Abstract
Objectives
We assessed the relationship between obesity and all-cause mortality in patients with acute respiratory distress syndrome (ARDS).
Methods
In this retrospective cohort study, patient data were extracted from the eICU Collaborative Research Database and the Medical Information Mart for Intensive Care Database III. Body mass index (BMI) was grouped according to World Health Organization classifications: underweight, normal weight, overweight, obese. Cox regression models estimated hazard ratios (HRs) and 95% confidence intervals (CIs) of all-cause mortality related to obesity.
Results
Participants included 185 women and 233 men, mean age 70.7 ± 44.1 years and mean BMI 28.7 ± 8.1 kg/m2. Compared with normal weight patients, obese patients tended to be younger (60.1 ± 13.7 years) and included more women (51.3% vs. 49.0%). In the unadjusted model, HRs (95% CIs) of 30-day mortality for underweight, overweight, and obesity were 1.57 (0.76, 3.27), 0.64 (0.39, 1.08), and 4.83 (2.25, 10.35), respectively, compared with those for normal weight. After adjustment, HRs (95% CIs) of 30-day mortality for underweight, overweight, and obesity were 1.82 (0.85, 3.90), 0.59 (0.29, 1.20), and 3.85 (1.73, 8.57), respectively, compared with the reference group; 90-day and 1-year all-cause mortalities showed similar trends.
Conclusions
Obesity was associated with increased all-cause mortality in patients with ARDS.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS) is an acute inflammatory lung injury associated with increased pulmonary vascular permeability, increased lung weight, and loss of aerated lung tissue, with severe hypoxemia. 1 , 2 ARDS is a global health care challenge with a reported mortality rate of more than 34%. 3 Despite advances in understanding of the mechanisms in ARDS, improvement in mortality remains less than satisfactory. 4 Hence, there is a pressing need to identify prognostic predictors of ARDS to assist physicians in making medical decisions and identifying patients with high-risk. 5
Obesity is considered to be a global epidemic resulting from a growing preference for energy-dense foods by populations worldwide. Obesity is a contributor to increased morbidity in hypertension, diabetes, coronary heart disease, cancer, and other diseases. 6 , 7 However, recent studies have revealed an “obesity paradox” in that obesity remains a risk factor for some diseases (e.g., heart failure, cancer, and stroke), but paradoxically presents a survival benefit in patients who have already developed these diseases.8–10 Several studies11–14 have investigated the relationship between obesity and outcome of ARDS. Ni et al. 15 conducted a meta-analysis pooling five trials. They found that compared with normal weight, obesity was more likely to result in lower mortality. Zhi et al. 16 also reported an obesity paradox in ARDS in a systematic review and meta-analysis. However, several important confounding factors, including alcohol use, severity of disease, and cardiovascular disease, were not considered in these studies. Thus, whether an obesity paradox exists with respect to ARDS remains controversial. 17 In our study, we evaluated the association between obesity and the risk of all-cause mortality in patients with ARDS.
Methods
Study population
We conducted a retrospective cohort study using the eICU Collaborative Research Database and the Medical Information Mart for Intensive Care Database III (MIMIC-III) version 1.4.18,19 The MIMIC-III database includes 58,976 patients admitted to the intensive care unit (ICU) at Beth Israel Deaconess Medical Center from 2001 to 2012. 18 The setting of this database has been approved by the Institutional Review Boards of Beth Israel Deaconess Medical Center and the Massachusetts Institute of Technology. To gain access to this database, we completed online courses required by the National Institutes of Health and completed all tests related to protecting human research participants. All personal information in the database has been de-identified to safeguard patient privacy. The reporting in our study conforms to the STROBE statement.
Of 58,976 distinct patients in the database, patients were included if (i) they were screened for ARDS, defined according to the American European Consensus Committee as follows: (1) hypoxemic respiratory failure requiring intubation and ratio of arterial oxygen partial pressure to fractional inspired oxygen ≤200 mmHg; (2) bilateral infiltrates on chest radiographs; (3) an absence of left atrial hypertension; 20 , 21 (ii) height and weight data were acquired on the first day of admission; (iii) the hospital stay was >2 days; and (iv) they were aged above 18 years. Exclusion criteria were >5% missing data and individual data values exceeding the mean ± 3 times the standard deviation.
Our institutional ethics review committee waived the need for approval of the study protocol and informed consent because this study involved a retrospective analysis using a public database.
Data extraction and management
We collected patient’s demographic data (age, sex, and ethnicity), clinical characteristics, comorbidities, and scale scores. Vital signs within 24 hours after ICU admission included heart rate, percutaneous oxygen saturation (SPO2), systolic blood pressure (SBP), and diastolic blood pressure (DBP). Comorbidities included hypertension, diabetes mellitus (DM), coronary heart disease (CHD), chronic heart failure (CHF), atrial fibrillation (AF), and acute kidney injury (AKI). The Simplified Acute Physiology Score II (SAPS II), Sequential Organ Failure Assessment (SOFA), 22 Glasgow Coma Scale (GCS), and Acute Physiology and Chronic Health Evaluation (APACHE) III 23 scores were included. 24
Body mass index (BMI) was calculated using height and weight recorded in the first 24 hours after patient admission. BMI was subdivided into four groups according to World Health Organization BMI classifications: underweight (BMI <18.5 kg/m2), normal weight (BMI 18.5–25 kg/m2), overweight (BMI 25–30 kg/m2), and obese (BMI ≥30 kg/m2). 25
Outcomes
Our primary outcome was 30-day mortality, and secondary outcomes were 90-day mortality and 1-year mortality. The date of enrollment was the start date of follow-up, and all participants were followed for at least 1 year. The date of death was obtained from Social Security Death Index records.
Statistical analyses
The statistical analysis was divided into three steps. First, participants were divided into groups according to baseline BMI. Continuous data are presented as mean with standard deviations and were compared using analysis of variance or the Kruskal–Wallis H test. Categorical data are presented as frequency and percentage and were compared using the χ2 test and Fisher’s exact test. Second, Cox proportional hazards regression models were used to estimate hazard ratios (HRs) with 95% confidence intervals (CIs) for the association between BMI and mortality. The group with normal weight (BMI: 18.5–25 kg/m2) was considered the reference group. In Model 1, no covariates were adjusted; model 2 was adjusted for age, sex, and ethnicity; model 3 was adjusted for confounders including age, sex, ethnicity, SBP, heart rate, SPO2, CHD, AF, DM, AKI, CHF, and APACHE III and SOFA scores. Confounders were selected based on change in the effect estimate over 10%. 26 , 27 Third, smooth curve fitting (penalized spline method) was used to identify nonlinear correlations of BMI with 30-day mortality. A two-piecewise linear regression model was applied to examine the threshold effect of BMI on 30-day mortality. The inflection point of BMI, at which the correlation of BMI with 30-day mortality began to reverse, was determined using a recursive method.
We used IBM SPSS version 22.0 (IBM Corp., Armonk, NY, USA) for the statistical analysis. A P value <0.05 (two-sided) was considered statistically significant.
Results
Participant characteristics
For this study, from a total of 58,976 of individuals, 418 participants were ultimately included in the analysis (Figure 1). The baseline characteristics of 418 patients with ARDS are listed in Table 1. The participants included 185 women and 233 men, with mean age 70.7 ± 44.1 years and mean BMI 28.7 ± 8.1 kg/m2. At baseline, obese patients tended to be younger (60.1 ± 13.7, P = 0.003) and included more women (51.3% vs. 49.0%, P = 0.001) compared with normal weight patients (Table 1). The proportion of patients with DM increased with increased BMI.

Flow chart illustrating inclusion and exclusion criteria of study cohort.
Characteristics of study participants according to BMI.

Data are presented as mean ± SD unless otherwise noted.
P value: difference among the four groups.
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; SPO2, percutaneous oxygen saturation; CHD, coronary heart disease; CHF, chronic heart failure; AF, atrial fibrillation; DM, diabetes mellitus; SOFA, Sequential Organ Failure Assessment; GCS, Glasgow Coma Scale; ICU, intensive care unit; APACHE, Acute Physiology and Chronic Health Evaluation; AKI, acute kidney injury.
Association between BMI and outcome of ARDS
Table 2 displays the effect sizes in the association between BMI and outcome of ARDS. For 30-day mortality, 85 deaths occurred during the follow-up period. In Model 1 (unadjusted model), the HRs (95% CIs) of 30-day mortality for underweight, overweight, and obesity were 1.57 (0.76, 3.27), 0.64 (0.39, 1.08), and 4.83 (2.25, 10.35), respectively, (P < 0.0001) in comparison with normal weight. In Model 2, after adjusting for age, sex, and ethnicity, a similar trend was observed for 30-day mortality and the risk was more evident with higher BMI. In Model 3, after adjusting for confounders including age, sex, ethnicity, SBP, heart rate, SPO2, CHD, AF, DM, AKI, CHF, and APACHE III and SOFA scores, the HRs (95% CIs) of 30-day mortality for underweight, overweight, and obesity were 1.82 (0.85, 3.90), 0.59 (0.29, 1.20), and 3.85 (1.73, 8.57) respectively, compared with the reference group. For 90-day mortality and 1-year mortality, a similar trend was also observed, and the risk was more evident with higher BMI.
HRs (95% CIs) for mortality across groups of BMI.

Model 1: no covariates were adjusted.
Model 2 adjusted for age, sex, and ethnicity.
Model 3 adjusted for age, sex, ethnicity, SBP, heart rate, SPO2, CHD, AF, DM, AKI, CHF, APACHE III, and SOFA.
BMI, body mass index; HR: hazard ratio; CI: confidence interval; ref: reference; SBP, systolic blood pressure; SPO2, percutaneous oxygen saturation; CHD, coronary heart disease; CHF, chronic heart failure; AF, atrial fibrillation; DM, diabetes mellitus; SOFA, Sequential Organ Failure Assessment; APACHE, Acute Physiology and Chronic Health Evaluation; AKI, acute kidney injury.
To demonstrate nonlinearity in BMI and 30-day mortality, we performed smooth curve fitting (penalized spline method). After adjusting for age, sex, ethnicity, SBP, heart rate, SPO2, CHD, AF, DM, AKI, CHF, and APACHE III and SOFA scores, nonlinear relationships were observed (Figure 2).
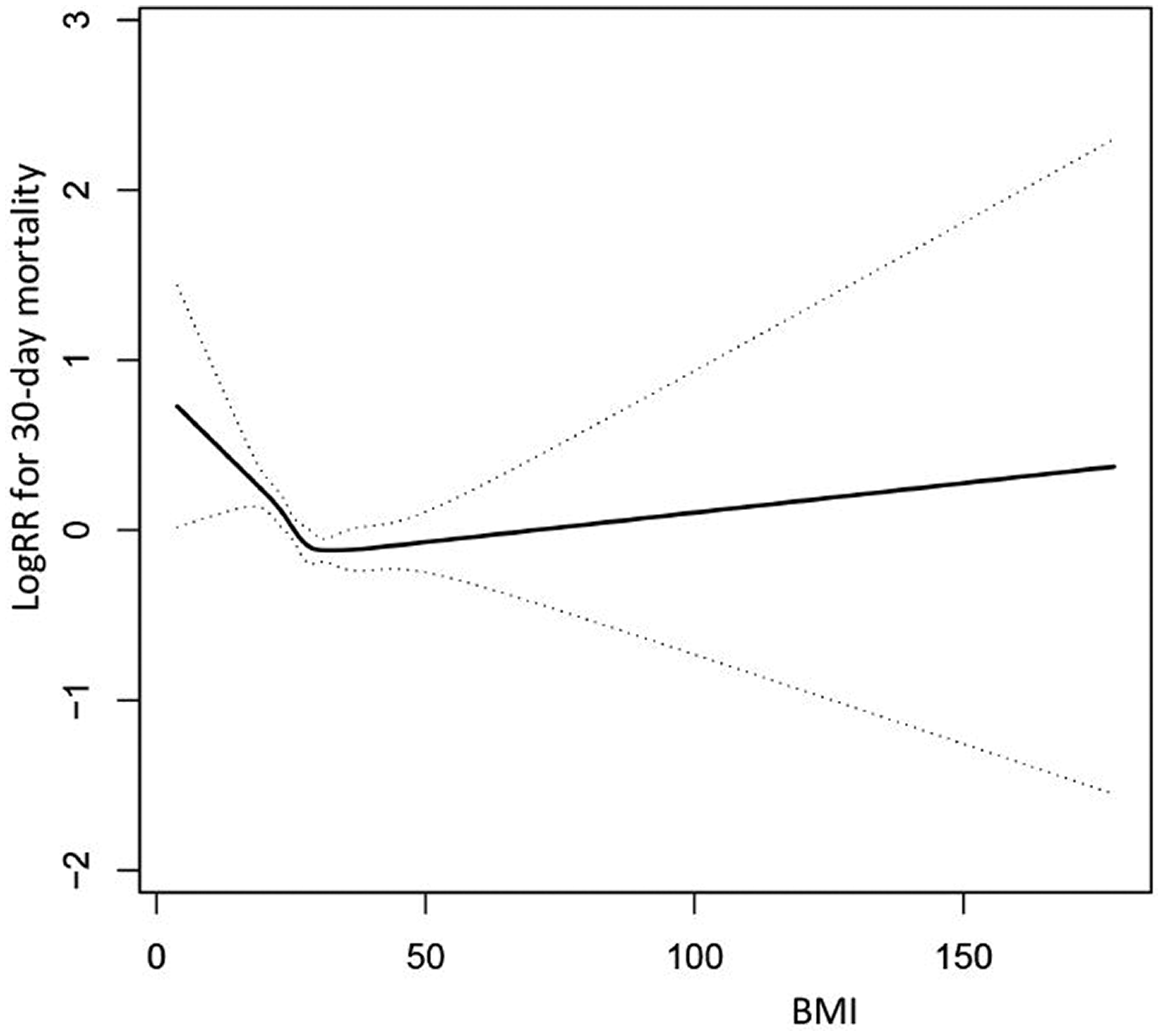
Fitting curve between BMI and log RR for 30-day mortality.
Discussion
In this observational database study, we found that obesity was associated with increased risk of short- and long-term all-cause mortality in patients with ARDS, compared with their normal weight counterparts. However, there was no significant difference in odds of mortality among patients with ARDS who were underweight, overweight, and normal weight. Moreover, the relationship between BMI and ARDS mortality was nonlinear.
The results of epidemiological studies showing the impact of obesity on outcomes in patients with ARDS remain controversial 16 , 28 , 29 In 1999, Fleischmann et al. 30 first reported that increased BMI reduced mortality in patients with end-stage renal disease. This connection was also observed in CHF, CHD, chronic obstructive pulmonary disease, and critical illness.31–34 The phenomenon was called the “obesity paradox”. In a meta-analysis, 16 it was suggested that ARDS morbidity was higher for patients with obesity than those with normal weight, but obesity was significantly associated with a reduction in ARDS mortality. 15 , 16 However, these studies did not adjust for cardiovascular disease or AKI. Obese patients are more likely to have comorbidities, cardiovascular disease, respiratory illness, and AKI. Our study used detailed clinical, laboratory, and physiologic data to strengthen our findings. We adjusted for multiple factors such as cardiovascular risk and severity of disease, and we applied curve fitting to the relationship between BMI and ARDS prognosis; a U-shaped relationship was observed. According to two-stage analysis, the cutoff point of BMI was 31.5 kg/m2. Although the lower mortality rate in the overweight BMI range was surprising, this finding had been demonstrated in the general population as well for all-cause mortality. 35
The incidence of ARDS is higher in obese individuals; however, the mortality rate of ARDS is lower in obese patients than in normal-weight patients. The underlying pathophysiologic mechanisms are still undefined; possible explanations are as follows. First, the inflammatory status of obese patients with ARDS may be attenuated or altered. ARDS is characterized by an overreaction to inflammation in the lungs. 36 Obese patients with respiratory failure are reported to have lower plasma levels of proinflammatory cytokines than normal-weight patients. 13 Moreover, Weisberg et al. 37 showed that the increased adipocyte volume in healthy obese people can lead to the accumulation of M1 subsets of macrophages (proinflammatory properties). Because macrophages are known to switch between M1/M2 phenotypes during critical illness, 38 protection against severe disease in obese patients may depend on switching to predominantly M2 (anti-inflammatory properties) activation in the already large number of macrophages. Second, lipids and lipoproteins in adipose tissue can bind potentially toxic metabolites. The additional antioxidant stores in obese patients may help them during the initial catabolic phase, thus contributing to survival. 39 Third, previous studies have suggested that the likelihood of requiring mechanical ventilation in obese patients is higher than in patients with low or normal weights. The application of mechanical ventilation may reduce the likelihood of respiratory muscle weakness, which partly explains the relative decrease in mortality among obese patients with ARDS. Another theory is that obesity leads to a protective preconditioning response that may induce a low-grade inflammatory state. In this process, the lungs may be protected from a more aggressive secondary attack, such as via ventilator-induced lung injury or sepsis. 40 Further research is needed to clarify the mechanisms underlying the relatively low mortality rate in obese patients with ARDS.
Several study limitations should be acknowledged. First, this was a retrospective cohort study; we cannot prove a causal relationship between mortality and BMI. Second, as with any cohort study, we aimed to adjust for possible risk factors, such as cardiovascular diseases and comorbidities; however, residual confounders cannot be completely ruled out, including proinflammatory factors, marital status, and other known or unknown confounders. Third, BMI values used were only those within the first 24 hours of admission; however, the relationship between subsequent changes in BMI and prognosis was not evaluated. BMI on admission may not be a good index of obesity owing to many factors. For example, positive fluid balance during resuscitation may falsely increase body weight. Using only baseline data increased the risk of misclassification bias.
Conclusions
In this retrospective single-center cohort, obesity was found to be associated with increased risk of short- and long-term all-cause mortality in patients with ARDS. After adjusting for multiple factors, the increase in risk declined but remained statistically significant. However, there was no significant difference in the odds of death among patients with ARDS who were underweight, overweight, and normal weight. The relationship between BMI and all-cause mortality in patients with ARDS was nonlinear.
