still be verified by follow-up visits. 11
This is the first report of a patient with malignant glioma treated with concurrent TTF and a PARP inhibitor during radiotherapy. This combined treatment effectively prevented rapid progression of the tumor, and the patient showed good tolerance during the treatment. However, its efficacy still needs to be explored on a larger scale because the aforementioned findings were only the results of a single case.
For rapidly progressing glioblastomas or histological grade II/III IDHwt astrocytomas, the combination of TTF and a PARP inhibitor during radiotherapy may have a synergistic effect on tumor control and is well tolerated by patients.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics
The patient provided written and verbal informed consent for treatment as well as consent for publication of the report. The study protocol was approved by Tianjin Huanhu Hospital ethics review committee.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
