Abstract
Objective
To explore the role of miR-223 and miR-126 in predicting treatment responses to dual antiplatelet therapy (DAPT) in patients with ST-elevation myocardial infarction (STEMI).
Methods
Plasma miR-223 and miR-126 levels were measured before treatment. Treatment responses and 2-year survival were determined. In vitro experiments were performed to explore the mechanism of action.
Results
Patients with resistance to DAPT had a lower level of miR-223 and miR-126. Cardiac-event-free survival was shorter in patients with lower miR-223 or miR-126 levels. MiR-223 and miR-126 independently predicted DAPT resistance. Modulating miR-223 or miR-126 in platelets in vitro significantly changed the response to clopidogrel by regulating platelet aggregation.
Conclusion
MiR-223 and miR-126 play a role in DAPT resistance and may provide potential biomarkers in patients with STEMI.
Keywords
Introduction
Clot formation induced by platelet activation, adhesion, and aggregation is an important event in the pathogenesis of acute coronary syndromes (ACSs) and their complications. Dual antiplatelet therapy (DAPT) with aspirin and clopidogrel has been demonstrated to be effective for the treatment of ACS, prevention of myocardial infarction, and reduction in ACS-related deaths. 1 However, patients with ACS who receive DAPT show different response rates,2,3 and the underlying mechanisms remain unclear. Therefore, it is important to identify predictors for the efficacy of DAPT and understand the underlying mechanisms.
MicroRNA (miR) is a small non-coding RNA that modulates the expression of several mRNAs by regulating their translation and degradation. Platelets are an important source of circulating miRs, among which miR-223 has gained particular attention as it is found predominantly in platelets. 4 An important achievement in platelet research is the discovery that activated platelets participate in cell communication by releasing microparticles containing complexes of miR-223 and Argonaute 2, which are internalized by other platelets.5,6
Previous results regarding the effects of miR-223 on responses to DAPT (aspirin combined with clopidogrel or another P2Y12 antagonist) are controversial. It has been reported that miR-223 interacts with the 3ʹ-untranslated region of the mRNA encoding the P2Y12 adenosine 5ʹ-diphosphate (ADP) receptor to inhibit P2Y12 expression. 6 Decreased levels of miR-223 in platelets and plasma have been observed in patients with troponin-negative non-ST-elevation ACS treated with DAPT who have concurrent high on-treatment platelet reactivity,7,8 indicating a poorer outcome. 9 In contrast, it has been shown that reduced levels of miR-223 are associated with enhanced platelet inhibition during DAPT. 10 Therefore, further study is required to explore the role of miR-223 in predicting DAPT outcomes.
The levels of other miRs, such as miR-126, have been reported to be altered in patients with ACS during DAPT. For instance, circulating levels of miR-126 were decreased after DAPT therapy. 10 However, increased levels of miR-126 before therapy served as an indicator of adverse events after percutaneous coronary intervention in patients receiving DAPT. 11 Whether the plasma levels of miR-126 before treatment can be used to predict responses to DAPT in patients with ACS has not been explored.
In the present study, we measured the plasma levels of miR-223 and miR-126 in patients with ST-elevation myocardial infarction (STEMI) and with or without DAPT resistance to confirm whether circulating miR-223 and miR-126 can predict treatment resistance and patient survival. More importantly, we performed in vitro studies by modulating the level of miR-223 or miR-126 in platelets collected from patients to observe the response to DAPT. Our results provide novel insight into predicting the prognosis of patients with STEMI after DAPT.
Methods
Patients
In this prospective study, consecutive adult patients with STEMI were recruited between January and September 2017 after excluding those who did not meet the inclusion criteria. The exclusion criteria were as follows: (1) those who were allergic or intolerable to clopidogrel or aspirin; (2) those who experienced cerebral vascular events in the past 6 months; (3) those who had bleeding disorders; (4) those who had severe liver diseases, including liver fibrosis or cirrhosis; (5) those who suffered terminal diseases, such as advanced tumors or uremia; (6) those who were on platelet glycoprotein IIb/IIIa inhibitors; (7) those who had hematological disorders; (7) those whose platelet count was lower than 100 × 109/L or greater than 450 × 109/L; and (8) those who refused to attend this study. Patients were diagnosed with STEMI according to the 2016 American College of Cardiology/American Heart Association guideline. 12
In addition to a dose of oral aspirin (100 mg daily), all patients received a loading dose of oral clopidogrel (300 mg) and a maintenance dose of oral clopidogrel (75 mg) daily after admission. All recruited patients were followed up for at least 2 years after the initial administration. A composite endpoint of heart disease-related death, recurrent ACSs, and heart failure was documented. Severe side effects were also recorded. The major side effect refers to major bleeding events, which are defined as intracranial bleeding, a decrease in hemoglobin concentration by >5 g/dL, or a decrease in hematocrit by at least 10%. No healthy volunteers from the general public were recruited.
Determination of responses to clopidogrel
Responses to clopidogrel were determined by the VerifyNow method (Accumetrics, Inc., San Diego, CA, USA) in accordance with the manufacturer’s instruction and previous publications. 13 Briefly, VerifyNow is designed to determine the blockage of P2Y12 in platelets after treatment with ADP. Clopidogrel resistance was defined as P2Y12 reaction units (PRU)>208 based on our preliminary results and publications.14–16
Whole blood samples were collected from each individual at 6 hours after the loading dose and 7 days after clopidogrel treatment. In accordance with the manufacturer’s instructions, 2 mL of whole blood was loaded into the system, and PRU rates were recorded.
miR quantification
miRs were extracted from blood samples before and after treatment. Plasma samples were obtained by centrifugation at 4000×g for 30 minutes at 4°C. A total of 400 μl of plasma was used for miRNA extraction with a commercial miRNA isolation kit (Life Technologies, Carlsbad, CA, USA). Reverse transcription targeting miRs was performed using a TaqMan MicroRNA Reverse Transcription Kit (Life Technologies). Quantitative polymerase chain reaction (qPCR) was conducted using TaqMan primers and probes (Life Technologies) following the manufacturer’s guides. All samples were run in triplicate and quantified using the comparative threshold cycle method (2−ΔΔCt) method with miR-16 as an endogenous control.
Evaluation of the role of miR in vitro
To perform gain-of-function or loss-of-function assays, we transfected the collected platelets with a miRNA overexpression plasmid or siRNA targeting miRNA in vitro in accordance with a published protocol. 17 Platelets were purified as previously published. 18 Vectors for miR-223 or miR-126 and non-targeting control siRNA were purchased from Sigma-Aldrich (St. Louis, MO, USA) and transfected into platelets using a commercial transfection kit (Lipofectamine 2000, Invitrogen, Carlsbad, CA, USA) in accordance with the manufacturer’s instruction. The expression levels of miR-223 and miR-126 were validated by qPCR as described above. Following transfection, platelets were diluted to the circulating concentration in humans and collected for the VerifyNow assay.
Light transmission aggregometry
Platelets were collected and transfected with miR mimics or inhibitors as described above. After transfection, platelets (200 × 109/L) were used for aggregation assays. Platelet aggregation was induced by adding ADP (purchased from Sigma-Aldrich) at a concentration of 2 μM in the presence or absence of clopidogrel (30 μM) in the medium, as previously reported.19,20 The process was performed on a Chronolog Optical Lumi-Aggregometer (Havertown, PA, USA) at 37°C and 240×g. Data were collected at 15 minutes after ADP stimulation.
Statistical analysis
Data were presented as the mean±standard deviation, median (range), or n (%), according to the type of variables. Student t-tests were used to compare continuous data when two groups were compared. One-way ANOVA with the Bonferroni post-hoc test was used to compare data among more than two groups. Chi-squared tests were used to analyze categorical variables. Event-free survivals were assessed with the Kaplan–Meier method. Area under the curve (AUC) analysis was also performed to determine the ability of miR-223 and miR-126 to predict the resistance to DAPT in patients with STEMI. The correlation between variables and treatment responses to DAPT was analyzed by logistic regression. The effects of variables on event-free survival were analyzed by Cox regression. Normal distributions were confirmed by Shapiro–Wilk tests. Homogeneity of variance was analyzed by Hartley tests. All data were analyzed using GraphPad Prism 5 (Windows Edition, GraphPad Software, San Diego, CA, USA) and SPSS Statistics for Windows, Version 17.0 (SPSS Inc., Chicago, IL, USA). A p value of less than 0.05 was considered statistically significant.
Ethical consideration
The Ethics Committee of Ningbo First Hospital at Ningbo University reviewed, approved, and supervised this study (approval number: 20150014). All patients involved in this study agreed to their participation and signed written consent forms.
Results
Patient characteristics
The selection of patients involved in this study is presented in Figure 1. A total of 120 consecutive patients with STEMI were included in the present study. Thirty-six patients had a PRU larger than 208 U and were considered resistant to DAPT (referred to as non-responders), and the remaining patients were responders. All recruited patients received lose-dose daily aspirin (75–100 mg oral daily) prior to hospital admission. The characteristics of responders and non-responders are compared in Table 1. Non-responders were older and more frequently used proton-pump inhibitors than responders. No severe side effects were observed in either group.
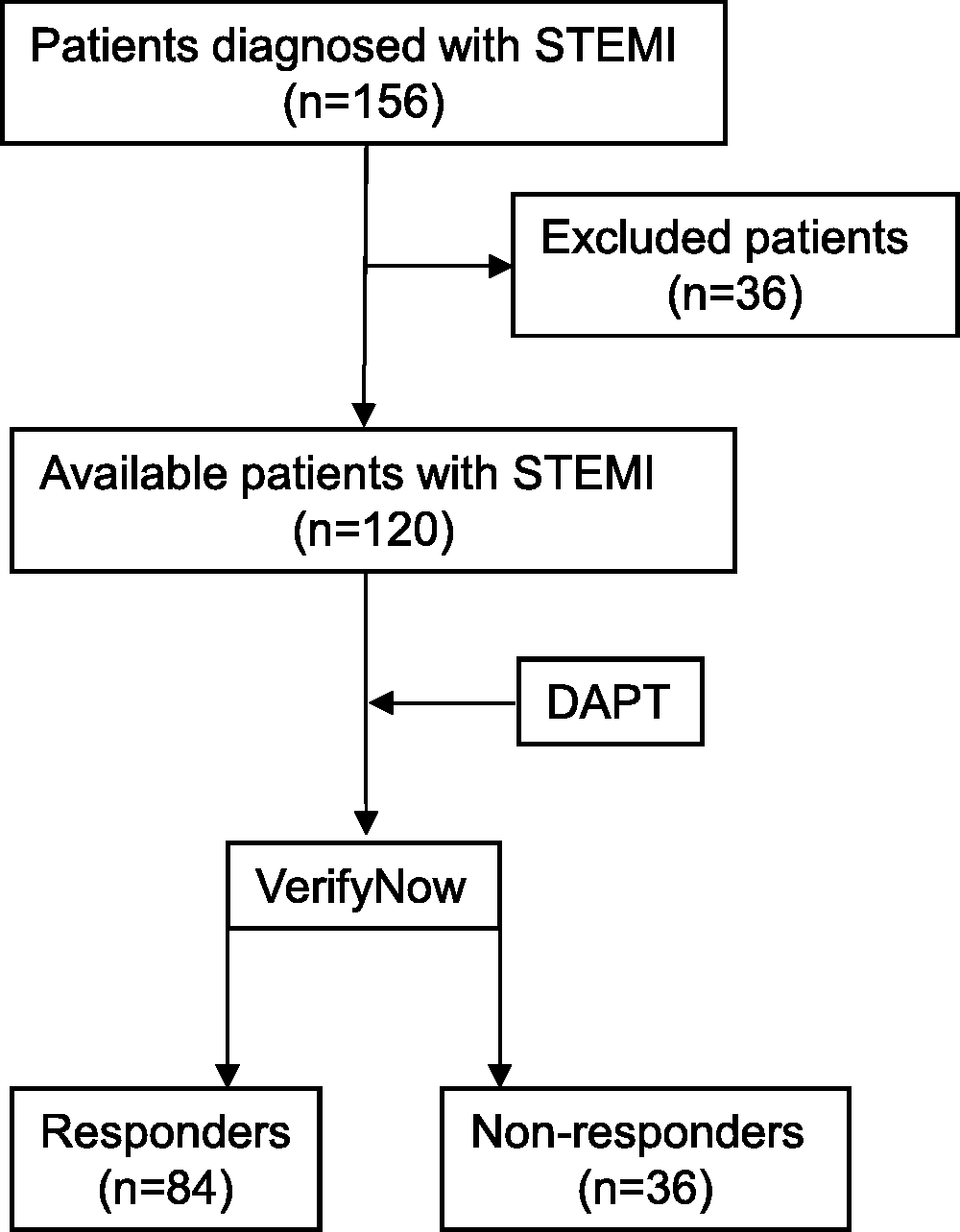
Patient selection in this study. A total of 120 consecutive patients with STEMI were included in the present study. Responses to clopidogrel were determined by the VerifyNow method. Thirty-six patients had a PRU larger than 208 U and were considered resistant to DAPT (referred to as non-responders), and the remaining patients were responders.
Patient characteristics.

BMI, body mass index; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; PCI, percutaneous coronary intervention; LVEF, left ventricular ejection fraction; PPI, proton-pump inhibitor.
Association of plasma miR-223/miR-126 levels with resistance to DAPT
As shown in Figure 2a, non-responders had a significantly lower plasma level of miR-223 compared with that of responders before the initiation of DAPT (responders vs. non-responders, 1.117±0.02 vs. 0.342±0.045, p < 0.0001). Similarly, a lower level of miR-126 before treatment was observed in non-responders (responders vs. non-responders, 1.34±0.025 vs. 0.8±0.1265, p < 0.0001) (Figure 2b). These data indicate the non-responders to DAPT have lower levels of miR-223 and miR-126 in the plasma before treatment.

Association of plasma miR-223/miR-126 levels with resistance to DAPT. a. Serum levels of miR-223 in responders and non-responders before DAPT. b. Serum levels of miR-126 in responders and non-responders before DAPT. c. Correlation between PRU and relative miR-223 levels in the serum. d. Correlation between PRU and relative miR-126 levels in the serum. ****p < 0.0001, by two-tailed Student t-tests.
Because the PRU value is an important indicator of DAPT resistance, we then determined whether there was a correlation between the PRU and plasma levels of miR-223 or miR-126. We performed linear regression analyses between these variables. As indicated in Figure 2c and d, there were significant correlations between the PRU and levels of miR-223 or miR-126 (p < 0.0001), further confirming that miR-223 and miR-126 are associated with DAPT responses.
Predictive values of plasma miR-223 or miR-126 for DAPT resistance
AUC analysis was then performed using miR-223 and miR-126 to predict the resistance to DAPT in patients with STEMI. As shown in Table 2, both plasma miR-223 and miR-126 had significant value in predicting DAPT resistance. The AUC of miR-223 for predicting resistance to DAPT was 0.996, which was superior to miR-126 (0.905). The cut-off value of plasma miR-223 levels was 0.70 with a sensitivity of 88.9% and a specificity of 97.6% (p < 0.0001), and the cut-off value of plasma miR-126 levels was 1.02 with a sensitivity of 88.9% and a specificity of 95.2% (p < 0.0001).
Predictive values of serum miR-223 and miR-126 in DAPT resistance.
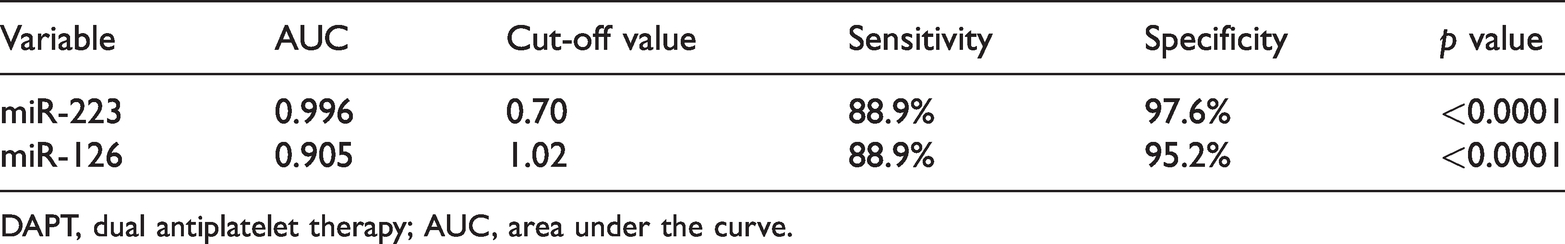
DAPT, dual antiplatelet therapy; AUC, area under the curve.
Risk factors for DAPT resistance
We subsequently aimed to identify risk factors for DAPT resistance using the logistic regression method. As revealed in Table 3, univariable logistic regression analyses showed that older patients (>64 years old), the use of proton-pump inhibitors, low plasma miR-223 levels (≤0.70), and low plasma miR-126 levels (≤1.02) were associated with DAPT resistance. Moreover, we input these parameters into a multivariable logistic regression analysis, which demonstrated that the use of proton-pump inhibitors, low plasma miR-223, and low plasma miR-126 were independent risk factors for DAPT resistance.
Logistic regression analyses of risk factors for DAPT resistance.

*p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
DAPT, dual antiplatelet therapy; OR, odds ratio; CI, confidence interval; BMI, body mass index; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; PCI, percutaneous coronary intervention; LVEF, left ventricular ejection fraction; PPI, proton-pump inhibitor.
Patients with low levels of miR-223 or miR-126 had shorter cardiac-event-free survival
In addition to DAPT responses, we followed up all recruited patients to analyze heart disease-related events within 2 years. We divided patients into two groups according to the cut-off values of plasma miR-223 or miR-126 (Table 2). As shown in Figure 3a and b, patients with lower levels of miR-223 (≤0.70) or miR-126 (≤1.02) had significantly shorter cardiac-event-free survival than those with higher levels of miR-223 or miR-126 (p < 0.0001).

Patients with low levels of miR-223 or miR-126 have shorter cardiac-event-free survival. All recruited patients were followed up since the initial administration for up to 24 months. A composite endpoint of heart disease-related death, recurrent ACS, and heart failure was evaluated. a. Patients with low miR-223 levels in the serum had shorter cardiac-event-free survival. b. Patients with low miR-126 levels in the serum had shorter cardiac-event-free survival.
miR-223 and miR-126 mediate DAPT responses in vitro
To explore the role of miR-223 and miR-126 in mediating the response to DAPT, we manipulated the levels of these two miRs in vitro and evaluated the PRU. According to the cut-off values obtained in Table 2, we classified these patients into two groups based on miR-223 values (≥0.70 or ≤0.70) and miR-126 values (≥1.02 or ≤1.02). Accordingly, miR overexpression vectors were added to the platelets obtained from patients with low levels of the respective miR, whereas miR siRNAs were used to transfect platelets obtained from those with high levels of the corresponding miR. As shown in Figure 4a, miR-223 plasmid transfection into platelets from the decreased miR-223 group potently reduced the average PRU from over 208 U to lower than 208 U (Figure 4a). Meanwhile, miR-223 siRNA transfection into platelets from the elevated miR-223 group increased the PRU (Figure 4b). Similar results were also observed in platelets transfected with miR-126 plasmids or siRNAs (Figure 4c and d). These data suggest that the platelet levels of miR-223 or miR-126 potently regulate the response of patients with STEMI to DAPT.

Modulating miR-223 or miR-126 in vitro changed the PRU. a. miR-223 oe plasmid transfection of platelets collected from patients in the decreased miR-223 group reduced the PRU. b. miR-223 siRNA transfection of platelets collected from those in the elevated miR-223 group increased the PRU. c. miR-126 oe plasmid transfection of platelets collected from those in the decreased miR-126 group reduced the PRU. d. miR-126 miRNA transfection of platelets collected from those in the elevated miR-126 group increased the PRU. **p < 0.01; ****p < 0.0001, by two-tailed Student t-tests. Each dot represents the mean of triplicate samples for each individual.
miR-223 and miR-126 regulate platelet aggregation in response to clopidogrel
Finally, we explored whether the contribution of miR-223 or miR-126 to DAPT responses was mediated by platelet aggregation in vitro. ADP (2 μM) was added to the medium 15 minutes prior to miR plasmid or siRNA transfections to stimulate platelet aggregation in the presence or absence of clopidogrel (30 μM). Using light transmission aggregometry, we determined that transfection with overexpression plasmids or siRNAs for either miR-223 or miR-126 had no effect on platelet aggregation in the absence of clopidogrel (Figure 5a–d). However, when we increased miR-223 levels in platelets collected from patients in the decreased miR-223 group, platelets treated with miR-223 mimics showed a substantial decrease in aggregation following clopidogrel treatment (Figure 5a), indicating an improved response to clopidogrel. In contrast, when miR-223 levels were decreased in the elevated miR-223 group, platelets showed enhanced aggregation after clopidogrel treatment (Figure 5b). Similar results were observed when the platelet levels of miR-126 were manipulated in vitro (Figure 5c and d). Taken together, these data indicate that miR-223 and miR-126 play important roles in clopidogrel responses by regulating platelet aggregation.
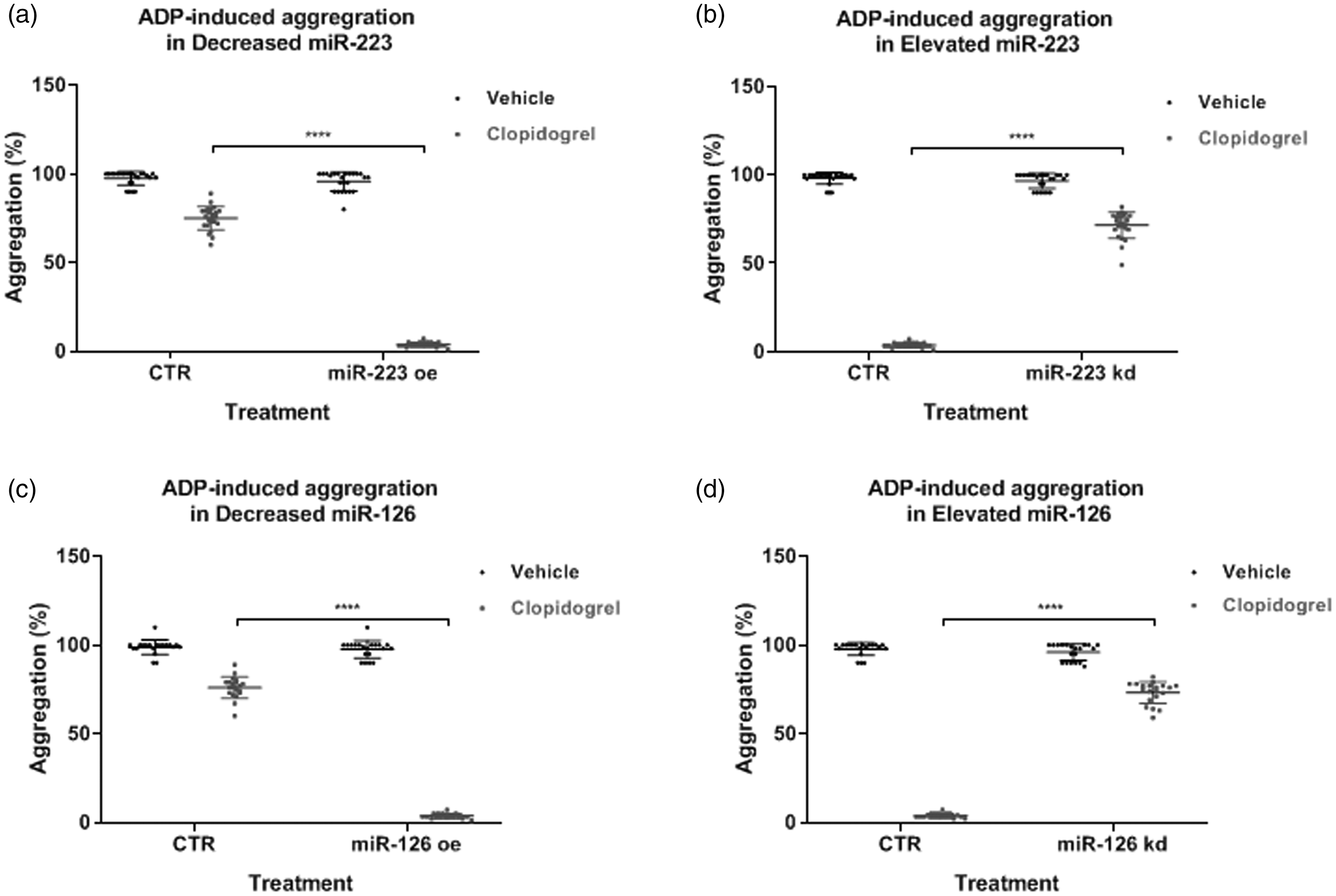
Modulating miR-223 or miR-126 in vitro regulated platelet aggregation in the presence of clopidogrel. a. Transfecting miR-223 plasmids into platelets collected from those in the decreased miR-223 group reduced platelet aggregation in the presence of clopidogrel. b. Knockdown of miR-223 in platelets collected from those in the elevated miR-223 group increased platelet aggregation in the presence of clopidogrel. c. Transfecting miR-126 plasmids into platelets collected from those in the decreased miR-126 group reduced platelet aggregation in the presence of clopidogrel. d. Knockdown of miR-126 in platelets collected from those in the elevated miR-126 group increased platelet aggregation in the presence of clopidogrel. ****p < 0.001, by one-way ANOVA with Bonferroni post-hoc tests.
Discussion
In the present study, we presented the following results: (1) patients with STEMI exhibited a response rate of 70% to DAPT; (2) patients with lower levels of plasma miR-223 or miR-126 at the start of DAPT showed treatment resistance; (3) lower plasma concentrations of miR-223 or miR-126 and the use of proton-pump inhibitors are risk factors for DAPT resistance; (4) patients with STEMI and low levels of plasma miR-223 or miR-126 have shorter cardiac-event-free survival; and (5) more importantly, reducing the levels of miR-223 or miR-126 in platelets collected from patients with high concentrations in the circulation impaired the responsiveness of platelets to clopidogrel.
Only a certain number of patients show a good response to clopidogrel measured by the inhibition of platelet aggregation.21–25 More importantly, those who have a poor response to clopidogrel are at high risk for ischemic events and other cardiac events.21–25 With respect to these previous reports, we found that approximately 70% of patients in our cohort showed a good response to DAPT, which was determined using the VerifyNow system. Our data, in addition to previous publications, highlight the importance of predicting responses to DAPT before treatment.
Platelets and platelet microparticles (PMPs) are a source of miRs, including miR-126, miR-197, miR-223, miR-24, and miR-21, which can be detected in the circulation. 10 More importantly, the concentrations of individual miRs in the circulation are correlated with their levels in platelets, 10 as these miRs are exclusively expressed in platelets. Platelet-derived miRs may have several important physiological effects. For instance, after antiplatelet therapy, the concentrations of certain miRs are reduced in the circulation,10,26 which likely results from reduced PMP shedding during platelet inhibition. However, further investigations, including pre-clinical and clinical trials, are warranted to understand the roles of circulating miRs expressed in platelets.
Antiplatelet therapy, including DAPT, is critical for the treatment of ischemic cardiac diseases. The two medications used in this regimen have different but synergetic inhibitory effects on platelets. Aspirin inhibits thromboxanes produced by platelets, whereas P2Y12 inhibitors (such as clopidogrel) suppress ADP-induced platelet aggregation. In the present study, we found that patients with high plasma miR-223 or miR-126 levels showed a good response to DAPT and longer cardiac-event-free survival, consistent with previous reports. 27 It has been reported that miR-223 binds to the 3ʹ untranslated region of P2Y12, which results in the suppression of protein expression.27,28 However, the precise roles of these platelet-derived miRs in DAPT responses warrant further experiments.
In this report, we found that decreased levels of miR-223 or miR-126 predicted undesired outcomes in patients with STEMI, although the results from a previous study show that increased levels of miR-223 or miR-126 are correlated with ischemic events within 30 days or 1 year. 29 It is worth noting that there may be differences in the design of these two studies. For instance, the ethnicity of the patients in the study mentioned above is unknown but is presumably Caucasian or Eastern European, whereas our cohort was a Han Chinese group. Differences in the genetics between these two cohorts might explain the varied results. Furthermore, a fraction of patients in the Prague-18 study received two medications (ticagrelor and clopidogrel or prasugrel and clopidogrel). 29 Another major difference between their study and ours is the definition of endpoints. Hromadka et al. 29 used a combined endpoint of ischemic events. However, using an individual endpoint rather than a combined one may change the results. Regardless of these differences, an international collaboration involving more ethnic groups at multiple institutes is warranted to explore the value of miR-223 or miR-126 in predicting adverse events in patients with STEMI.
This study has a few limitations. First, the cohort is relatively small; thus, a larger research population is needed in the future. Second, we only used the VerifyNow system to measure platelet functions. Although other measurement methods exist in practice, the above system is the most widely used and well tested in clinical trials with stable results. 13 Lastly, the assay we used to determine the levels of plasma miRs is time-consuming, which might delay the therapeutic decision. Rapid test kits for miR levels in plasma are needed in clinical practice.
This study has clinical significance. Because ischemic cardiac events, such as STEMI, require urgent therapeutic intervention, identifying non-responders in advance is crucial to select more effective and individual therapeutic options. Another issue is to identify these non-responders to avoid unnecessary side effects. Here, we showed that high plasma levels of miR-223 or miR-126 were correlated with a good response to DAPT, indicating their potential as promising predictors of treatment outcomes. Mechanistically, we determined that decreasing the levels of miR-223 or miR-126 in platelets collected from patients with high circulating miR concentrations induced therapy resistance. In contrast, increasing the levels of these miRs in platelets collected from patients with low circulating concentrations improved the responses of platelets to clopidogrel. These data were consistent with the observations in patients with STEMI. More importantly, these data indicate miR-223 or miR-126 as a direct regulator of platelet responses to clopidogrel, highlighting the potential of modulating their levels in clinical practice.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Natural Science Foundation of Ningbo (2017A610198).
Author Contributions
XL and QY were responsible for the conceptualization and performance of experiments. HC, JY, NW, YL, YZ, YZ, and JS were responsible for sample collection and patient information organization. XC and YX were responsible for the conceptualization and supervision of the study.
