Abstract
Cryptococcus neoformans is an environmental fungal pathogen that causes opportunistic infections and severe disseminated meningoencephalitis, mainly in immunocompromised patients such as those with acquired immunodeficiency syndrome (AIDS). In this study, the clinical characteristics, treatment protocols, and outcomes of 70 patients with AIDS and Cryptococcus neoformans infection at Beijing Ditan Hospital were retrospectively analyzed. We performed antimicrobial sensitivity tests and multilocus sequence typing (MLST) on C. neoformans isolates from these patients. The most common symptoms were headache (58.6%), fever (54.3%), and high cerebrospinal fluid pressure (≥200 mm H2O) (71.4%). All patients were positive for C. neoformans antigen in blood or cerebrospinal fluid. The CD4 cell counts of 92.8% (65/70) of patients were <100 cells/µL. In total, 74 C. neoformans isolates were obtained from the 70 patients. The 65 isolates that could be typed fell into 12 sequence types (STs) by MLST: ST5, ST31, ST63, ST202, ST237, ST289, ST295, ST296, ST298, ST324, ST337, and ST359. ST5 was the major type, accounting for 78.5% of isolates (51/65). This study comprehensively assessed the clinical and molecular epidemiology of C. neoformans in patients with AIDS and may inform the development of targeted prevention and treatment strategies for immunocompromised patients with C. neoformans infection.
Keywords
Introduction
Cryptococcus neoformans is an opportunistic fungal pathogen that causes severe disseminated meningoencephalitis in humans. Severe disease occurs mainly in immunocompromised patients such as those with acquired immunodeficiency syndrome (AIDS). Globally, cryptococcal infection leads to approximately 957,900 cases of cryptococcal meningitis in patients with AIDS each year. Approximately 624,700 of these patients die within 3 months after infection. 1 From 1985 to 2010, 8769 patients developed cryptococcosis in China; 16% of these patients were human immunodeficiency virus (HIV) positive. Mortality rates vary from 23% to 35% depending on treatment. 2
Many molecular typing methods are used in molecular epidemiological analyses such as random amplified polymorphic DNA, restriction fragment length polymorphism, amplified fragment length polymorphism, internal transcribed spacer rDNA, and multilocus sequence typing (MLST). 3 Different typing methods can categorize isolates into subgroups with a variety of names. In 2007, the ISHAM Cryptococcus Working Group decided to use MLST as the gold standard for molecular typing of Cryptococcus species because of its high resolution and good reproducibility. Seven housekeeping genes (CAP59, GPD1, LAC1, PLB1, SOD1, URA5, and IGS1) were selected as loci in the global consensus plan.
The genus Cryptococcus contains many species. C. neoformans and C. gattii cause the majority of infections in humans. 4 Epidemiological studies have shown that C. neoformans is distributed worldwide and mainly causes infection in patients with HIV/AIDS, especially in patients with CD4 T cell counts <50 cells/μL. 5 , 6 In contrast, C. gattii is mainly found in tropical and subtropical regions and most infections occur in immunocompetent individuals. 7
Nearly all C. neoformans strains analyzed in China in previous studies were isolated from HIV-negative patients. 5 , 8 , 9 Little is known about the clinical characteristics, drug sensitivity, and genotypes of C. neoformans infection in HIV-positive patients in China. There are substantial differences in the epidemiology, clinical features, therapeutic approaches, and outcomes of cryptococcosis in HIV-positive and -negative patients. The objective of this study was to summarize the clinical characteristics, drug sensitivity, and genotypes of C. neoformans in patients with HIV in Beijing, China. Our results will serve as a basis for further molecular epidemiological studies and to guide therapeutic interventions.
Materials and methods
Ethics
The study protocol was reviewed and approved by the Institutional Review Committee of Beijing Ditan Hospital (approval number: 2018-004). The requirement for informed consent was waived based on analysis of isolates for routine drug susceptibility testing only. No information that could potentially be used to identify patients was analyzed in the study.
Clinical data and strains
Patients with HIV and C. neoformans infection at Beijing Ditan Hospital were enrolled from June 2017 to July 2019. We retrospectively analyzed the clinical characteristics of patients and collected data from cerebrospinal fluid (CSF) analyses including protein, glucose, and chlorine levels as well as CD4 and CD8 cell counts. All C. neoformans strains were isolated from clinical samples including blood, CSF, and bronchial alveolar lavage fluid. The privacy of patients was protected and all details were de-identified that the identity of the patients could not be ascertained.
Isolation, characterization, and antimicrobial sensitivity testing of C. neoformans
Clinical specimens were processed strictly according to the procedures of the National Center for Clinical Laboratories (Beijing, China). Briefly, blood cultures were monitored using an automated blood culture instrument (BACTEC™ FX, BD Biosciences, Franklin Lakes, NJ, USA). When pathogenic yeasts in blood cultures grew to a certain degree, the instrument triggered an alarm and cultures were transferred to blood agar and MacConkey agar plates (Thermo Fisher Scientific, Waltham, MA, USA). CSF specimens were centrifuged at 3000 × g for 15 minutes. The pellet was used to inoculate blood agar and chocolate agar plates. A 10-μL sample of bronchial alveolar lavage fluid was used to inoculate blood agar, MacConkey agar and Sabouraud gentamicin chloramphenicol (SGC) agar plates. All plates were incubated at 37°C for 48 hours.
After incubation, yeast-like colonies were selected for Gram staining and transferred onto SGC plates. Colonies were identified after 24 hours of incubation using API 20 C AUX strips (bioMérieux, Marcy-l'Étoile, France) and antifungal drug sensitivity was performed using ATB™ FUNGUS 3 strips (bioMérieux). Briefly, yeast-like colonies were prepared as McFarland Standard 2.0 suspensions, then 20 μL of each suspension was added to ATB F2 culture medium. A 135-µL sample of the mixture were added to the ATB™ FUNGUS 3 strips. After incubating for 48 hours in a 35°C incubator, the strips were evaluated according to the manufacturer’s instructions. Kit instructions indicated breakpoints for only 5-fluorocytosine and fluconazole. For 5-fluorocytosine, a minimum inhibitory concentration (MIC) ≤4 mg/L was interpreted as sensitive, a MIC of 8 to 16 mg/L was interpreted as intermediate, and a MIC ≥32 mg/L was interpreted as resistant. For fluconazole, a MIC ≤4 mg/L was interpreted as sensitive, a MIC of 8 mg/L was interpreted as intermediate, and a MIC ≥16 mg/L was interpreted as resistant. In addition to characterization using API 20 C AUX strips, all isolates were assessed using matrix-assisted laser desorption/ionization mass spectrometry (VITEK® MS, bioMérieux).
CSF analyses and CD4/CD8 cell counts
Levels of glucose and protein in CSF were assessed using a Hitachi 7600 automatic biochemical analyzer (Hitachi, Tokyo, Japan). C. neoformans in CSF was examined using India ink staining. Cryptococcal antigens in serum were detected using a latex agglutination assay (IMMY, Norman, OK, USA). CD4 and CD8 T cells were assayed using flow cytometry (FACSCalibur, BD Biosciences).
MLST
Genomic DNA was extracted from colonies on SGC medium and purified using DNeasy Plant Mini Kits (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. MLST was performed using the published scheme (CAP59, URA5, GPD1, SOD1, LAC1, IGS1, PLB1). 10 The C. neoformans MLST scheme and allele definitions, the sequences of the primers, and the polymerase chain reaction conditions are available from the Fungal MLST Database (http: //mlst.mycologylab.org/cneoformans). The genetic relationships among sequence types (STs) were analyzed by constructing a minimal spanning tree using Phyloviz 2.0 software. 11
Statistical analysis
All statistical analyses were conducted using SPSS 19.0 software (IBM, Armonk, NY, USA). Differences in survival by genotype were assessed using the chi-square test. One-way analysis of variance was used to differences in survival or genotype by susceptibility to five antifungal drugs. Values of p < 0.05 were considered statistically significant.
Results
Clinical characteristics of patients and C. neoformans strain isolation
Among the 70 patients with HIV and C. neoformans infection, 87.1% were male (61/70) and 61.4% (43/70) were ≤40 years old. Most patients had symptoms of headache (58.6%) and fever (54.3%) (Table 1), and 71.4% (50/70) had relatively high CSF pressures (≥200 mm H2O). In 85.7% of patients, CSF leukocyte counts were >10 × 106 cells/L. CSF India ink staining was positive in 74.3% of patients. All patients were positive for C. neoformans antigen in blood or CSF, and 92.8% (65/70) of patients had CD4 cell counts of <100 cells/µL. However, only 20 patients (28.5%, 20/70) had CD8 cell counts of <500 cells/µL (Table 1).
Clinical characteristics and laboratory parameters of 70 patients AIDS and Cryptococcus neoformans infection.

*Other includes fatigue, poor appetite, decreased consciousness, decreased appetite, tinnitus, dyspnea, blurred vision, or diarrhea.
CSF, cerebrospinal fluid; AIDS, acquired immunodeficiency syndrome; WBC, white blood cell.
We isolated 74 C. neoformans strains from the 70 patients with HIV; in four patients, we isolated strains from both blood and CSF. Among the 74 strains (all identified as C. neoformans), 23 were isolated from blood, 45 were isolated from CSF, and six were isolated from alveolar lavage fluid.
Drug susceptibility, treatments, and outcomes
We analyzed the susceptibility of the 74 C. neoformans strains to five commonly used antifungal agents (5-fluorocytosine, amphotericin B, fluconazole, itraconazole, and voriconazole). MIC values for these agents are shown in Table 2. Among the 74 strains, one was resistant to 5-fluorocytosine (1.4%) and five were resistant to fluconazole (6.8%).
Drug susceptibility of 74 Cryptococcus neoformans strains to five antifungal agents.

MIC, minimum inhibitory concentration.
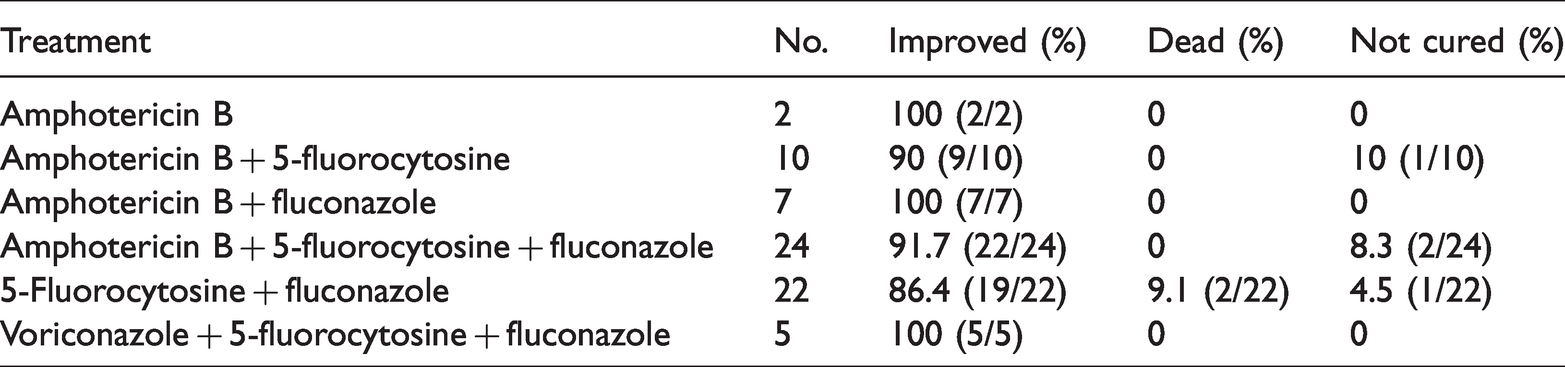
The main antifungal drugs used to treat patients were amphotericin B, 5-fluorocytosine, fluconazole and voriconazole. In most cases a combination of these drugs was used (Table 3). The treatment plan was consented to by each patient. Mannitol and glycerol fructose were also used simultaneously to reduce intracranial pressure, and glucocorticoids were used to suppress inflammatory responses. Overall, six treatment combinations were used among the 70 patients. Most patients had a good prognosis. Sixty-four patients (91.4%) improved after treatment and were discharged. Four patients chose to discontinue treatment and two patients died following combination therapy with 5-fluorocytosine and fluconazole.
Treatment and prognosis of patients included in the study.
MLST typing and phylogenetic analysis
Among the 74 strains of C. neoformans, 65 were successfully typed. Among typed strains, 12 STs were identified including ST5 (51 isolates), ST31 (2 isolates), ST63 (1 isolate), ST202 (1 isolate), ST237 (1 isolate), ST289 (1 isolate), ST295 (1 isolate), ST296 (2 isolates), ST298 (1 isolate), ST324 (1 isolate), ST337 (1 isolate), and ST359 (2 isolates) (Table 4, Figure 1). ST5 was the dominant ST, accounting for 78.5% (51/65) of strains. Clonal complex 5 (CC5), containing ST5 and five other STs distinct in one allele only, accounted for 89.2% (58/65) of strains. These results indicated that CC5 was the most prevalent clonal complex among the patients included in our study.
MLST typing of 65 Cryptococcus neoformans isolates.

*Allele sequence: CAP59, GPD1, IGS1, LAC1, PLB1, SOD1, and URA5.
MLST, multilocus sequence typing.

Minimum spanning tree based on MLST of Cryptococcus neoformans isolates. The tree was constructed with Phyloviz software based on the 74 clinical strains isolated at Beijing Ditan Hospital between 2009 and 2018. Each circle represents a unique ST with size proportional to the number of isolates within each ST. Numbers within circles represent STs, while numbers next to connecting lines indicate the number of allelic differences between two STs based on the seven gene MLST scheme (maximum five allelic differences are shown).
We separated the patients into two groups by CD4+ T cell count (>20 or ≤20 cells/µL). There were no significant differences in the distributions of strains or STs between the two groups.
Correlational analysis
There was no significant correlation between survival and C. neoformans genotype. MIC values of fluorocytosine, itraconazole and voriconazole were not affected by genotype, while MIC values of amphotericin B and fluconazole were significantly affected by genotype (p < 0.001 and p = 0.002, respectively). There was no significant correlation between survival and susceptibility to the five antifungal drugs (Table 5).
Correlations between survival, Cryptococcus neoformans genotypes, and susceptibility to five antifungal drugs.

Discussion
In China, the incidence of HIV is approximately 0.05%, and is growing at a rate of 30% every year (https://www.who.int/). This is a very serious problem. A variety of opportunistic infections occur in immunocompromised patients, of which C. neoformans is the most common central nervous system infection. 4 , 12 , 13
Previous studies showed that intracranial hypertension was an important indicator of poor prognosis in patients with cryptococcal meningitis. 14 , 15 Other indicators included elevated levels of CSF protein and reduced levels of sugar, although chlorine levels generally did not change significantly. These results were consistent with our findings (Table 1). In addition to CSF pressure, helper T cells were also an important indicator of prognosis. Cryptococcosis typically occurs in HIV patients with profound immunosuppression and functional abnormalities of CD4 T cells. The higher a patient’s CD4 T cell count, the lower their risk of secondary fungal infection. These patients also have better recovery following treatment. 16 Our results were consistent with these findings. Only 7.1% (5/70) of patients had CD4 T cell counts of >100 cell/µL and 45.7% (32/70) of patients had CD4 T cell counts < 20 cell/µL (Table 1).
Amphotericin B remains the cornerstone of induction therapy for cryptococcal meningitis. 17 Studies have suggested that higher daily doses of amphotericin B may be more effective in sterilization of the CSF. 18 The patients included in this study were at varying stages of the disease. Accordingly, six treatment regimens, and most patients had a good prognosis. Among 70 patients, 64 improved and were discharged, four did not improve and discontinued treatment, and two died. Both patients who died were treated with 5-fluorocytosine combined with fluconazole. In vitro drug sensitivity analysis of C. neoformans isolates from these two patients showed that one isolate was resistant to 5-flucytosine and showed intermediate sensitivity to fluconazole, while the other isolate showed intermediate sensitivity to 5-fluorocytosine and was resistant to fluconazole. The genotypes of both isolates were ST5. These results demonstrate that clinical treatment should be adjusted based on drug sensitivity testing results.
In addition to clinical characteristics, antifungal susceptibility, and treatment outcomes, we also analyzed the genotypes of C. neoformans isolated from the patients studied. To date, most research on C. neoformans genotypes has focused on environmental isolates or isolates from HIV-negative patients 19 , 20 in China. In a previous study, Chen 5 collected 129 C. neoformans isolates in China; only 11 isolates (8.5%) were from AIDS patients, in contrast with other studies. Studies in the United States and Africa reported that the majority of cryptococcosis patients had AIDS (>80% and >69%, respectively). 21 , 22
In this study, we enrolled 70 patients and isolated 74 C. neoformans strains. For four patients, C. neoformans was isolated from both the blood and CSF. Three of the four patients had the same genotype in blood and CSF (ST5), but one patient had different genotypes in blood and CSF (ST337 in blood and ST298 in CSF). Both these genotypes belong to CC5 and differ in only one allele. We extracted nucleic acids from 74 strains of C. neoformans for genotyping, but only 65 strains were successfully typed. In the other nine strains, typing failed because unsuccessful amplification and/or amplified sequences not matching with reference sequences. More experimentation is needed to further understand the specific reasons for genotyping failure in these specimens.
As shown in Table 4, C. neoformans isolates belonged to 12 STs. ST5 was the dominant ST, accounting for 78.5% (51/65) of isolates. In a previous study, Khayhan et al. 23 also used MLST to genotype 476 strains of C. neoformans from patients in eight Asian countries. Their results showed that C. neoformans isolates mainly belonged to ST5 in HIV-negative patients, while in HIV-positive patients, ST4, ST6 and ST5 accounted for 72 (31.6%), 68 (29.8%) and 27 (11.8%) of strains, respectively. These previous findings varied slightly from our results. One explanation may be differences in patient location. In the previous study, patients were included from various regions of Asia including Shanghai, Hong Kong, Tokyo, Japan, and Southeast Asia, whereas in our study, patients were in the Beijing area. Statistical analysis showed no significant correlation between survival and genotype or between survival and susceptibility to five antifungal drugs. However, MICs of amphotericin B and fluconazole were significantly affected by genotype (p<0.001 and p=0.002, respectively) (Table 5). It should be noted that STs were unevenly distributed and ST5 was the dominant type (51/65), which may have impacted our conclusions.
This study had several limitations including its retrospective nature, the relatively small number of patients studied, and relatively few C. neoformans isolates analyzed. Larger studies may reveal more STs, providing additional molecular epidemiological information. In addition, our study of strains was limited in its depth. If serotypes or mating types could be analyzed, the genetic characteristics of C. neoformans isolates from HIV-positive patients might be better understood.
Understanding the clinical characteristics and drug susceptibility of C. neoformans in combination with molecular typing is important to comprehensively assess the clinical and molecular epidemiology of this organism in patients with AIDS. This information could contribute important new insights into the pathogenesis, early recognition, treatment, and prevention of complicated cryptococcal infections.
Footnotes
Author’s contributions
Xinmin Xu and Yuanyuan Zhang contributed to the conception of the study, prepared the manuscript, and were involved in overall supervision. Yajie Wang revised the manuscript. Pengcheng Du and Huizhu Wang analyzed the data. Weijie Li and Xiaoling Yang collected the data. All authors read and approved the final manuscript.
Acknowledgments
We thank the members of the Department of Clinical Laboratory Medicine, Beijing Ditan Hospital, Capital Medical University for advice and assistance. We also thank the members of the Institute of Infectious Disease, Beijing Ditan Hospital for technical support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by Beijing Key Laboratory of Emerging Infectious Diseases Project (grant number DTKF201801).
