Abstract
Objective
Identifying novel biomarkers involved in the development of gastric cancer (GC) can provide potential therapeutic strategies and improve clinical prognosis. miR-301-3p and Cx43 are reportedly dysregulated in GC. miR-301-3p and Cx43 interaction, and their functions in GC progression, are still poorly understood.
Methods
The expression levels of miR-301-3p and Cx43 in GC tissues and cell lines with various differentiation degrees were evaluated by RT-qPCR. The interaction between miR-301-3p and Cx43 was assessed by dual-luciferase reporter assays. CCK8 and Transwell assays were employed to assess the effects of the miR-301-3p-Cx43 axis on GC cell proliferation, migration, and invasion.
Results
Cx43 was significantly downregulated in GC tissues and cell lines, while miR-301-3p expression was negatively correlated with Cx43 mRNA levels. The expression levels of Cx43 and miR-301-3p were closely associated with the differentiation, TNM stage, vascular invasion, and lymph node metastasis status of GC patients. Cx43 overexpression could suppress the proliferation, migration, and invasion of GC cells. Cx43 mRNA is a direct target of miR-301-3p, and transfection of an miR-301-3p mimic could reverse the inhibitory effects of Cx43.
Conclusion
The miR-301-3p-Cx43 axis is involved in the development and progression of GC by affecting the proliferation, migration, and invasion of GC cells.
Introduction
Cancer has become a major public health problem and its increasing incidence and mortality have made it a leading cause of death worldwide. 1 Gastric cancer is a common digestive tract malignancy. Although the incidence of gastric cancer has decreased in the past decades, it remains a major cause of cancer-related death. 2 The degree of cancer differentiation is an important factor in the development of gastric cancer. 3 Identifying effective biomarkers associated with the differentiation of gastric cancer can help physicians adjust the therapy used to treat this disease according to the development of an individual patient’s condition.
Connexins are part of a group of proteins that can form gap junction channels, which can benefit the intercellular communication between cells and facilitate the transfer of signaling molecules. 4 Connexin 43 (Cx43) is a common connexin protein isoform that is widely expressed in epithelial tissues. 5 The clinical significance of Cx43 has been reported in various human diseases, such as hepatocellular cancer, breast cancer, and ovarian cancer.6–8 Cx43 expression was found to be downregulated in gastric cancer and implicated in the occurrence, development, and metastasis of this disease. It can also enhance the chemosensitivity of gastric tumors.9–11 The expression and function of Cx43 in various differentiation levels of gastric cancer could possibly provide a clinical reference for risk prediction of this malignancy.
MicroRNAs (miRNAs) are part of a group of small non-coding RNAs that regulate gene expression. They are involved in tumorigenesis and control a variety of cellular pathways that affect proliferation, differentiation, migration, and invasion. miR-301-3p reportedly binds to the 3ʹ untranslated region (UTR) of Cx43 mRNA and participate in the pathogenesis of bacterial meningitis. 12 miR-301-3p also promotes the proliferation and cell cycle progression of hepatocellular carcinoma cells, while inhibiting apoptosis. 13 Additionally, miR-301-3p regulates various pathways in gastric cancer cells and promotes the development of this disease. 14 However, the interaction between miR-301-3p and Cx43 and its effects on the development of gastric cancer are not fully clear. Hence, this study focused on the expression of Cx43 in gastric cancer tissues of different differentiation levels and its interaction with miR-301-3p to identify novel biomarkers that affect the differentiation and progression of gastric cancer.
Materials and methods
Patients and tissue samples
Sixty gastric cancer patients with various differentiation degrees that underwent surgery at the Affiliated Hospital of Chengde Medical University from 2017 to 2019 were included in this study. The following criteria were required for inclusion: a) patients diagnosed with gastric adenocarcinoma of no special type (NST) and received surgical resection; b) tumors were M0 without any other malignancy; c) patients were not diagnosed with any other malignant tumor; d) patients had never received any type of chemotherapy or radiotherapy before surgery; e) patients had complete clinical data. Normal gastric tissues (5 cm from the tumor site) and gastric cancer tissues were collected by surgical resection and frozen in liquid nitrogen for further analyses. The collected tissues were diagnosed by at least two pathologists who reached an agreement. All patients never received any anti-tumor therapies or had a history of gastric cancer. This study obtained approval from the Ethics Committee of Affiliated Hospital of Chengde Medical University (Approval No. 2016135). Written informed consent was obtained from each participant. The reporting of this study confirms to the STROBE statement. 15
Cell lines and transfections
Various differentiated gastric cancer cell lines, including AGS (well-differentiated) and MKN-45 (poorly differentiated) cells, and a normal gastric epithelial cell line GES-1, were obtained from the American Type Culture Collection (ATCC; Manassas, VA, USA). All cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) (Sigma Aldrich, St. Louis, MO, USA) and penicillin (100 IU/mL) at 37°C in a 5% CO2 environment.
MKN-45 cells were transfected with 100 nM miR-301-3p mimic (Guangzhou RiboBio Co., Guangzhou, China) to overexpress miR-301-3p. Cx43 was overexpressed by transfecting cells with pcDNA 3.1-Cx43. pcDNA 3.1 (empty vector) was used as the negative control. Transfections were performed with Lipofectamine 2000 reagent (Invitrogen, Waltham, MA, USA) following the suggested protocols.
Immunohistochemistry (IHC)
Collected tissues were fixed with 4% paraformaldehyde solution, dehydrated, and embedded in paraffin. Cx43 protein expression was detected by IHC after the embedded tissues were cut into 4-µm sections. The affinity-purified rabbit polyclonal antiserum raised against the carboxy terminal 362-382 peptide segment of Cx43 was used to immunolabel Cx43 (Abcam, Cambridge, UK). The tissues were first incubated with the Cx43 primary antibody at 4°C overnight, then washed in PBS three times. Then, the secondary antibody was added and incubated at 37°C for 1 hour, followed by three more PBS washes. DAB, which yields brown reaction products, was then used as the chromogen, followed by hematoxylin counter staining. The stained tissues were observed under a microscope and analyzed with ImageJ software (https://imagej.nih.gov/ij/). The immunohistochemical scores were based on the percentage of positive cells in the observed field.
Quantitative reverse transcription PCR (RT-qPCR)
The expression levels of miR-301-3p and Cx43 mRNA were evaluated by RT-qPCR using the SYBR Green I Master Mix Kit (Invitrogen). Briefly, total RNA was isolated using Trizol Reagent (Invitrogen), then cDNA was generated using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Waltham, MA, USA) for Cx43 and the TaqMan Reverse Transcription Kit (Applied Biosystems) for miR-301-3p. The relative expression levels of miR-310-3p and Cx43 were normalized to U6 and β-actin mRNA, respectively, and calculated using the 2−ΔΔCt method. The primer sequences used were: miR‐301‐3p: forward, 5′‐TCCGACGAAACTGTTATAGTA‐3′ and reverse, 5′‐GTGCAGGGTCCGAGGT‐3′; Cx43: forward, 5′-
Cell proliferation assay
MKN-45 and AGS cells were seeded at a density of 1 × 105 cells/well in 96-well plates and cultured in DMEM with 10% FBS. The plates were incubated at 37°C with 5% CO2 for 0, 24, 48, or 72 hours, then a CCK8 Kit (Dojindo, Tokyo, Japan) was used to evaluate cell proliferation rates according to the manufacturer’s instructions. The absorbance values at 450 nm were detected using a microplate reader (Bio-Rad, Hercules, CA, USA).
Cell migration and invasion assays
MKN-45 and AGS cells were seeded at a density of 1 × 105 cells/well in the upper chambers of a 24-well Transwell chamber with a pore size of 8 µm (Thermo Fisher Scientific, Waltham, MA, USA) and cultured in serum-free DMEM. DMEM with 10% FBS was placed in the bottom chamber as a chemoattractant. The upper chambers were pre-coated with Matrigel (BD Biosciences, Franklin Lakes, NJ, USA) for the invasion assays. After incubating for 24 hours, the upper chamber and membrane were disposed. Migrated and invaded cells in the bottom chambers were stained with 0.1% crystal violet and quantified using a microscope (200× magnification).
Dual-luciferase reporter assay
The binding sites between miR-301-3p and Cx43 mRNA were predicted using the TargetScanHuman v7.2 database (http://www.targetscan.org/vert_72/). The wild-type Cx43 3ʹ UTR containing the miR-301-3p binding sites was cloned into the pGL3 luciferase reporter vector (Promega, Madison, WI, USA). A mutant version of this vector was generated by mutating the miR-301-3p binding sites. The luciferase reporter plasmid, miR-301-3p mimic, or miR-301-3p inhibitor (100 nM, Guangzhou RiboBio) were co-transfected into gastric cancer cells for 48 hours. The relative luciferase activity was evaluated with the Dual-Luciferase Reporter Assay Kit (Promega) according to the manufacturer’s instructions.
Statistical analysis
All data are presented as mean ± standard deviation (SD) obtained from at least three experiments. Statistical analyses were performed with the help of SPSS 23.0 software (IBM Corp., Armonk, NY, USA) and GraphPad Prism 7.0 (GraphPad Software, Inc., San Diego, CA, USA). Differences among three or more groups were analyzed by a one-way analysis of variance, while the differences between two groups were evaluated by a Student’s t-test. The differences were considered to be statistically significant when P < 0.05.
Results
Correlation of miR-301-3p and Cx43 expression in gastric cancer
miR-301-3p was significantly upregulated in well-differentiated and poorly differentiated gastric cancer tissues compared with normal tissues (P < 0.001), while there was no significant difference between the various differentiated tissues (P > 0.05, Figure 1a). miR-301-3p expression levels were also dramatically increased in well-differentiated and poorly differentiated gastric cancer cell lines compared with the normal gastric epithelial cell line GES-1 (P < 0.001, Figure 1b). On the contrary, Cx43 mRNA expression was dramatically downregulated in well-differentiated and poorly differentiated tissues (P < 0.001, Figure 1c). Moreover, relatively lower Cx43 expression was also found in gastric cancer cell lines compared with GES-1 cells (P < 0.001, Figure 1d). The expression levels of miR-301-3p were significantly negatively correlated with Cx43 mRNA levels, with a correlation coefficient value of −0.602 (P < 0.001, Figure 1e).

The expression levels and correlation of miR-301-3p and Cx43 mRNA in gastric cancer patient samples. (a) miR-301-3p was significantly upregulated in well-differentiated and poorly differentiated gastric cancer patient tissue samples compared with normal gastric tissues. (b) MiR-301-3p was significantly upregulated in AGS (well-differentiated) and MKN-45 (poorly differentiated) cells compared with the normal gastric cell line, GES-1. (c) Cx43 mRNA was significantly downregulated in gastric cancer patient tissue samples with various differentiation degrees compared with normal gastric tissues. (d) Cx43 mRNA was downregulated in AGS (well-differentiated) and MKN-45 (poorly differentiated) cells compared with GES-1 cells. (e) The expression levels of miR-301-3p were negatively correlated with Cx43 mRNA levels. r = −0.602, P < 0.001. ***P < 0.001 relative to normal tissues and cell lines.
Clinical significance of Cx43 and miR-301-3p in gastric cancer
According to the median Cx43 expression value in gastric cancer tissues, the 60 patients were divided into low expression and high expression groups. Patients that had relatively low Cx43 expression showed poor differentiation (n = 29, P = 0.001) and advanced TNM stage (n = 25, P = 0.004), and possessed vascular invasion (n = 26, P = 0.007) and lymph node metastasis (n = 23, P = 0.020). Similarly, miR-301-3p upregulation was also associated with poor tumor differentiation (P = 0.002), advanced TNM stage (P = 0.006), positive vascular invasion (P = 0.002), and positive lymph node metastasis status (P = 0.001) of gastric cancer patients (Table 1).
Associations between the expression levels of Cx43 or miR-301-3p and the clinical characteristics of gastric cancer patients.

Cx43 is a direct target of miR-301-3p
miR-301-3p could bind to the 3ʹ UTR of Cx43 mRNA, and these binding sites are shown in Figure 2a. The relative luciferase activity of the Cx43 reporter vector was inhibited following transfection of the miR-301-3p mimic, but was enhanced when the miR-301-3p inhibitor was transfected. However, the relative luciferase activity of the mutant vector was not affected by any of these transfections. These results indicate that a direct interaction occurs between miR-301-3p and Cx43 (P < 0.001, Figure 2b).

Cx43 mRNA is a direct target of miR-301-3p. (a) The binding sites between miR-301-3p and Cx43 wild-type (Cx43 WT) or Cx43 mutant-type (Cx43 MT). (b) Overexpression of miR-301-3p through mimic transfection significantly suppressed the luciferase activity of Cx43 WT, while knockdown of miR-301-3p with a specific inhibitor enhanced the luciferase activity of Cx43. The luciferase activity of Cx43 MT was not affected by miR-301-3p expression modulation. ***P < 0.001 relative to the mock group.
The biological function of Cx43 in the progression of gastric cancer
MKN-45 and AGS cells were co-transfected with pcDNA 3.1-Cx43 and miR-301-3p mimic to co-regulate the expression of Cx43 and miR-301-3p. As depicted in Figure 3a, the transfection of miR-301-3p mimic notably inhibited the expression of Cx43 (P < 0.001). Cx43 expression was significantly enhanced by the transfection of pcDNA 3.1-Cx43, which was reversed by the co-transfection of miR-301-3p mimic (P < 0.01, P < 0.001). The proliferation rates of MKN-45 and AGS cells were promoted by miR-301-3p mimic transfection and inhibited by Cx43 overexpression (P < 0.01, Figure 3b). miR-301-3p mimic transfection could also reduce the inhibitory effect of Cx43 upregulation on the proliferation of MKN-45 and AGS cells (P < 0.05, Figure 3b).
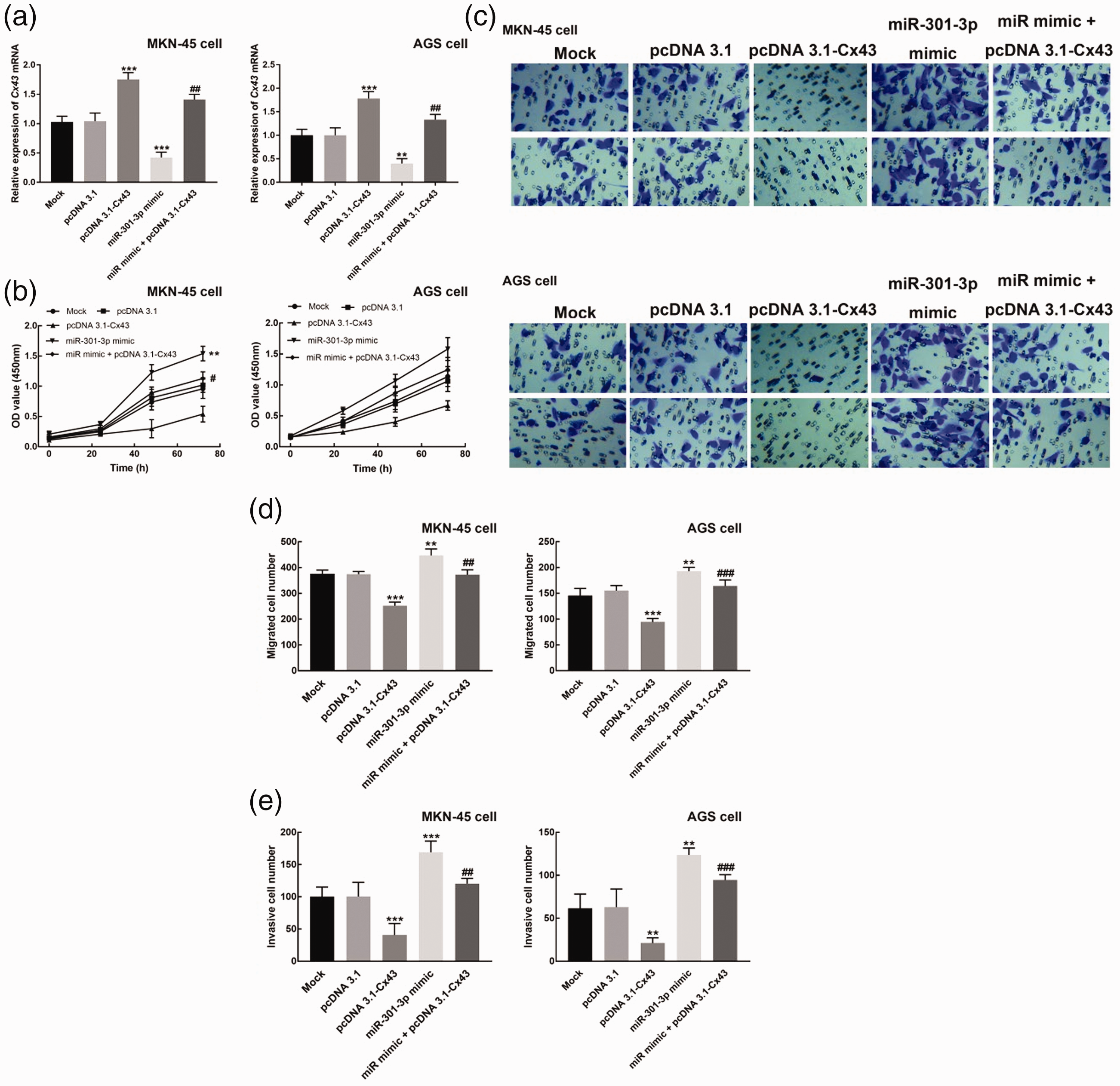
Cx43 affects various cell processes of gastric cancer cells. (a) Cx43 expression was inhibited by miR-301-3p mimic transfection, but promoted by pcDNA3.1-Cx43 transfection. The co-transfection of miR-301-3p mimic and pcDNA 3.1-Cx43 could reverse the upregulation of Cx43 by pcDNA3.1-Cx43. (b) The overexpression of Cx43 significantly suppressed the proliferation of MKN-45 cells, while miR-301-3p mimic transfection exerted the opposite effect. The co-transfection of miR-301-3p mimic and pcDNA 3.1-Cx43 could reverse the inhibitory effect of Cx43 overexpression on the proliferation of gastric cancer cells. (c) Representative images of the Transwell assays of the migration (top) and invasion (bottom) of MKN-45 and AGS cells. (d) The overexpression of Cx43 significantly suppressed the migration of MKN-45 cells, while miR-301-3p mimic transfection promoted the migration of MKN-45 cells dramatically. The co-transfection of miR-301-3p mimic and pcDNA 3.1-Cx43 could reverse the inhibitory effect of Cx43 overexpression on the migration of gastric cancer cells. (e) The overexpression of Cx43 significantly suppressed the invasion of MKN-45 cell, while miR-301-3p mimic transfection promoted the invasion of MKN-45 cells dramatically. The co-transfection of miR-301-3p mimic and pcDNA 3.1-Cx43 could reverse the inhibitory effect of Cx43 overexpression on the invasion of gastric cancer cells. **P < 0.01, ***P < 0.001 relative to the mock group. #P < 0.05, ##P < 0.01 relative to the pcDNA3.1-Cx43 group.
Additionally, the migration and invasion rates of MKN-45 and AGS cells were inhibited by Cx43 overexpression, but promoted by miR-301-3p upregulation (P < 0.001, P < 0.01, Figure 3d and e). However, the co-transfection of miR-301-3p mimic and pcDNA 3.1-Cx43 could reverse the inhibited migration and invasion of gastric cancer cells (P < 0.01, Figure 3d and e).
Discussion
The differentiation degree of cancer tissues is associated with disease development and clinical outcome of patients, together with other factors.16,17 Exploring molecular biomarkers associated with the development of gastric cancer could improve the diagnostic efficiency and risk prediction accuracy. Previous studies have identified a variety of biomarkers that affect the proliferation, migration, and invasion of gastric cancer cells and are involved in the diagnosis and prognosis of this disease.18,19 For example, prospero homeobox protein-1 (PROX1) could influence the proliferation, migration, and invasion of MKN-45 cells and was positively correlated with the cancer stage of patients, indicating that PROX1 is a possible indicator and molecular therapeutic target of gastric cancer. 20
Cx43 is a vital component of gap junction intercellular communication, which has previously been associated with the recurrence and metastasis of cancers, as well as with poor survival of patients. Cx43 inhibited the invasion and migration of cisplatin-resistant cells in testicular cancer. 21 Cx43 was downregulated in gastric cancer and was associated with the positive peritoneal metastasis of gastric tumors. 11 Several miRNAs were reported to regulate Cx43 expression in various diseases. For example, miR-382-3p could suppress the inflammatory response of chondrocytes induced by IL-1β via mediating Cx43 levels. 22 The miR-218-5p-Cx43 axis contributes to gemcitabine chemoresistance of breast tumors and can regulate the growth, migration, and invasion of breast cancer cells. 23 However, the clinical significance and biological function of Cx43, as well as its interaction with miR-301-3p, were still unclear for gastric cancer.
Consistent downregulation of Cx43 mRNA was observed in both gastric cancer tissues and cell lines with various differentiation degrees. Downregulated Cx43 was closely associated with the differentiation, TNM stage, vascular invasion, and lymph node metastasis status of gastric cancer patients, suggesting that these important clinical features could possibly indicate the disease development of these patients. Furthermore, these findings indicate that Cx43 is involved in the progression of this cancer. 24 miR-301-3p expression levels were also related to the critical characteristics of patients, as well as negatively correlated with Cx43 mRNA levels. This implies a potential interaction between miR-301-3p and Cx43 mRNA in the development of gastric cancer. miR-301-3p was reported to bind to the 3ʹ UTR of Cx43 mRNA and be involved in bacterial meningitis pathogenesis. 12 Interestingly, although both Cx43 and miR-301-3p expression were significantly associated with the differentiation of patients and showed a decreased trend in poorly-differentiated gastric cancer tissues, the differences were not statistically significant. The limited sample size may have contributed to these results. Therefore, a relatively large sample size is needed in further studies. Additionally, bias was potentially a factor during patient enrollment and may have limited the obtained clinical results.
For the biological function of Cx43, we found that overexpression of Cx43 could suppress the proliferation, migration, and invasion of gastric cancer cells, indicating that Cx43 has a tumor suppressive role in these cells. miR-301-3p reportedly serves as a tumor promoter in gastric cancer by accelerating cell proliferation and suppressing apoptosis, which is consistent with the results of our present research. 14 Moreover, transfection of a miR-301-3p mimic significantly inhibited the expression of Cx43 and reversed the inhibitory effect of Cx43 overexpression on proliferation, migration, and invasion of gastric cancer cells. Overall, these results suggest that the miR-301-3p-Cx43 axis could indicate the development of gastric cancer and mediate the proliferation, migration, and invasion of gastric cancer cells.
A further mechanism underlying the function of the miR-301-3p-Cx43 axis was not uncovered in this study. The mTOR/p70s6k signaling pathway was considered to be critical for the growth and apoptosis of various cancer cells, such as esophageal squamous cell carcinoma, ovarian cancer, and prostate cancer.25–27 Whether the mTOR/p70s6k signaling pathway is involved in the functional processes of the miR-301-3p-Cx43 axis requires validation in further studies. Additionally, Cx43 is an important member of the connexin protein family, which could improve the intercellular communication between cells and facilitate the transfer of signaling molecules. 4 The effects of the miR-301-3-Cx43 interaction on gap junctions were not evaluated, so future studies are needed.
In conclusion, we show that the miR-301-3p-Cx43 axis is significantly associated with the disease development of gastric cancer patients. miR-301-3p can regulate Cx43 expression to mediate proliferation, migration, and invasion of gastric cancer cells.
