Abstract
Managing type B aortic dissection (TBAD) involving Kommerell’s diverticulum (KD), aberrant right subclavian artery (ARSA), and isolated left vertebral artery (ILVA), is extremely challenging. As treatment, we described a one-stage hybrid technique combined with thoracic endovascular aortic repair (TEVAR) with open surgery through a supraclavicular incision. A 57-year-old man with TBAD and the three anomalies successfully underwent hybrid TEVAR. A side-to-side artificial bypass between the ARSA and the right common carotid artery was established through a right supraclavicular incision before TEVAR. The release of the stent-graft was designed from the distal aortic ostium to the left common carotid artery (LCCA) to cover the ILVA, ARSA, and left subclavian artery (LSA). Then, the ILVA and LSA were transposed to the LCCA through a left supraclavicular incision. Intraoperative angiography confirmed complete false lumen exclusion and KD, with all branches patent and without endoleaks. Computed tomography angiography 7 days and 1 year postoperatively demonstrated well-perfused ARSA, LSA, and ILVA, and a fully expanded stent-graft with no endoleaks, migration, disconnection, or stenosis. TBAD involving KD, ARSA, and ILVA in one case is rare. This is the first report to treat this pathology with a one-stage supraclavicular hybrid procedure.
Keywords
Introduction
Aberrant right subclavian artery (ARSA), Kommerell’s diverticulum (KD), and isolated left vertebral artery (ILVA) are three aortic arch anomalies that seldom exist together. Moreover, people with these aortic arch anomalies are usually susceptible to aortic dissection that could benefit from surgery under certain conditions. The reported incidences of ARSA and ILVA are 0.6% and 3.4% in the general population, and 1.8% and 6.3% in patients with thoracic aortic disease, respectively. 1 KD has also been found in 60% of patients with ARSA. 2 The high morbidity and mortality rates with open surgery with total aortic arch replacement limit the scope of this approach. Conventional thoracic endovascular aortic repair (TEVAR) is also a challenging approach because the proximal landing zone may be insufficient for the endograft owing to involvement of the ostia of the left subclavian artery (LSA) and ILVA because of dissection. This report demonstrates a hybrid approach involving TEVAR, double supraclavicular transposition, and single bypass to treat type B aortic dissection (TBAD) involving the three anomalies.
Case report
A 57-year-old male patient with a long history of hypertension was admitted to our hospital with acute chest and back pain for 12 hours. Computed tomography angiography (CTA) demonstrated TBAD combined with ILVA, ARSA, and KD, and that the ILVA was dominant (Figure 1a–d). The maximum KD diameter was 32.8 mm (Figure 1c), and the entry tear was located around KD and ARSA (Figure 1e). The distances between the ostia of the LSA and ILVA and the entry tear were 3.0 mm and 13.0 mm, respectively (Figure 1f and 1g). If traditional TEVAR were performed to treat the TBAD, the ARSA, ILVA, and LSA would have to have been covered to achieve an adequate landing distance, and, to ensure cerebral perfusion, the ILVA, LSA, and ARSA would have to have been revascularized. Because the fenestrated TEVAR (fTEVAR) and chimney TEVAR (chTEVAR) techniques are complicated, expensive, and prone to complications, a hybrid TEVAR with concomitant double supraclavicular transposition of the LSA and ILVA to the LCCA as well as single artificial bypass of the ARSA to the RCCA was performed through bilateral supraclavicular incisions.

CTA findings confirming type B aortic dissection involving three rare anatomical anomalies. (a) 3D image showing type B aortic dissection involving anatomical anomalies, namely KD (red arrow), ARSA (white arrow), and ILVA (yellow arrow). (b) 3D image showing the ARSA and KD (red arrow), with a maximum KD diameter of 21.3 × 32.8 mm. (c) 3D image showing the ILVA (yellow arrow). (d) 2D image showing the ILVA directly arising from the aortic arch (yellow arrow). (e) The KD and ARSA were involved owing to dissection. (f) The distance between the entry tear and the LSA ostium (green arrow) on 2D imaging is 3.0 mm. This image shows the four supra-arch branches: RCCA (pink arrow), LCCA (blue arrow), ILVA (yellow arrow), and LSA (green arrow). (g) The distance between the entry tear and the ILVA ostium on 2D imaging is 13.0 mm
The patient underwent standard drug therapy after admission, which lasted for 1 month. Symptoms of chest and back pain persisted. The hybrid procedure was then performed under general anesthesia in a hybrid operating room. The right common femoral artery was punctured, and a gold-marked pigtail catheter (Cook Medical Inc., Bloomington, IN, USA) was inserted into the ascending aorta to perform angiography, which confirmed the CTA results (Figure 2). Two transverse incisions were made in bilateral supraclavicular fossae. The clavicular end points of the sternocleidomastoid muscle and anterior scalene muscle were transected to expose the LCCA, LSA, ILVA, RCCA, and ARSA (Figure 2). A side-to-side bypass between the ARSA and RCCA was established using an 8- to 40-mm artificial vessel (W.L. Gore & Associates, Inc., Flagstaff, AZ, USA) (Figure 2). The proximal ARSA was not transected during the bypass procedure. Next, a 0.035-inch Lunderquist guidewire (Cook Medical Inc.) was advanced into the ascending aorta via right common femoral artery access. A 38- × 32- × 180-mm thoracic aortic stent-graft (DiNovA Medtech, Hangzhou, China) was introduced into the aortic arch through the Lunderquist guidewire and then deployed to cover the entry tear as well as the ostia of the LSA, ILVA, and ARSA. A 40- × 34- × 180-mm bare stent (DiNovA Medtech) and a 34- × 34- × 120-mm bare stent (DiNovA Medtech) were sequentially deployed in the distal descending aorta. Angiography showed that the first entry tear, LSA, and ILVA resolved, and the true lumen was fully expanded. Both the proximal ILVA and LSA were transected and ligated, and the distal arteries were anastomosed to the LCCA end-to-side (Figure 2). Completion angiography demonstrated that the artificial bypass and all of the supra-arch branches were patent (Figure 2). The procedure required 6.5 hours, including 7 minutes of X-ray exposure time. The total fluoroscopy dose was 56 K mGy, and the iodine contrast dose was 100 mL.
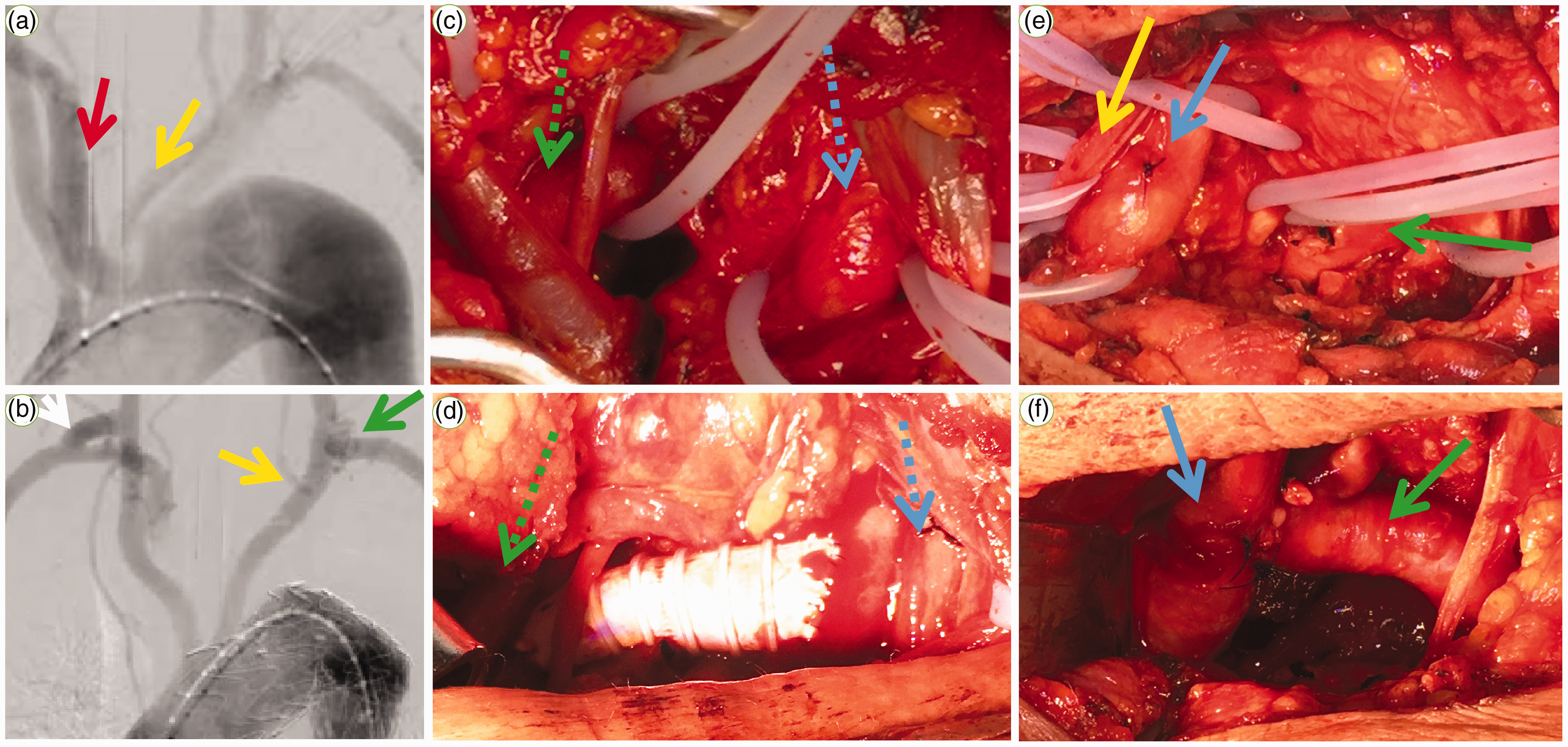
Intraoperative images. (a) Angiography before constructing the artificial bypass showing the ARSA (red arrow) and ILVA (yellow arrow) directly originating from the aortic arch. (b) Completion angiogram demonstrating that the artificial bypass (white arrow), ILVA-LCCA transposition (yellow arrow), and LSA-LCCA transposition (green arrow) were patent, with the initial part of the ARSA not developed. (c) Intraoperative photograph of the ARSA (green dotted arrow) and RCCA (blue dotted arrow). (d) The side-to-side artificial bypass established between the distal RSA (dotted green arrow) and RCCA (dotted blue arrow). (e) Intraoperative photograph of the ILVA (yellow arrow), LCCA (blue arrow), and LSA (green arrow). (f) End-to-side transposition of the LSA (green arrow) to the LCCA (blue arrow)
The patient experienced no complications of stroke, dizziness, upper limb claudication, abnormal neck movement, or poor wound healing perioperatively. He spent 1 day in the intensive care unit (ICU), with stable vital signs. CTA on postoperative day 7 showed that the stent-graft was fully expanded, with no endoleak, migration, disconnection, or stenosis, and that the false lumen was completely excluded (Figure 3a and 3g). The artificial bypass and all of the supra-arch branches were patent (Figure 3b–d). Contrast agent was not found in the proximal ARSA and KD (Figure 3e and 3f), suggesting complete exclusion of KD. The patient was discharged on postoperative day 14. Follow-up CTA 1 year postoperatively showed that the main graft was fully expanded, with all branches patent (Figure 4). Moreover, no complications occurred during the follow-up.

CTA on postoperative day 7.

CTA findings 1 year postoperatively.
Discussion
ARSA was first described by Hunauld in 1735. 3 ARSA is one of the most common congenital aortic arch anatomical anomalies reported worldwide, and it is located distal to the LSA ostium on the aortic arch and passes through the posterior mediastinum to the right upper limb. Over 60% of patients have a wider attachment between the ARSA and the aortic arch, which was described and named Kommerell’s diverticulum by Kommerell 4 in 1936. As another congenital aortic arch anatomical anomaly, the ILVA arises directly from the aortic arch and is located proximal or distal to the LSA. Affected patients with all of the common aortic arch anomalies, such as ARSA, ILVA, and a bovine arch usually have higher morbidity related to aortic dissection than does the general population. 1 The formation of ARSA and ILVA is associated with involution of the right dorsal aorta and abnormal development of the seventh intersegmental artery during fetal development.5,6
Cases combining all three anomalies (ARSA, ILVA, and KD) with aortic dissection or aneurysm have been reported rarely. We reviewed previous cases with one or more anomalies (Table 1).2,7–20 Nonami et al. 7 reported that a patient with ARSA and ILVA with aortic aneurysm successfully underwent aortic arch replacement with five branches, in 1998. Before the development of TEVAR, traditional treatment was accompanied by a high risk of complications and injuries. Compared with open surgery, endovascular therapy has the advantages of lower mortality and morbidity, and operation duration and length of hospital stay.21,22 Elghoneimy et al. 8 reported a patient with all three anomalies with TBAD who successfully underwent traditional TEVAR without reconstruction of any aortic arch branches because the entry point was 2 cm distal to the ARSA, and the landing distance was adequate. The patient recovered without morbidity during the postoperative follow-up. However, traditional TEVAR is unsuitable for all patients because of landing zone limitations. Other surgeons have reported aortic dissection or aneurysm with ARSA or ILVA occurring alone, treated with total endovascular repair,9–12 hybrid procedure,12–17 or open surgery.18–20
Recent cases of aortic dissection or aneurysm with ARSA, ILVA, or KD.

ARSA, aberrant right subclavian artery; ILVA, isolated left vertebral artery; KD, Kommerell’s diverticulum; M, male; F, female; TEVAR, thoracic endovascular aortic repair.
In our patient, TEVAR was beneficial in slowing the progression of TBAD and reducing continuous pain. Performing TEVAR 1 month after admission may improve clinical outcomes owing to satisfactory vascular conditions in the subacute phase. 23 The distances between the ostia of the ILVA and LSA, and the entry tear were 13.0 mm and 3.0 mm, respectively. As the landing distance was inadequate, it was necessary to cover the ILVA and LSA to provide a sufficient landing zone. To prevent ischemia of the left upper limb, LSA revascularization was unavoidable. CTA showed that both vertebral arteries were dominant, ensuring cerebral perfusion and suggesting that the ARSA and ILVA should also be revascularized. 9 Although fTEVAR or chTEVAR techniques could extend the landing distance, these procedures are complicated, expensive, and prone to complications when revascularizing two or more branches. Furthermore, the chimney and periscope techniques could lead to higher incidences of endoleak and retrograde dissection.2,9,24,25 Branched endografts or physician-modified fenestrated endografts were considered difficult to design and deploy accurately in this case owing to the complicated anatomical structure with the three rare anomalies.9,10,26 Open surgery with total aortic arch replacement was not considered because of the high risk of complications and injury. Although all of the above methods were achievable in our center, none was considered the best solution. Thus, a hybrid approach constituting TEVAR, double supraclavicular transposition, and single bypass was designed.
Some cases of TBAD with ARSA or ILVA have been treated successfully with a supraclavicular hybrid operation.12–17 Supraclavicular hybrid TEVAR has the advantages of less invasiveness, and better efficacy and cost compared with open surgery with total arch replacement and total endovascular treatment, such as with multiple fTEVAR or chTEVAR. Thus, we designed a hybrid TEVAR with concomitant double transposition of the LSA and ILVA to the LCCA and single artificial bypass of the ARSA to the RCCA through bilateral supraclavicular incisions. The reason for using artificial bypass to revascularize the ARSA was that the dissection had formed in the proximal ARSA. We deployed a bare metal stent distally because it was favorable in the short-term.27,28 This is the first report describing supraclavicular hybrid TEVAR for a patient with TBAD combined with three rare anomalies.
Three anomalies combined with TBAD is rare in a single patient. Supraclavicular hybrid TEVAR can decrease the risk of open surgery with total arch replacement as well as total endovascular treatment, such as multiple fTEVAR or chTEVAR. The hybrid approach is an effective, safe, and economical treatment for TBAD involving aortic arch anomalies.
Ethics statement
This study was granted written approval from the Ethics Committee from Tianjin Medical University General Hospital (approval number: No. IRB2021-WZ-021). Because the patient had already been discharged and lived far from the hospital at the time of writing of this report, verbal informed consent by telephone was obtained from the patient for publication of this case report and accompanying images.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211020241 - Supplemental material for One-stage supraclavicular hybrid procedure for type B aortic dissection involving three rare anatomical anomalies: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605211020241 for One-stage supraclavicular hybrid procedure for type B aortic dissection involving three rare anatomical anomalies: a case report and literature review by Junhang Chen, Xiangchen Dai, Jiechang Zhu, Fanguo Hu, Peng Li, Yudong Luo, Hailun Fan, Zhou Feng and Yiwei Zhang in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Authors’ contributions
All data generated or analyzed during this study are included in this article. Junhang Chen collected the case data and made substantial contributions to the writing of the manuscript. Yudong Luo, Hailun Fan, Zhou Feng, and Yiwei Zhang contributed substantially to the analysis of the case. Xiangchen Dai, Jiechang Zhu, Fanguo Hu, and Peng Li performed the operation and assisted in preparing the manuscript. All authors read and approved the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
