Abstract
Beta-lactam antibiotics commonly cause immune thrombocytopenia. Cross-reactivity in patients with drug-induced immune thrombocytopenia has rarely been reported. In this study, we describe the case of a 79-year-old man who developed thrombocytopenia after receiving piperacillin–tazobactam for an acute exacerbation of chronic obstructive pulmonary disease, and he was successfully treated with meropenem and cefotiam. However, thrombocytopenia recurred after cefoperazone–sulbactam administration. This indicated that cross-reactivity of platelet-specific antibodies occurred between piperacillin–tazobactam and cefoperazone–sulbactam. However, the responsible drug structures remain unknown, requiring further investigation. Likewise, chemical structure similarities among beta-lactam antibiotics must be examined to determine the risk of immune thrombocytopenia in the clinical setting.
Introduction
Drug-induced thrombocytopenia (DIT) is a sudden and severe hematologic complication. Diagnosing DIT remains a challenge because its etiology is complex and multifactorial. It can have fatal outcomes with a platelet count nadir of <100,000/µL and a decrease of ≥30%. 1 The incidence of DIT is 10 cases per million population per year, with a prevalence of approximately 25% in critically ill patients. 2
Beta-lactam antibiotics commonly cause thrombocytopenia. They can decrease platelet production secondary to bone marrow suppression; however, these drugs are more likely to cause drug-induced immune-mediated thrombocytopenia (DITP). 3 Cross-reactivity might exist between beta-lactam antibiotics because of the immunogenic nature of DITP and the structural similarities between the drugs. Some case reports or series on DITP suggest that no cross-reaction occurs between the penam and cephem groups.4–6 However, these reports involved few types of beta-lactam antibiotics, and they did not identify the antibodies responsible for DITP. Therefore, the safety of using an alternative beta-lactam antibiotic must be determined in patients with DITP.
In this case report, we describe a patient with immune thrombocytopenia induced by piperacillin–tazobactam that was successfully treated with meropenem and cefotiam but not cefoperazone–sulbactam.
Case report
In July 2020, a 79-year-old man presented with an acute exacerbation of cough, dyspnea, and wheezing. The patient had a history of type 2 diabetes mellitus, hypertension, and chronic obstructive pulmonary disease (COPD), for which he was receiving gliclazide, losartan, and budesonide–formoterol, respectively. Physical examination, laboratory testing, and imaging studies revealed multifocal pneumonia. The baseline complete blood count (CBC) revealed a hemoglobin level of 145 g/L, white blood cell (WBC) count of 10.34 × 109/L, and platelet count of 150 × 109/L. The patient was administered doxofylline, bromhexine, methylprednisolone, and piperacillin–tazobactam (4.5 g IV guttae q8h). On day 4 of hospitalization, his cough and shortness of breath had improved. Repeat laboratory investigation disclosed a hemoglobin level of 133 g/L, WBC count of 8.3 × 109/L, and platelet count of 76 × 109/L. On day 8 of hospitalization, the patient’s general condition was good, and he was discharged from the hospital.
In March 2021, the patient was re-hospitalized for acute exacerbation of COPD (AECOPD) and pulmonary infection. The baseline CBC revealed a hemoglobin level of 115 g/L, a WBC count of 9.91 × 109/L, and platelet count of 149 × 109/L. The patient was administered the same medications used in the previous admission, except that piperacillin–tazobactam (4.5 g IV guttae q8h) and levofloxacin (0.5 g IV guttae qd) were administered for pulmonary infection. However, on day 3, repeat laboratory investigation revealed a rapid decrease of the platelet count to 25 × 109/L. Other hematologic indices were unremarkable, and the patient had no neurological deficits, purpura, petechiae, mucosal bleeding, or epistaxis. The condition was considered thrombocytopenia associated with antibiotics; therefore, piperacillin–tazobactam and levofloxacin were discontinued. Meropenem (1 g IV guttae q8h) was administrated to complete the treatment of pulmonary infection. His platelet count gradually increased to 96 × 109/L 9 days after discontinuing piperacillin–tazobactam (Figure 1), and no other treatments were given. On day 14 of hospitalization, improvement of the patient’s cough and dyspnea was noted, and he was discharged from the hospital. On day 7 post-discharge, the patient was followed-up in the outpatient clinic, and his platelet count had returned to normal.
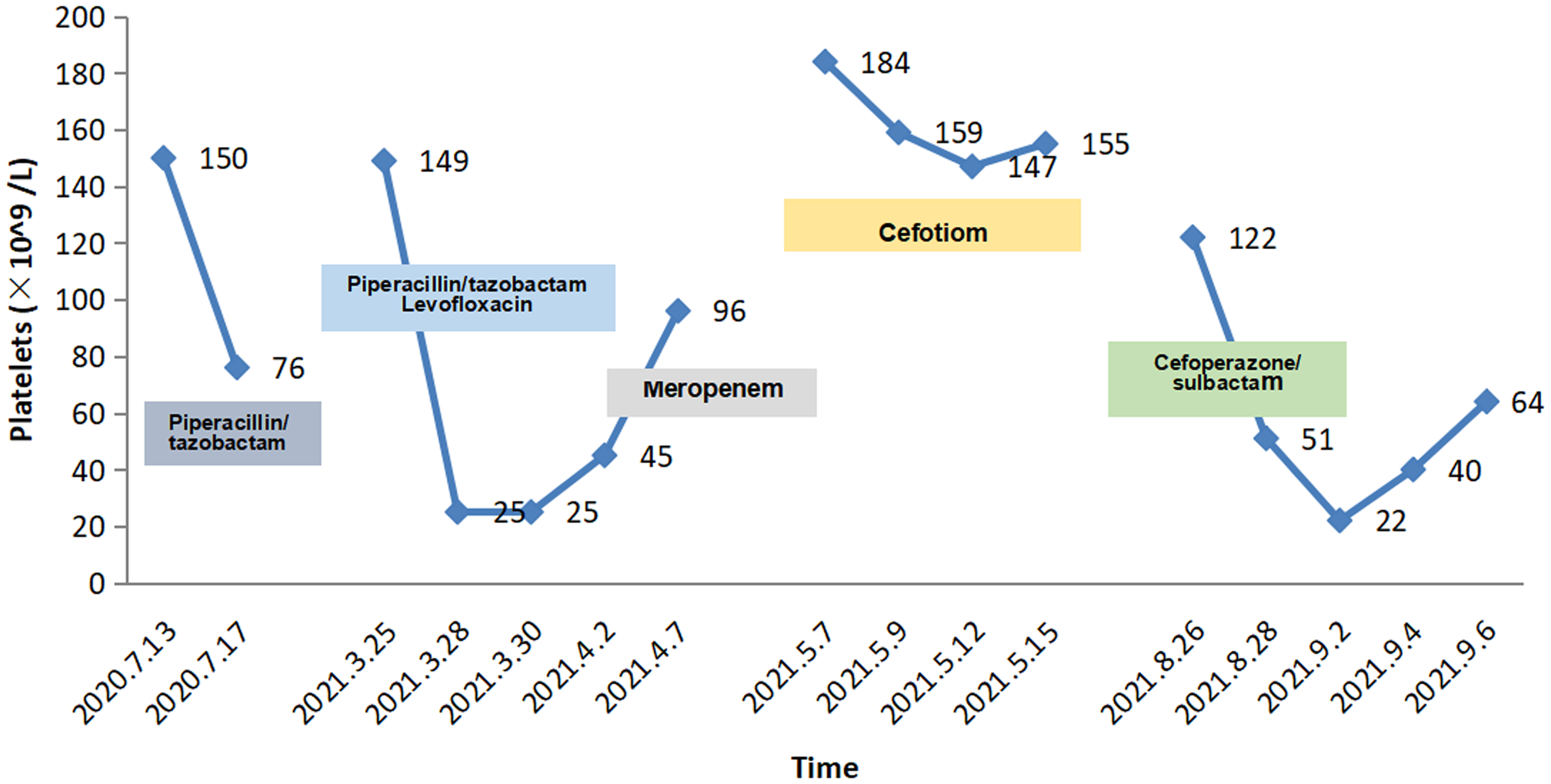
Depiction of the patient’s time course of thrombocytopenia and exposure to potentially causative antibiotics.
In May 2021, the patient was re-admitted for AECOPD and pulmonary infection with an initial platelet count of 184 × 109/L. The patient was administered the same medications used in the previous admission, except that cefotiam (2 g IV guttae q12h) was administered for infection. The platelet counts on days 3 and 9 of hospitalization were 159 × 109 and 155 × 109/L, respectively. On day 11 of hospitalization, improvements of the patient’s cough and sputum were noted, and he was discharged.
In August 2021, the patient was readmitted for AECOPD and pulmonary infection. The baseline CBC revealed a hemoglobin level of 119 g/L, WBC count of 7.71 × 109/L, and platelet count of 122 × 109/L. The anti-infection regimen was switched to cefoperazone–sulbactam (4 g IV guttae q12h). However, his platelet count decreased to 51 × 109/L after 2 days of administration and 22 × 109/L after 7 days. Meanwhile, his WBC count was 5.68 × 109/L, and his hemoglobin level was 118 g/L. The patient had no neurologic deficits, purpura, petechiae, mucosal bleeding, or epistaxis. The clinical pharmacist examined the patient’s medication list (Figure 1) and thought that thrombocytopenia was associated with antibiotic use. Further, according to the grading criteria for blood and lymphatic disorders, the severity of the condition was classified as grade 3. 7 Cefoperazone–sulbactam was discontinued because it was the suspected cause of thrombocytopenia. Within 4 days, the patient’s platelet count increased from 22 × 109/L to 64 × 109/L (Figure 1). His cough and shortness of breath improved, and he was discharged. After 2 weeks of follow-up, the platelet count had returned to normal.
The patient and his family provided approval for his therapeutic plan. Written informed consent was obtained from the patient for publication of the case details. The reporting of this study conforms to CARE guidelines. 8 This case report did not contain personal medical information about an identifiable individual. Therefore, the requirement for ethics approval was waived.
Discussion
In this study, we described the case of a patient with a history of COPD. The patient required antimicrobial therapy for pulmonary bacterial pneumonia; however, the treatment was complicated by thrombocytopenia associated with piperacillin–tazobactam use. The patient was successfully treated with meropenem and cefotiam; however, the patient did not tolerate cefoperazone–sulbactam.
Diagnosing DIT remains a challenge because its etiology is complex and multifactorial. Therefore, doctors should be aware of the risks of DIT, and drugs should be discontinued following the detection of this condition. The Naranjo scale was used to assess the causal relationship between the development of thrombocytopenia and the use of piperacillin–tazobactam and cefoperazone–sulbactam. 9 This yielded a probability score of +9 for piperacillin–tazobactam, suggesting that it was the definite cause of thrombocytopenia, and the probability score for cefoperazone–sulbactam of +7 suggested that it was a probable cause of thrombocytopenia. We further used the criteria for evaluating DIT and obtained the same results.10,11
Two mechanisms underlying beta-lactam antibiotic-induced thrombocytopenia have been proposed: (1) drug-induced bone marrow suppression and (2) DITP. However, DITP, an idiosyncratic immune-mediated reaction, is more common. 3 Two mechanisms underlying beta-lactam antibiotic DITP have been elucidated.12,13 A limited number of case reports have confirmed DITP caused by piperacillin–tazobactam by identifying positive drug-induced anti-platelet antibodies, 5 which is important for confirming the etiology of DITP. 14 However, this was not feasible at our institution. Meanwhile, bone marrow suppression is often accompanied by simultaneous decreases in hemoglobin levels and platelet and leukocyte counts. Given the nature of its mechanism, bone marrow suppression often develops gradually over a few weeks. By contrast, DITP often develops rapidly after 7 to 14 days of therapy or 1 to 3 days of therapy in patients with prior exposure. 12 Therefore, because of the absence of decreases of white blood cell counts and hemoglobin levels or a rapid decline of platelet counts, we speculated that the patient had DITP associated with piperacillin–tazobactam and cefoperazone–sulbactam.
Previous reports described the use of other beta-lactam antibiotics, such as carbapenems,15–18 cefepime,3,19,20 cefoperazone, 15 and aztreonam, 16 in patients with thrombocytopenia induced by piperacillin–tazobactam. In our case, the patient did not tolerate cefoperazone–sulbactam despite successful treatment with meropenem and cefotiam. Given the immunogenic nature of DITP and the structural similarities among beta-lactams, the structural moiety of the piperacillin–tazobactam antibodies responsible for this immunogenic response must be identified. Likewise, the safety of other beta-lactam antibiotics in the clinical setting must be evaluated. To the best of our knowledge, only three studies have described drug cross-reactions in DITP caused by beta-lactams. In one study, 4 two ceftriaxone-dependent platelet-reactive antibodies failed to cross-react with five other cephalosporins. In another study, 5 three piperacillin-induced antibodies failed to cross-react with two other penam and five cephem beta-lactam drugs. In the last study, 6 5 of 14 antibodies specific for penam drugs had strong cross-reactivity with other penam drugs. Additionally, 8 of 18 antibodies specific for cephem drugs had strong cross-reactivity with other cephem drugs. However, antibodies induced by penam drugs did not have strong cross-reactivity with cephem drugs and vice versa. Nevertheless, these reports covered few types of beta-lactam antibiotics, excluding piperacillin and cefoperazone.
Based on the patient’s medications, Table 1 lists the possible drug structures of piperacillin–tazobactam and cefoperazone–sulbactam that might cross-react to cause thrombocytopenia. First, piperacillin and cefoperazone have similar beta-lactam structures. However, cefotiam also has a similar structure. Thus, the beta-lactam rings of penam and cephem drugs are less likely to induce the production of anti-platelet antibodies. Previous reports observed a strong correlation between cross-reactivity and similar or identical R1 side groups of beta-lactams. 6 We found that piperacillin and cefoperazone had similar R1 side chains, which induce the production of platelet-reactive antibodies. Therefore, we hypothesized that the identical R1 side chains of piperacillin and cefoperazone could explain the development of thrombocytopenia during piperacillin–tazobactam and cefoperazone–sulbactam treatment in our patient. However, a case report found that cefoperazone can be successfully administered to patients with DITP associated with piperacillin–tazobactam, 15 making the aforementioned hypothesis debatable. Second, we found that similarly as piperacillin, sulbactam contains a beta-lactam structure, which might be responsible for the induction of platelet-reactive antibody production. This case could be similar to a cross-reaction between sulbactam allergy and other beta-lactams with a similar core as that of sulbactam. 21 Hence, there was a possibility of cross-reactivity between piperacillin and sulbactam in the development of thrombocytopenia. Third, in a previous study, tazobactam-specific antibodies were obtained from a patient with DITP caused by piperacillin–tazobactam. 22 Thus, tazobactam might be a crucial drug in our case. Moreover, the structural similarities between tazobactam and sulbactam suggested the possibility of a cross-reaction between piperacillin–tazobactam and cefoperazone–sulbactam. However, multiple pathways could be involved in different immune reactions. Therefore, the exact antibodies responsible for piperacillin–tazobactam-induced immune thrombocytopenia cannot be clearly defined, and further investigation is required.
The possible drug structures responsible for the cross-reactivity of platelet-reactive antibodies in our patient with immune thrombocytopenia.

The proper treatment of DITP usually entails withdrawal of the suspected drug. Meanwhile, supportive care entails platelet transfusions because bleeding complications can be severe. 23 To date, no evidence suggests that corticosteroids are efficacious in DITP. 23 This might explain why our patient experienced DITP despite continuous methylprednisolone use. However, a previous case report observed a minimal decrease in platelet count upon the administration of a higher corticosteroid doses. 24
In summary, we reported the first case of piperacillin–tazobactam-induced immune thrombocytopenia that was successfully treated with meropenem and cefotiam; however, the patient was unable to tolerate cefoperazone–sulbactam as a treatment for pneumonia. Further, the exact antibodies responsible for piperacillin–tazobactam-induced immune thrombocytopenia remain unknown, and further investigation is necessary. Likewise, the chemical structure similarities among beta-lactams must be examined to determine the risk of DITP in the clinical setting.
Footnotes
Acknowledgements
We would like to thank the patient and his family for providing consent to use his data in this case report.
Author contributions
XH and WH conceived the original idea and wrote the manuscript. XW and XL performed literature search and collected the clinical data. QC and LD revised and edited the manuscript. All authors revised, read, and approved the submitted version.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
