Abstract
Objective
Whether direct stenting (DS) without predilatation during primary percutaneous coronary intervention (PPCI) reduces microvascular dysfunction in patients with ST-elevation myocardial infarction is unclear. We performed a randomized study to assess the effect of DS on microvascular reperfusion.
Methods
Seventy-two patients undergoing PPCI were randomly assigned to the DS or conventional stenting (CS) with predilatation groups. The primary endpoint was the post-PPCI index of microcirculatory resistance (IMR). We compared thrombolysis in myocardial infarction myocardial perfusion (TMP) grades, ST-segment resolution, and long-term clinical outcomes between the groups.
Results
Microvascular reperfusion parameters immediately after PPCI (e.g., the IMR, TMP grade, and ST-segment resolution) were not different between the groups. However, significantly fewer patients in the DS group had the IMR measured because of no-reflow or cardiogenic shock during PPCI than those in the CS group. No differences were found in left ventricular functional recovery or clinical outcomes between the groups.
Conclusions
This trial showed no effect of DS on the IMR. However, our finding should be interpreted with caution because the number of patients who could not have the IMR measured was higher in the CS group than in the DS group. A larger randomized trial is required (Research Registry number: 8079).
Keywords
Introduction
Timely reperfusion by primary percutaneous coronary intervention (PCI) salvages the myocardium and reduces mortality in patients with ST-elevation myocardial infarction (STEMI). However, successful restoration of epicardial coronary artery patency does not always lead to adequate reperfusion at the microvascular level. Microvascular obstruction following PCI is highly prevalent and independently associated with adverse clinical outcomes.1,2 This condition is related to embolization of plaques or thrombotic material downstream in the infarct-related artery,1,3 and distal embolization can occur spontaneously or by means of mechanical fragmentation during predilatation and stenting.4,5 Direct stenting without predilatation during primary PCI is thought to reduce distal embolization and thereby microvascular obstruction. This strategy has been widely investigated, and some previous studies have shown its beneficial effects on microvascular protection.6,7
It should be noted that there were potential selection biases and confounding factors in these retrospective analyses, and there is a lack of existing randomized, controlled trials. Therefore, we performed the Efficacy of Direct Stenting without Predilatation during Percutaneous Coronary Intervention in Acute Myocardial Infarction: a randomized trial evaluating microvascular dysfunction with the index of microcirculatory resistance (DIPPAMI-IMR). This trial aimed to investigate the efficacy of direct stenting without predilatation in reducing microvascular dysfunction and improving clinical outcomes compared with conventional PCI in patients with STEMI using the index of microcirculatory resistance (IMR). 8
Materials and methods
Selection of patients
Between March 2011 and December 2012, hemodynamically stable patients who had a first attack of STEMI and fulfilled the following criteria were screened and prospectively included in this study. The inclusion criteria were as follows: 1) chest pain lasting
Study flow
The study flowchart is shown in Figure 1. Patients with STEMI who met the criteria and underwent primary PCI were finally enrolled in this study. Prior to primary PCI, all patients received 300 mg of aspirin, 600 mg of clopidogrel, and weight-adjusted heparin (70 units/kg). The patients were randomly assigned to the direct stenting group (DS group) or the conventional stenting group (CS group) at the catheterization laboratory using a computerized system after achievement of TIMI 2 or 3 grade flow by TA or guide wire passage during primary PCI. TA using the Thrombuster II (Kaneka Inc., Osaka, Japan) was performed on infarct-related arteries with a TIMI flow grade of 0 or 1, thrombotic occlusion, or evidence of visible thrombi on coronary angiography. The administration of a glycoprotein IIb/IIIa inhibitor, Clotinab (Abciximab; Isu Co., Ltd., Seongnam, Korea), was recommended when the TIMI flow was less than grade 1 before TA or as a bail-out modality after unsuccessful PCI. When the antegrade flow was established, intracoronary nitrate was administered to ensure maximal epicardial vasodilation. Subsequently, stents were deployed in patients in the DS group.

Selection of patients and randomization.
In patients in the CS group, balloon angioplasty was carried out before the stenting step. In the DS group, cross-over to balloon predilatation and stenting were permitted when the stent could not be advanced through the target lesion according to the operator’s judgement. All patients were treated with second-generation drug-eluting stent (DES) implantation. A DES was selected by operators at the time of procedure and included zotarolimus-eluting stents (Endeavor or Integrity Medtronic, Minneapolis, MN, USA), everolimus-eluting stents (Xience; Abbott Vascular, Santa Clara, CA, USA or Promus; Boston Scientific, Marlborough, MA, USA), and Biolimus A9-eluting stents (BioMatrix; Biosensors International, Singapore or Nobori: Terumo, Tokyo, Japan). Standard therapies after PCI included aspirin, clopidogrel, beta-blockers, lipid-lowering agents, and angiotensin-converting enzyme inhibitors or angiotensin-II receptor blockers in accordance with the guidelines. 10 Clinical follow-up was performed periodically at 6-month intervals by a clinical visit or a telephone interview. All patients were adherent to medication after hospital discharge during the follow-up periods.
Assessment of microvascular function
The IMR was measured to assess the status of the microcirculation independently of the epicardial area after successful primary stenting in all patients. A coronary pressure/temperature sensor-tipped guidewire (Radi pressure wire; Certus-Radi Medical Systems, Uppsala, Sweden) was introduced distally in the culprit lesion. Hyperemia was induced by 140 µg/kg/minute of intravenous adenosine and preceded by a 2-mL intracoronary bolus of 200 µg of nitrate. The mean aortic and distal coronary pressures were recorded during maximal hyperemia. The IMR was calculated as distal coronary pressure multiplied by the mean transit time of a 3-mL bolus of saline at room temperature during maximal coronary hyperemia.11,12 The proportion of an IMR >40, which is associated with a poor clinical outcome, 13 was compared between the groups. We also evaluated the TIMI myocardial perfusion (TMP) grade as described previously 14 from the final recorded images as follows: 0, no myocardial blush; 1, minimal myocardial blush or contrast density; 2, moderate myocardial blush or contrast density, but less than that obtained during angiography of a contralateral or ipsilateral non-infarct-related coronary artery; and 3, normal myocardial blush or contrast density, similar to that obtained during angiography of a contralateral or ipsilateral non-infarct-related coronary artery.
Study endpoints
The primary endpoint was myocardial perfusion assessment using post-PCI IMR values. The secondary endpoints were the TMP grade, ST-segment resolution, left ventricular function recovery, and the occurrence of major adverse cardiovascular events (MACE), which are a composite of cardiac death, target-vessel related myocardial infarction (MI), and target-lesion revascularization (TLR). ST-segment resolution was defined as the percentage of ST-segment reduction between baseline and 90 minutes after PCI. Incomplete ST-segment resolution was defined as an ST-segment resolution <50%. Clinical events were analyzed in a post-hoc manner in accordance with the definition by the Academic Research Consortium. 15 Death was considered cardiac, unless an evident noncardiac cause could be established. MI was defined by an elevation in cardiac biomarkers with at least one value above the 99th percentile of the upper reference limit, with concomitant ischemic symptoms or electrocardiographic findings indicative of ischemia unrelated to an interventional procedure. Target vessel-related MI included MI that could not be clearly related to another vessel. TLR was defined as any repeated PCI of the target lesion, or bypass surgery of the target vessel performed for restenosis or for other target lesion complications. Transthoracic echocardiography was performed within 24 hours of presentation. Follow-up echocardiography was obtained approximately 6 months after the acute event. The percentage change in the ejection fraction (EF) between baseline and the 6-month follow-up was calculated as the difference between the follow-up EF and the baseline EF, divided by the baseline EF, and multiplied by 100% to assess left ventricular functional recovery. All angiographic and IMR data were centralized and analyzed separately by two cardiologists. Electrocardiographic and echocardiographic data were blindly analyzed for the IMR and angiographic data. All analyses were on an intention-to-treat basis.
Statistical analysis
The primary endpoint of our study was the IMR value. A sample size calculation was not performed because there have only been a few studies that referred to the IMR value at the beginning of the study. This study planned to register patients with STEMI who visited the hospital for 1 to 2 years. Therefore, this study can be considered to be a pilot study. Continuous variables are reported as the mean with standard deviation or median, and categorical variables are reported as percentages. Continuous variables were analyzed using the Mann–Whitney U-test and the Wilcoxon signed-rank test. Categorical variables were compared using the Pearson χ2 test or Fisher’s exact test. Log-rank and Kaplan–Meier tests were used to compare the event rate of MACE between the DS and CS groups. These analyses were carried out using IBM SPSS version 23.0 (IBM Corp., Armonk, NY, USA) and R Statistical Software (version 3.6.0; www.r-project.org). All tests were two-sided, and the results were considered statistically significant at P < 0.05.
Results
Patients’ characteristics
Seventy-two patients who underwent primary PCI were randomized to undergo either direct (n = 34) or conventional stenting (n = 38). The baseline characteristics were well matched between the DS and CS groups (Table 1). There were no significant differences in the door-to-balloon time, symptom onset to balloon time, or the use of medication at discharge between the groups. Angiographic and procedural characteristics of the two groups are shown in Table 2. Manual TA was performed in 65 (90.3%) patients. Direct stenting failed in three (8.8%) patients in whom pre-dilatation was necessary before successful stenting could be achieved. The baseline TIMI flow grade was similar in the two groups. The stent size, and the rates of using TA, a glycoprotein IIb/IIIa inhibitor, and adjuvant ballooning were also comparable between the groups.
Baseline characteristics.

LDL, low-density lipoprotein; HDL, high-density lipoprotein; CK-MB, creatine kinase-MB; BNP, brain natriuretic peptide; hsCRP, high-sensitivity C-reactive protein; RWMSI, regional wall motion score index; ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker.
Angiographic and procedural characteristics.

TIMI, thrombosis in myocardial infarction.
Postprocedural findings
The procedural outcomes immediately after PCI are shown in Table 3. The incidence of angiographic no-reflow during PCI and a final TIMI flow grade of 2 after PCI appeared to be higher in the CS group than in the DS group, but this was not significant. Cardiogenic shock during PCI occurred in one (2.9%) patient in the DS group and in four (10.5%) patients who were in the CS group. In 11 patients, the IMR could not be measured because of the development of cardiogenic shock, ventricular fibrillation, or no-reflow during the procedure. The proportion of patients in whom the IMR was unable to be measured for those reasons was significantly higher in the CS group than in the DS group (P = 0.036). The post-PCI IMR value was not different between the DS and CS groups (Table 3 and Figure 2). The number of patients with an IMR >40 was also similar between the groups. Although no significant difference in the mean IMR value was found between the two groups, the number of patients with hemodynamic instability where the actual IMR might be high, but could not be measured, was high in the CS group. Microvascular dysfunction evaluated by the TMP grade immediately after PCI and electrocardiographic findings 90 minutes after PCI, such as the sum of the height of residual ST elevation and the percentage of ST-segment resolution, tended to be worse in the CS group than in the DS group. However, these findings were not significant. Additionally, these findings were consistent, regardless of the lesion territory between the left anterior descending artery (LAD) and non-LAD infarction groups (Figure 3).
Study outcomes.
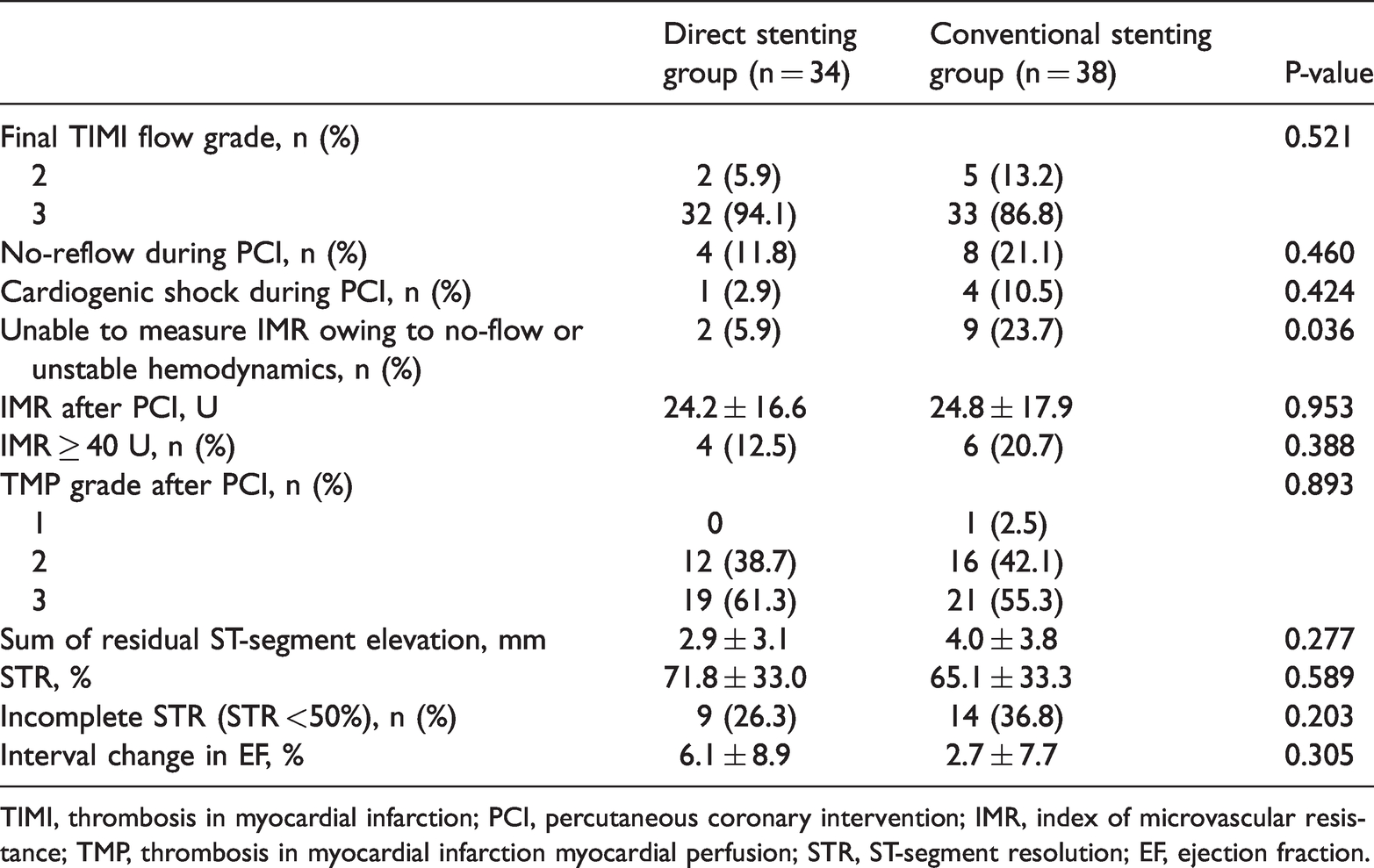
TIMI, thrombosis in myocardial infarction; PCI, percutaneous coronary intervention; IMR, index of microvascular resistance; TMP, thrombosis in myocardial infarction myocardial perfusion; STR, ST-segment resolution; EF, ejection fraction.

Primary endpoint according to treatment groups.

Comparison of LAD and non-LAD infarctions.
Left ventricular functional recovery and long-term clinical outcome
Between baseline and the 6-month follow-up, no difference was observed in the change in the left ventricular EF in the two groups (Figure 4). The mean clinical follow-up duration was 71.8 ± 36.0 months. The Kaplan–Meier curve showed that there was no difference in the cumulative incidence of MACE between the CS and DS groups (Figure 5). The overall rates of MACE during the follow-up were 3.2% (1 TLR) in the DS group and 7.9% (1 cardiac death, 1 target vessel-related MI, and 1 TLR) in the CS group.

Comparison of left ventricular functional recovery in the direct stenting and conventional stenting groups.

Kaplan–Meier curves for MACE in the direct stenting and conventional stenting groups.
Discussion
In this prospective, randomized pilot study, direct stenting without a predilatation strategy did not show a significant effect on reducing microvascular dysfunction or adverse cardiac events compared with conventional PCI after predilatation. However, the number of patients in whom we were unable to measure the IMR owing to the no-reflow phenomenon or cardiogenic shock during PCI, but who might have had severe microvascular dysfunction, was significantly less in the DS group than in the CS group. This finding may have contributed to our conclusion that the DS strategy did not significantly affect post-PCI IMR. The ST-segment resolution, recovery of EF, and long-term clinical outcomes appeared to be more favorable in the DS group, but this was not significant. Our study suggested that direct stenting can be applied safely and effectively in selected patients with STEMI after TA. A further, larger, randomized trial is required to confirm the benefit of this strategy for reducing microvascular dysfunction.
Currently, primary PCI using DESs is performed in patients with STEMI as a standard therapy. This strategy is successful in reopening the infarct-related artery with achievement of a TIMI flow grade of 3 in >90% of patients.16,17 Nevertheless, distal embolization of a fragmented thrombus or atheromatous debris, which can result in greater microvascular obstruction and a poor clinical outcome, frequently occurs during the procedure and remains a main limitation of the treatment.1,2,18,19 Therefore, development of better strategies aimed at reducing distal embolization and microvascular dysfunction is of great interest, in which direct stenting could be one of such approaches. Several possible advantages of direct stenting over conventional PCI have been previously proposed. 20 Skipping balloon predilatation in direct stenting might reduce the risk of mechanical thrombus fragmentation and distal embolization, and thus decrease subsequent microvascular obstruction. Additionally, vessel wall injury and an inflammatory response might also be reduced, which in turn could allow the endothelium to be better preserved. Furthermore, this could promote re-endothelialization of the stented vessel segment, which may decrease the risk of stent thrombosis and neointimal hyperplasia. 20 In addition, direct stenting may shorten the procedural time and reduce radiation and contrast exposure.21–23 However, failure to reach or cross the lesion, underestimation of the vessel size or incorrect placement of the stent due to incomplete lesion visualization, and stent underexpansion in calcified lesions can be potential disadvantages of direct stenting. 20
In previous studies, direct stenting appeared to improve myocardial reperfusion as observed by an improvement in ST-segment resolution and a considerable reduction in no-reflow, and was also associated with decreased adverse outcomes.7,24 However, such results should be interpreted cautiously because they were from non-randomized studies, which allowed a choice of stenting strategies according to the operator’s discretion. The selection of open vessels before stenting, with more focal or less complex lesions, in the DS group could have affected the reperfusion time, which is an important factor for infarct size and patients’ survival. Recently, using merged patient-level data from the three largest randomized trials on routine TA vs. PCI alone,25–27 the Thrombectomy Trialists Collaboration 22 reported that direct stenting in combination with TA during primary PCI did not improve clinical outcomes or myocardial reperfusion measurements. Although routine application of manual TA is not recommended in the current guideline, 28 TA can facilitate direct stenting in total obstruction of the infarct-related artery. Direct stenting was more frequently performed in patients assigned to TA than in patients assigned to PCI alone in the analysis mentioned above, but there were no synergic effects or significant interactions. 22 Although the study performed propensity matching, the conclusion was limited owing to the non-randomized nature of comparison. In our study, with TA implementation in >90% of the patients and the randomized design, there was no relevant benefit regarding direct stenting in terms of microvascular reperfusion and the long-term clinical outcome.
Randomized, controlled studies comparing these two PCI strategies are sparse. There have been five randomized trials with a small sample size.29–33 A meta-analysis of these trials, which included 754 patients, reported a higher ST-segment resolution rate (69% vs. 60%; P = 0.05) and a lower rate of in-hospital cardiovascular death (0.3% vs. 2.1%; P = 0.02) in the DS group than in the CS group. 34 However, these findings should be interpreted with caution because they were all conducted between the late 1990s and early 2000s with older treatments. These older treatments were different from those in the current PCI era, including the use of ticlopidine as antiplatelet therapy and implantation of bare-metal stents or first-generation DESs. In addition, the inclusion criteria of all but one trial 33 were not limited to patients with STEMI, but also to patients with a broad spectrum of acute coronary syndrome and even those with stable angina. In contrast, in our study, only patients with STEMI were enrolled, and all patients were treated with second-generation DESs.
Patients showing an initial TIMI flow grade of 0 or 1 comprised 74% of the total cohort, and most of them had TA performed. Furthermore, approximately 50% of patients were administered glycoprotein IIb/IIIa inhibitors, which can enhance myocardial perfusion in those with a large thrombotic burden. By using all common practices in the contemporary era and admitting patients with STEMI and a high thrombotic burden, our study managed to demonstrate the effect of direct stenting more accurately than previous studies. Although we reached the conclusion that direct stenting did not have a significant benefit on microvascular reperfusion and clinical outcomes, direct stenting might have some benefit in microvascular protection. This benefit is possible because the IMR could not be measured in some patients with predilatation, and they were likely to have severe microvascular dysfunction. Therefore, a further, larger scale, randomized trial using other modalities that can evaluate microvascular function in addition to the IMR is required to confirm the efficacy of the direct stenting.
This study has several limitations. First, our results were obtained from a single institution with experienced interventionists. Therefore, our results cannot be applied to all patients with STEMI. Second, because a sample size calculation was not performed, a small sample size limited the power to detect significant differences in microvascular dysfunction, and it is the main limitation to our study. Third, evaluating microvascular function with functional techniques, such as the IMR, which was the primary endpoint, cannot always be measured in urgent situations. Eleven patients were unable to have the IMR measured owing to insufficient coronary flow recovery or unstable vital signs, and they were likely to have a high IMR. Therefore, caution should be paid to the interpretation of our results. Fourth, other modalities, such as radionuclide 99 m technetium-sestamibi single-photon emission tomography myocardial perfusion imaging or cardiovascular magnetic resonance imaging, which can quantitatively measure the degree of microvascular dysfunction, were not implemented in our study. Fifth, ticagrelor or prasugrel, which are currently widely used in patients with STEMI, were not used in this cohort. Sixth, microvascular function assessment was only performed immediately after PCI. Seventh, the procedure and fluoroscopic time and requirement of contrast, which can be advantages of direct stenting, were not analyzed in this study. Finally, although there were no significant differences in any variables between the groups, the uncontrolled use of TA and glycoprotein IIb/IIIa inhibitors might be potential biases that affected the results. Nevertheless, this study is the most recent randomized, controlled trial that compared direct stenting versus conventional stenting. Our trial also reflects the current clinical practice with second-generation DES implantation, and highlights the high application rate of TA and glycoprotein IIb/IIIa inhibitors in common procedures in real practice.
In conclusion, this study showed that, among patients who were able to have the IMR measured, direct stenting during primary PCI did not have a remarkable benefit regarding microvascular protection or the long-term clinical outcome in the current DES era. However, the number of patients in whom the IMR was unable to be measured owing to subsequent cardiogenic shock events or no-reflow was significantly fewer in those who had direct stenting than in those who had conventional stenting. Therefore, direct stenting can be performed safely and might be considered in selected patients with STEMI who achieve a TIMI grade 2 or 3 flow by TA. An adequately-powered, randomized trial is warranted to definitely address the efficacy of direct stenting in the future.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221127888 - Supplemental material for Effect of direct stenting on microvascular dysfunction during percutaneous coronary intervention in acute myocardial infarction: a randomized pilot study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221127888 for Effect of direct stenting on microvascular dysfunction during percutaneous coronary intervention in acute myocardial infarction: a randomized pilot study by Byung Gyu Kim, Sung Woo Cho, Jongkwon Seo, Gwang Sil Kim, Moo-Nyun Jin, Hye Young Lee, Young Sup Byun, Byung Ok Kim in Journal of International Medical Research
Footnotes
Author contributions
Conceptualization, B.O.K.; methodology, B.O.K.; formal analysis, B.G.K. and S.W.C.; investigation, B.O.K. and Y.S.B.; resources, J.S., M.N.J., G.S.K. and Y.S.B.; data curation, H.Y.L.; writing—original draft preparation, B.G.K.; writing—review and editing, S.W.C. and B.O.K.; visualization, B.G.K.; supervision, B.O.K.; funding acquisition, B.O.K. and B.G.K. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Korean Society of Cardiology (201003-05) and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2020R1F1A1076096).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
