Abstract
Objective
We aimed to determine the ocular manifestation and refractive error prevalences among people living with HIV (PLHIV) in Pune, India.
Methods
This cross-sectional study included HIV-infected adults attending a National AIDS Research Institute clinic. Ophthalmologic examination included visual acuity estimation, refraction, orthoptic evaluation, slit lamp and fundus examination, and photography.
Results
In total, 441 HIV-infected individuals were enrolled. The participants' median age was 44 (interquartile range 38–49) years and 227 (51.5%) were men. Refractive errors occurred in 132 (29.9%) individuals. Ocular manifestations were present in 93 (21.1%) participants and the most frequent was cataract in 59 (13.4%) participants. Multivariate logistic regression analysis showed that participants who were illiterate (adjusted odds ratio [AOR] 2.80, 95% confidence interval [CI] 1.47–5.33) and those aged greater than 40 years (AOR 5.59, 95% CI 2.69–11.61) were more likely to have ocular manifestations. The odds of having ocular manifestations were greater in participants with treatment substitution or changes (AOR 2.11, 95% CI 1.16–3.82).
Conclusions
Cataract and refractive errors were prevalent among PLHIV. PLHIV should be encouraged to have regular ophthalmic checkups. Individuals with lower education levels and older age should be counseled regarding eye care and timely reporting of ocular symptoms.
Introduction
Nearly 34 million people are currently living with HIV/AIDS and ocular complications are common in these individuals, affecting 50% to 75% of people living with HIV (PLHIV) at some point during the course of their illness. 1 The spectrum of ocular complications with HIV infection may be changing in the current era owing to the impact of highly active antiretroviral therapy (HAART). 2 Although antiretroviral therapy (ART) has reduced the risk of opportunistic infections, newer manifestations of known diseases and ocular lesions as a result of adverse drug reactions are being seen. 3 There has also been recognition of new clinical presentations in the form of immune recovery uveitis, which is now a leading cause of severe vision loss in patients with HIV. 4 Ocular opportunistic infections may present with significant inflammatory reactions in patients receiving ART. Ocular tuberculosis (TB) is common in India, although not to the extent of systemic TB among PLHIV. Paradoxical worsening post-TB infection has also been reported in PLHIV who are receiving HAART and antitubercular treatment (ATT). 5 HAART has been reported to cause recrudescence of uveitis in patients who are severely immunocompromised. Improvement in CD4 counts can lead to signs of uveitis. 6 Literature is scarce regarding the impact of HIV on refractive errors. A study in the United Kingdom identified accommodative failure in a considerable proportion of PLHIV aged between 26 and 35 years. 7 Accommodative failure leads to impaired near vision, which is important for tasks of daily living such as using mobile phones, recognizing currency, and household chores apart from reading and writing. Although ocular morbidity in PLHIV may not be vision threatening in the HAART era, it may affect quality of life. With this background, we aimed to investigate ocular manifestations and refractive errors among PLHIV from Pune, India.
Methods
Study design, setting, and selection of participants
This study was conducted in a clinic at the Indian Council of Medical Research-National AIDS Research Institute (ICMR-NARI). This outpatient clinic is affiliated with the National AIDS Control Organization (NACO) and caters to a proportion of PLHIV population in Pune city. Adults (age ≥18 years) were included in the study from January 2019 through September 2019. The study was approved by the Institutional Ethics Committee of ICMR-NARI (approval number NARI EC/2018-23), and each participant was recruited after we had obtained their written informed consent. No personal identifier was linked with any of the study data for dissemination purposes. The reporting of this study conforms to the STROBE statement. 8
Sample size was estimated to detect a prevalence of 30% of ocular manifestations 9 among PLHIV, with absolute precision of 5% at a 95% confidence level. The number of individuals required for this observational study was 355, with a 10% non-response rate, randomly drawn from the list of regular attendees at the ICMR-NARI clinic. Individuals with newly detected HIV infection and those who recently initiated ART under “test and treat” guidelines of the NACO during the study period were also enrolled (ART duration ≤6 months).
Data collection
A semi-structured questionnaire was used to collect sociodemographic, clinical, and laboratory data pertaining to investigations conducted earlier. ART cards served as an additional data source in this regard. Ocular examinations were conducted by a trained ophthalmologist and the findings were entered into a database created for each participant. Approximately 30 minutes were required for each participant to complete the study procedures.
Ocular examination
Distance and near vision were tested using the Snellen distance and Jaeger near vision charts, respectively. Anterior segment examination was carried out using a three-step slit lamp biomicroscope and intraocular pressure was measured using a non-contact tonometer. Color perception was measured using the Ishihara test and contrast sensitivity was assessed with the Pelli–Robson chart. Contrast sensitivity was measured as it may be affected prior to visual acuity in early optic nerve dysfunction. Ocular motility was also examined. Study participants underwent refraction for distance and near vision. Fundus examination with direct and indirect ophthalmoscopy was conducted. Positive findings were documented using a digital camera and recorded on a predesigned clinical record form. Spectacles were dispensed free of cost to the study participants.
Laboratory tests
Blood samples were tested via routine hemogram, biochemistry, and CD4 counts. We carried out serologic tests (enzyme-linked immunosorbent assay for immunoglobulin IgM and IgG antibodies) to detect Toxoplasma and Cytomegalovirus infection using commercial kits (Dia.pro, Milan, Italy). Rapid plasma reagin (RPR) and Treponema pallidum hemagglutinin assay (TPHA) were performed to rule out syphilis. All samples were sent to SRL Diagnostics Laboratory in Mumbai, which follows internal and external quality control procedures including a Bio-Rad Monthly Program, Compliance Audit Program, and National Accreditation Board for Testing and Calibration Laboratories.
Outcome measure and explanatory variables
In this study, the outcome variable was ocular manifestations as diagnosed clinically. Any anatomical deviation or pathology or loss of function in the eye was considered an ocular manifestation. In each participant, if one eye had better vision than the other, the former was considered the better eye and the latter was considered the eye with lower visual acuity in the analysis. Visual acuity assessment was conducted and the better eye was classified per World Health Organization classification 10 as blind (<3/60), severe visual impairment (3/60 to <6/60), moderate visual impairment (6/60 to 6/24), mild visual impairment (6/18), and no visual impairment (6/12 to 6/5). A refractive error of −0.5 sphere or greater magnitude was considered myopia. A refractive error of +0.75 or more was considered hypermetropia. Refractive error that differed by 0.75 or more in any meridian was classified as astigmatism. Presbyopia was defined as the inability to read the N6 line even with distant correction but able to do so with the addition of plus lenses.
Age, sex, education, income, marital status, employment, alcohol use, and smoking-related information were collected as explanatory variables. Other exposure-related information collected to explain the outcome of interest were type of ART regimen, duration on ART, regimen changes or substitutions, diabetes mellitus, hypertension, Cytomegalovirus and Toxoplasma seropositivity, and CD4 count at study entry. Participants having one or more ocular manifestations and those with no ocular manifestations were categorized accordingly. In this study, we used a cutoff of 6/18 vision for the better eye and the eye with lower visual acuity. Visual acuity of less than 6/18 was defined as visual impairment. 10
Statistical analysis
We analyzed the association of explanatory variables with ocular manifestations using binary logistic regression. Variables with a significant association in univariate analysis were entered into a multivariate logistic regression model. We reported odds ratios (ORs) with their respective 95% confidence intervals (CIs) for all associations. IBM SPSS 20.0 (IBM Corp., Armonk, NY, USA) was used for the data analysis. The demographic and clinical characteristics of participants were summarized using mean (standard deviation; SD) and median (interquartile range; IQR).
Results
Demographic and clinical characteristics
We enrolled 441 individuals in the study, among which 55 (12.5%) were ART naïve, 53 (12%) had initiated ART within 6 months, and 333 (75.5%) were on ART for more than 6 months. Nearly half of participants (227/441, 51.5%) were men. Participants’ median age and CD4 count at study enrollment were 44 years (IQR 38–49) and 473.5 cells/mm3 (IQR 281.8–685.0), respectively. Of the total participants, 374 (84.8%) were literate and 334 (75.7%) were employed. Two hundred and seventy-four (62.1%) participants were married and living with a partner. Median duration of HIV infection detected among participants was 7.9 years (IQR 2.7–12.1). Of the 386 participants receiving ART, 148 (38.3%) were on a tenofovir-based regimen and 173 (44.8%) were on a zidovudine-based regimen at enrollment. The median duration on ART was 7 years (IQR 2.5–9.8). As per NACO technical guidelines, the ART regimen was either changed or substituted among 184 (47.7%) participants for various reasons such as treatment failure, toxicity, ATT, or a change in the national guidelines. Diabetes was present in 16 (3.7%) PLHIV and hypertension in 157 (35.7%). Thirteen (3%) participants were reactive for syphilis (RPR and TPHA). Cytomegalovirus IgM and Toxoplasma IgM antibodies were detected in 15 (3.4%) and 12 (2.7%) participants, respectively.
Ocular manifestations
One-fifth of participants (93/441, 21.1%) had ocular manifestations (Table 1). The most frequent finding was cataract in 59 (13.4%) participants, followed by pseudophakia in 7 (1.6%), corneal opacity in 4 (0.9%), and glaucoma in 4 (0.9%) individuals. The mean age of individuals with cataract or pseudophakia was 55.3 (SD: 8.9) years.
Demographic factors associated with ocular manifestations among PLHIV
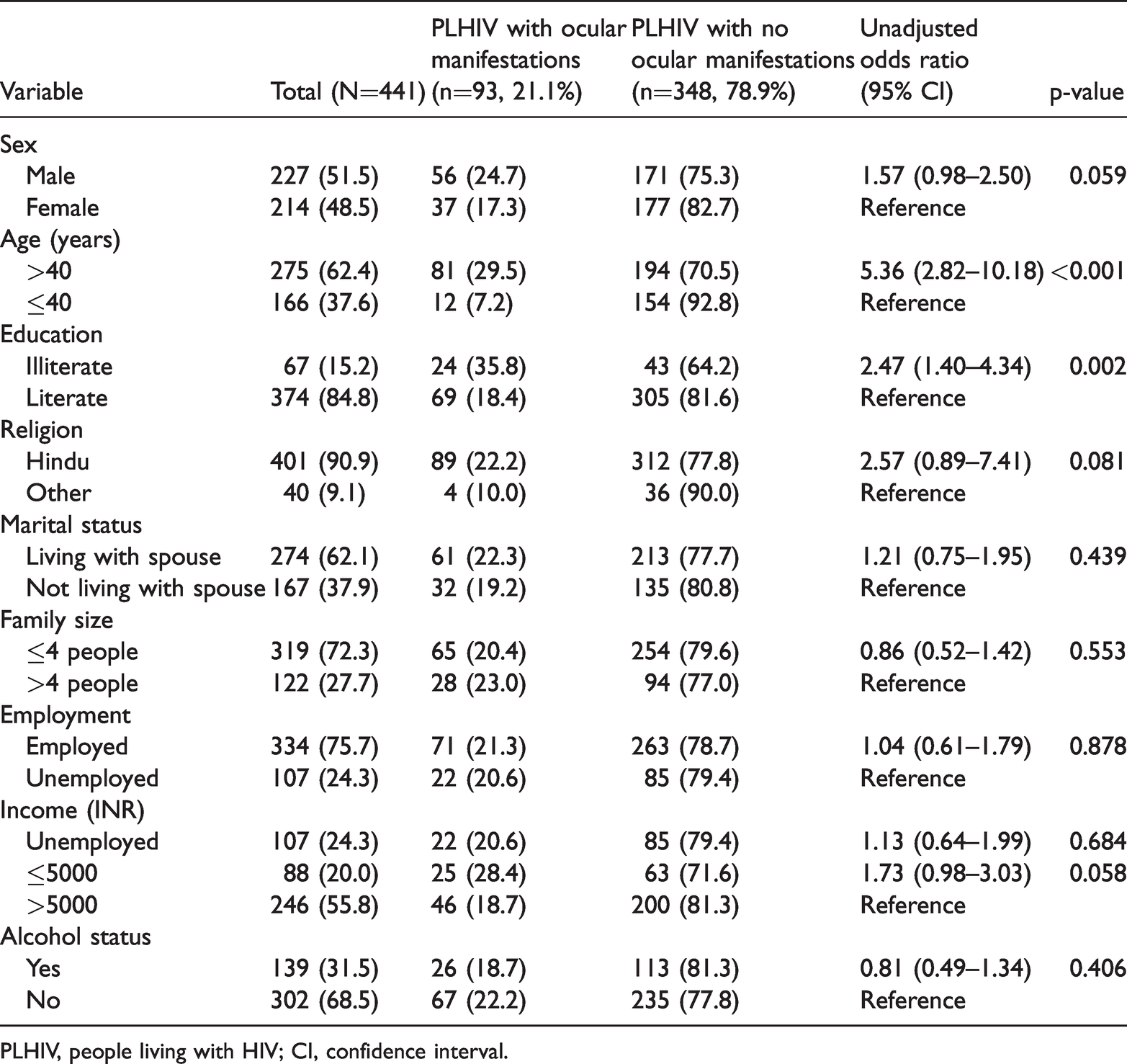
PLHIV, people living with HIV; CI, confidence interval.
Uveitis, myopic fundus, and squint were each observed in 3 (0.7%) participants whereas amblyopia and choroiditis were seen in 2 (0.5%) participants. Other manifestations such as hard exudate, hypertensive retinopathy, chorioretinal atrophy patch with dilated and tortuous blood vessels with prior retinal edema, proliferative diabetic retinopathy with vitreoretinal fibrovascular proliferation, ptosis, and pterygium were observed in 1 (0.2%) participant each.
Visual acuity, refractive error, and color perception
Ocular examination showed that 2 (0.5%) PLHIV had blindness, 5 (1.1%) had severe visual impairment, 49 (11.1%) had moderate visual impairment, 50 (11.3%) had mild visual impairment, and 335 (76%) participants had normal vision in the better eye.
Considering the eye with lower visual acuity, 16 (3.6%) participants had blindness, 5 (1.1%) had severe visual impairment, 89 (20.2%) had moderate visual impairment, 48 (10.9%) had mild visual impairment, and 283 (64.2%) PLHIV had normal vision.
Participants age above 40 years were 1.9 times more likely to have <6/18 vision (OR 1.96, 95% CI 1.03–3.71; p = 0.039) and 3.0 times more likely to have ocular symptoms (OR 3.04, 95% CI 1.27–7.31; p = 0.013) in the better eye. In participants over age 40 years with lower visual acuity, the odds of having <6/18 vision were twice those of their counterparts with better vision (OR 2.09, 95% CI 1.29–3.39; p = 0.003).
Refractive errors were detected in 132 (29.9%) individuals. Of these, hypermetropia, myopia, and astigmatism were present in 40 (9.1%), 6 (1.4%), and 65 (14.7%) participants, respectively. Twenty-one (4.8%) participants had more than one refractive error. Three hundred and eleven (70.5%) participants had presbyopia, and a significant increase was observed with increasing age (p < 0.001). Thirty of 311 (9.6%) PLHIV with presbyopia were between age 35 and 39 years and 79/311 (25.4%) were between age 40 and 44 years. Contrast sensitivity was reduced in 13 (2.9%) individuals using the Pelli–Robson chart. Eight individuals had color vision deficiencies and five had impaired color vision.
There was improvement in near vision among all participants with the spectacles provided in this study. Seventy-eight (17.7%) individuals had improvement in distant vision by at least one line owing to optical or surgical intervention. Two participants underwent cataract surgery, two received intravitreal anti-vascular endothelial growth factor injections, and panretinal photocoagulation was conducted in two participants.
Univariate analyses
Overall, one-fifth of our study participants (93/441, 21.1%) had ocular manifestations. Univariate analyses showed that participants aged more than 40 years had a greater probability of having ocular manifestations than their counterparts (OR 5.36, 95% CI 2.82–10.18; p < 0.001). Illiterate participants had higher occurrence of ocular manifestations (OR 2.47, 95% CI 1.40–4.34; p = 0.002) than literate individuals (Table 1). Ocular manifestations were twice as likely (OR 1.90, 95% CI 1.16–3.09; p = 0.011) among participants who had ART changes or substitutions to another regimen, and participants with a duration of HIV detection >1 year were approximately three times more likely to have ocular manifestations than their counterparts (OR 2.82, 95% CI 1.15–6.89; p = 0.023) (Table 2). However, no association was found between marital status, employment, duration of ART, CD4 counts at ART initiation, Toxoplasma or Cytomegalovirus seropositivity, and ocular manifestations (Tables 1–3).
Clinical factors associated with ocular manifestations among PLHIV

PLHIV, people living with HIV; ART, antiretroviral therapy; CI, confidence interval.
Laboratory factors associated with ocular manifestations among PLHIV

PLHIV, people living with HIV; ART, antiretroviral therapy; CI, confidence interval; Ig, immunoglobulin.
Multivariate analyses
Variables included in the multivariate model were age, education, duration of HIV detection, and ART changes and substitutions. After adjustment for all the aforementioned variables, factors that retained an independent association with ocular manifestations were illiteracy (adjusted OR [AOR] 2.80; 95% CI 1.47–5.33) and age more than 40 years (AOR 5.59, 95% CI 2.69–11.61). Moreover, the odds of ocular manifestations among PLHIV with ART changes or substitutions were twice those of their counterparts (AOR 2.11, 95% CI 1.16--3.82) (Table 4).
Multivariable analyses of association between variables and ocular manifestation among PLHIV

PLHIV, people living with HIV; CI, confidence interval, ART, antiretroviral therapy.
Discussion
Various reports have shown that ocular manifestations commonly occur among HIV-infected individuals in India.11–15 In the present study, most participants were receiving ART and the overall prevalence of ocular manifestations was 21.1%. Sight-threatening ocular morbidity in our study was lower than what has been cited in other reports from India.3,11–15 The relatively lower prevalence of sight-threatening morbidities in our study might be owing to differences in the study design, duration of ART, and CD4 count at study initiation. A study by Karnataka reported a prevalence of 37.6% among HIV-infected individuals in an ART center where PLHIV with ocular complaints were referred to an ophthalmology clinic for treatment. 12 Studies from other parts of the country have reported a prevalence of 40% to 63%, which is higher than that in our study.16,17 It is expected that the prevalence of ocular manifestations would be higher in symptomatic individuals. In Chennai, a study was conducted among 1000 consecutive HIV-infected individuals, with a reported prevalence of ocular manifestations of 68.5%. 3 This high prevalence might be owing to a lower median CD4 count at study initiation (224.5 cells/mm3) as compared with our study (473.5 cells/mm3), as well as the study site being a referral center from where participants were recruited. 3 A study conducted in Mumbai that included participants with lower CD4 counts (<200 cells/mm3) as the preliminary survey conducted among patients with higher CD4 counts (>200 cells/mm3) did not find any ocular complications. 18 The authors mentioned that better immunological function might have been responsible for the reduction in the burden of ocular manifestations in their study. 18
Noteworthy findings in our study were those regarding cataract, followed by those involving corneal opacity and glaucoma. The mean age for cataract in our study was similar to that reported in a study conducted among a general population. 19 However, the prevalence of cataract in our study population was higher than that reported amongst PLHIV elsewhere in India.20–22 This might be owing to the use of slit lamp and dilated fundus examination conducted in all individuals who did not improve to 6/6, where even small lenticular opacities would have been detected. A report on cataract risk in HIV-infected individuals stated that compartmentalized residual intraocular HIV viremia, despite ART, and secondary chronic inflammation could be factors promoting cataract in these individuals. 23 That report highlighted the need for conducting more studies to assess whether compartmentalized intraocular HIV replication persists with ART and whether any link exists with subsequent cataract risk. The prevalence of corneal opacities in our study concords with that of Karnataka 12 whereas uveitis appeared to be less prevalent in comparison with previous reports.4,24 Overall, the observed prevalence of corneal opacity, uveitis, and glaucoma was higher in our study than in studies conducted among several populations. 25
Surprisingly, HIV-related ocular diseases, especially posterior segment findings, were uncommon in our study. This might be owing to the current government guidelines of “test and treat” and the newer, more effective regimen in the national program. Because previous guidelines had lower cutoffs for CD4 count at ART initiation, more HIV-related ocular diseases were observed in past reports, especially posterior segment findings.
We found that participants with lower education levels had a comparatively higher risk of ocular manifestations. A cross-sectional study among Asian Malays (≥40 years old) in Singapore reported that inadequate literacy was independently associated with visual impairment and poorer visual functioning. 26 Identifying issues and developing strategies to improve literacy around visual impairment and improving patient–clinician communication would help to address such issues.
Studies have revealed that HIV infection may accelerate the aging process in individuals27–30 and that such changes might be reflected in the eyes. Some studies have reported corneal endothelial cell loss, increased crystalline lens cell density, loss of retinal nerve fiber layer, and retinal vessel caliber as evidence of accelerated aging among HIV-infected individuals.31–33 Presbyopia, caused by a reduction in the amplitude of accommodation, has been reported as an age-related change in some studies.25,34
We found more than twice the number of participants with hypermetropia, as compared with myopia, although population-based studies have shown hypermetropia to be less common than myopia.25,35 A decrease in the amplitude of accommodation can manifest as latent hypermetropia. The reduction in the amplitude of accommodation in our study at a comparatively early age might also be indicative of an accelerated aging process. A larger sample with a longer follow-up is needed to determine whether the onset of cataract and presbyopia is earlier among PLHIV. We did not observe a significant association of comorbidities and lower CD4 counts with ocular manifestations. HIV-infected individuals in our study showing antibodies to Cytomegalovirus and Toxoplasma did not have any ocular manifestations.
Limitations in this cross-sectional study included the inability to determine a causal relationship. This study showed that ocular vascular and inflammatory conditions were less common and age-related conditions like cataract and presbyopia were more common among PLHIV; however, owing to the study design, we could not determine whether HIV accelerates ageing and we could not compare the findings with controls. Additionally, the severity of cataract could not be graded owing to the cross-sectional design. Long-term follow-up studies will be useful to provide in-depth information related to cataract among PLHIV. Data on ocular manifestations in some of our participants might have been missed owing to recall bias. Last, we did not specifically investigate dry eye, although dry eye is common among PLHIV according to some reports.
Conclusions
The findings of this study highlight the need for increasing awareness among health care providers regarding ocular manifestations in PLHIV. The National AIDS Control Programme recommends fundus examination for PLHIV with low CD4 counts. We wish to emphasize that regular counseling on ocular manifestations should be considered for PLHIV irrespective of age or CD4 count. Referral to an ophthalmology clinic in a timely manner is also needed for early diagnosis and treatment of ocular manifestations and refractive errors, to improve quality of life among PLHIV.
Footnotes
Acknowledgments
The authors acknowledge Dr. Meena Kharat for help with the ocular examination and Mr. Shrivallabh Sane for supporting the data analysis. The authors sincerely thank The Community Eye Care Centre for providing funding to distribute free spectacles to all participants with refractive errors. The authors acknowledge Mrs. Sunita Kumbhar and Mr. Vinayak Chavan for their help with technical support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received intramural funding from the ICMR-National AIDS Research Institute, Pune. The ophthalmology examination instruments and spectacles for PLHIV were provided by the Community Eye Care Foundation in Pune.
