Abstract
Objective
To assess the efficacy, acceptability, and tolerability of a vaginal cream based on plant extracts for treating signs and symptoms of vulvovaginitis (VV) (Zelesse cream®), either as monotherapy (non-infectious VV) or adjuvant to antimicrobial therapy (infectious VV).
Methods
This prospective, observational, multicenter study included women who attended outpatient offices for VV. The severity of signs (vaginal discharge, erythema, and edema) and symptoms (pruritus, burning, and dysuria) was assessed before and after 15±5 days of daily treatment with Zelesse cream on a 4-point scale (18-point global score).
Results
The study included 58 women aged 43.0±13.2 years, including 42 who were treated with Zelesse cream only and 16 who used Zelesse cream as adjuvant to antimicrobial therapy. All participants showed significantly reduced scores and absolute prevalence of individual signs and symptoms in both groups. Similarly, the median signs/symptoms decreased by 4.0 and 8.0 points in women using Zelesse only and those using Zelesse plus antimicrobial therapy, respectively. This product was well tolerated and had high acceptability.
Conclusions
Zelesse cream relieves signs and symptoms of VV, either as monotherapy in non-infectious VV or as adjuvant to antimicrobial therapy in infectious VV. Future randomized, placebo-controlled trials with larger sample sizes are warranted.
Keywords
Introduction
Vulvovaginitis (VV) is the most common gynecological diagnosis in primary care and most women have at least one episode during their lives. 1 VV is clinically characterized by pruritus, stinging, burning, and dysuria, and it typically presents with an increase in vaginal discharge (leukorrhea), genital inflammation, and vulvar or vaginal pain.1–3 Etiologically, VV can be infectious (e.g., caused by fungi, such as Candida albicans, bacteria, or flagellated protozoa, such as Trichomonas sp.) or non-infectious when caused by unspecific processes related to irritant or allergic contact dermatitis, foreign bodies, or atrophic vaginitis.4,5 VV, and particularly non-infectious or unspecific VV, are associated with multiple risk factors, such as obesity, pregnancy, certain sexual behaviors, poor hygiene, steroids, oral contraceptives, and antibiotics.1,6
The therapeutic approach of VV is primarily based on addressing predisposing factors, alleviating the symptoms, and eliminating the underlying microorganism when there is evidence of infection.1,3 Notably, the diagnosis of VV is often established empirically on the basis of the medical history and a general examination. Some authors have raised concerns on the inappropriate administration of antibiotics for VV, which can ultimately increase the risk of complications, recurrence, and antibiotic resistance.7,8 Regardless of the etiological treatment for VV, various products containing probiotics and plant extracts have shown benefits in relieving vaginal signs and symptoms of VV.9,10
Zelesse cream® is a vaginal cream based on plant extracts (chamomile, burdock, and aloe vera), with anti-pruritic, anti-inflammatory, moisturizing, and re-epithelialization properties. Zelesse cream has been marketed since 2016 for the intended use of improving complaints of VV. The use of a washing solution based on the same composition showed the ability to effectively improve the signs and symptoms of VV in adults while showing good tolerability.11,12 Furthermore, these results were confirmed in female children aged 2 to 8 years with non-infectious VV. 13 In this observational study, we aimed to assess the efficacy, tolerability, and acceptability of Zelesse cream in adult women with signs and symptoms of VV when using this cream as either monotherapy or adjuvant to antibiotic/antifungal treatment.
Patients and methods
Study design and patients
This was an observational, prospective, descriptive, multicenter, clinical study that was performed by 15 investigators (i.e., gynecologists) from private and public centers (the Zelina study). The study included adult women who attended outpatient gynecology offices in Spain for concerns about symptoms (e.g., pruritus, burning, and dysuria) and signs (e.g., erythema, vaginal discharge, and edema) of VV and for whom their gynecologist considered recommending Zelesse cream (ITF Research Pharma S.L.U., Alcobendas, Madrid, Spain) for alleviating their symptoms and signs, either as monotherapy or as adjuvant treatment to antibiotic/antifungal therapy. Participants were recruited consecutively between 1 September 2016 and 30 October 2016 as they started treatment with Zelesse cream. Women who were pregnant, had conditions contraindicating or causing hyper-sensibility to the product’s components, or used local treatments, including hydrating agents, phytoestrogen, or estrogen up to 1 week before the initial visit, were excluded from the study.
To prevent interference with prescription patterns in each study site, treatment was established at the physician’s discretion on the basis of a review of medical records, a physical and gynecological examination, including vaginal pH and assessment of signs/symptoms, and a microscopic examination of a sample of vaginal discharge. 14 When considered necessary according to the investigator’s criteria, vaginal exudate was analyzed to identify specific microorganisms. All study participants were advised to apply Zelesse cream to the vulvoperineal area (with a gentle massage until its complete absorption) twice a day for 15±5 days, preferably after a bath. Participants were assessed at baseline and the end of treatment. The study protocol was approved by the independent ethics committee of the University Hospital Puerta de Hierro-Majadahonda, Spain (approval number: 05.16; March 30, 2016), and all study participants provided written informed consent to participate and were eligible to participate. The study results have been reported by following the STROBE guidelines for observational studies.
Assessments
The demographic and clinical characteristics of the study participants were recorded at a baseline visit by a structured interview. Efficacy was assessed by changes in the prevalence and severity of symptoms/signs between the baseline visit and the final visit upon completion of treatment. Symptoms and signs were rated by the gynecologist on a 4-point scale as follows: 0=absence, 1=mild intensity, 2=moderate intensity, and 3=severe intensity. The sum of the individual scores of all symptoms/signs yielded a global score, which ranged from 1 to 18 points. Total recovery from a VV episode was considered when all signs and symptoms were absent. At the end of treatment, participants and gynecologists were asked about the contribution of Zelesse cream to improvement of signs and symptoms as follows: “great contribution”, “some contribution”, “slight contribution”, or “no contribution”.
Tolerability of Zelesse cream was assessed by the incidence of adverse events and the users’ acceptability of the product, and was rated as “very well accepted”, “well accepted”, “fairly well accepted”, “poorly accepted”, or “very poorly accepted” in the final visit. Upon completion of treatment, participants were also asked about the following outstanding features of Zelesse cream: itching relief properties, soothing action, moisturizing action, antipruritic action, wearing comfort, and others.
Statistical analysis
All participants who used Zelesse cream at least once were included in the analysis. Before performing the statistical analysis, all collected data were validated by double-checking the medical records of the study participants. Quantitative variables are described as the mean and standard deviation (SD) or the median and interquartile range (IQR, defined by 25th and 75th percentiles), whereas categorical variables are described as frequency and percentage of the available data. Results regarding tolerability, acceptability, and preferences were reported for the overall study sample. However, the efficacy outcome was analyzed for two diagnostic groups, including non-infectious VV (Zelesse cream used as monotherapy) and infectious VV (Zelesse cream used as adjuvant treatment to antibiotic/antifungal therapy). Participants who were diagnosed with infectious VV, but did not have an antibiotic/antifungal prescription by a gynecologist, were assigned to the non-infectious VV group because Zelesse cream was administered as monotherapy. Changes in the severity of symptoms and signs from baseline to the final visit were analyzed with Fisher’s exact test or the chi-square test for categorical variables and the signed-rank Wilcoxon test for quantitative variables. Statistical significance was established at an alpha error of 0.05, and no multiplicity adjustments were applied. All statistical analyses were performed using SAS® version 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
Subjects and treatment characteristics
During the recruitment period, 64 women consented to participate in the study. Of them, six were excluded from the analysis for not ever using Zelesse cream (n=2) or for lacking validity control on their data (n=4). Therefore, the full analysis set included 58 women who had used Zelesse cream at least once. Forty-two women were treated with Zelesse cream only (non-infectious VV) and 16 used Zelesse cream as adjuvant treatment to antibiotic/antifungal therapy (infectious VV). Table 1 shows the clinical and demographic characteristics of the study sample and those of each study group. The mean age of the women was 43.0±13.2 years. Overall, women in the non-infectious VV group more frequently reported predisposing factors, such as menopause (most frequent condition), regular use of panty liners, perfumes, and similar products, and use of tight or synthetic fiber underwear, than those in the infectious VV group. Women with previous VV episodes during the previous year in the non-infectious and infectious VV groups had experienced a median of 4.0 and 2.9 episodes, respectively (range: 1–9 in both groups). Most women in the infectious VV group were associated with candidiasis, and the main etiology of non-infectious VV was atrophy (Table 1). Four participants did not receive antibiotic/antifungal agents despite the presence of microorganisms in their vaginal exudates. Therefore, these women were considered non-infectious for analysis.
Demographic and clinical characteristics of the study participants who were grouped by type of diagnosis.
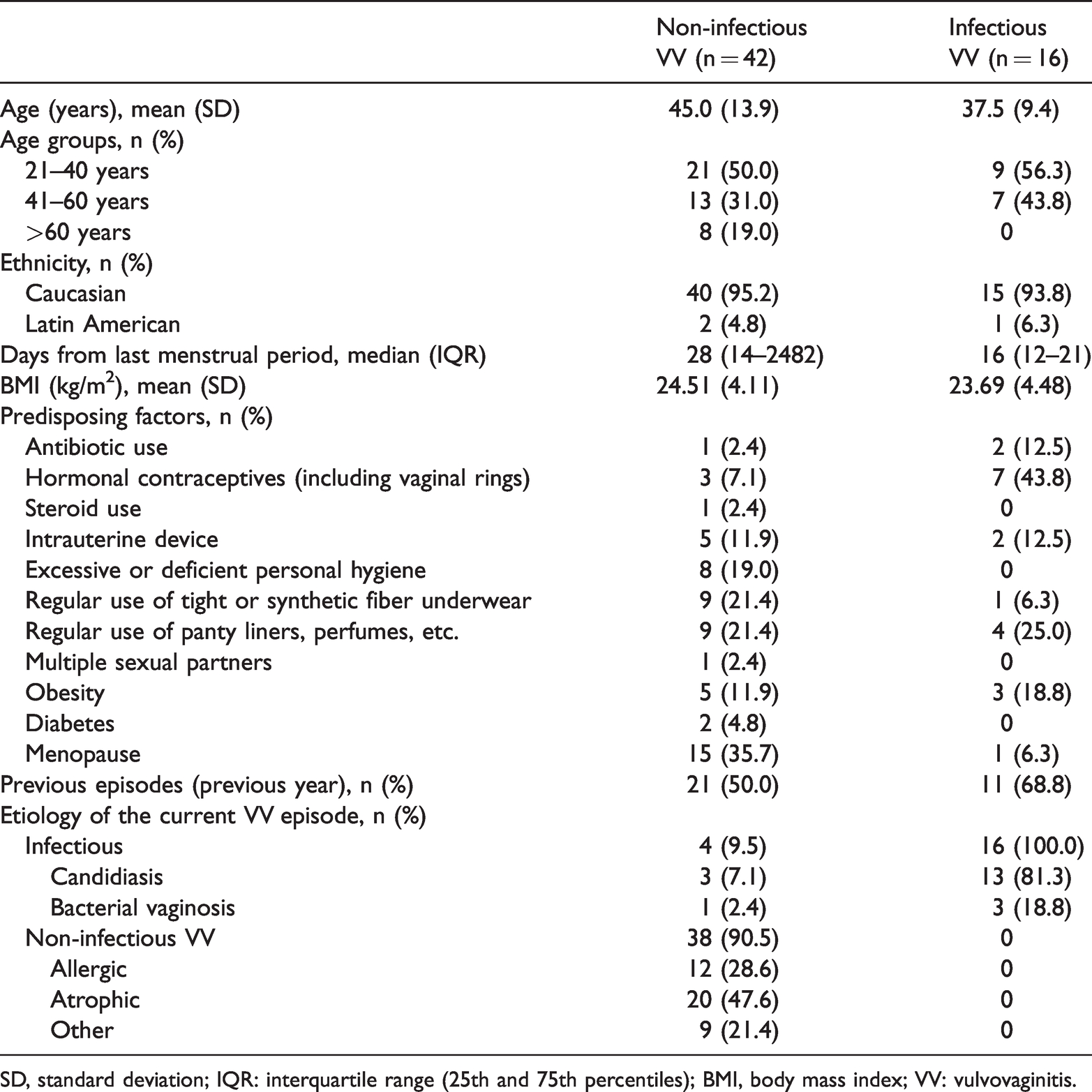
SD, standard deviation; IQR: interquartile range (25th and 75th percentiles); BMI, body mass index; VV: vulvovaginitis.
Participants used Zelesse cream for a median of 14 days (IQR: 14–17), and 6 (10%) of them interrupted treatment at least once. Most of the participants used Zelesse cream twice daily with no interruption, with 86% and 100% in the non-infectious and infectious VV groups, respectively. Most women with infectious VV (n=15, 94%) complied with antibiotic treatment, which was topical and systemic in 14 (88%) and 3 (19%) participants, respectively.
Efficacy
Of all participants who used Zelesse cream for at least 1 day, 43 (74%) experienced great improvement or complete recovery of the VV episode, including 29 (69%) and 14 (88%) in the non-infectious and infectious VV groups, respectively. Patients in the non-infectious VV group perceived improvement of the VV symptoms after a mean of 4.8 days (range: 1–10 days) and a reported a recovery time of 6.8 days (range: 1–10 days). The corresponding times to perceived improvement and recovery in the non-infectious VV group were 3.3 days (range: 0–10 days) and 5.9 days (range: 2–14 days), respectively.
The scores (Table 2) and absolute prevalence (Figure 1) of individual symptoms and signs were significantly decreased after treatment compared with baseline in the two study groups (all p < 0.05, except for dysuria in the infectious VV group [p=0.06]). Similarly, the global scores of symptoms and signs were significantly decreased after treatment compared with baseline in both study groups as follows: the median (IQR) reduction for participants in the non-infectious and infectious VV groups was 4.0 (5.0–3.0) and 8.0 (11.0–5.0), respectively (p < 0.001 for both groups).
Changes in symptom and sign scores from baseline to the final visit by group.

Results are presented as the median and interquartile range (25th and 75th percentiles). Baseline and final scores were compared using the Wilcoxon signed-rank test. Range of the score of individual signs/symptoms: 0 to 3; range of the global score: 0 to 18.
aMean (SD) change was −0.4 (0.7) and−0.3 (0.9) for infectious VV and non-infectious VV, respectively.
bMean (SD) change was −1.3 (1.1) and −0.8 (0.9) for infectious VV and non-infectious VV, respectively.
cMean (SD) change was −0.9 (0.7) and −0.4 (0.7) for infectious VV and non-infectious VV, respectively.
VV, vulvovaginitis; SD, standard deviation.
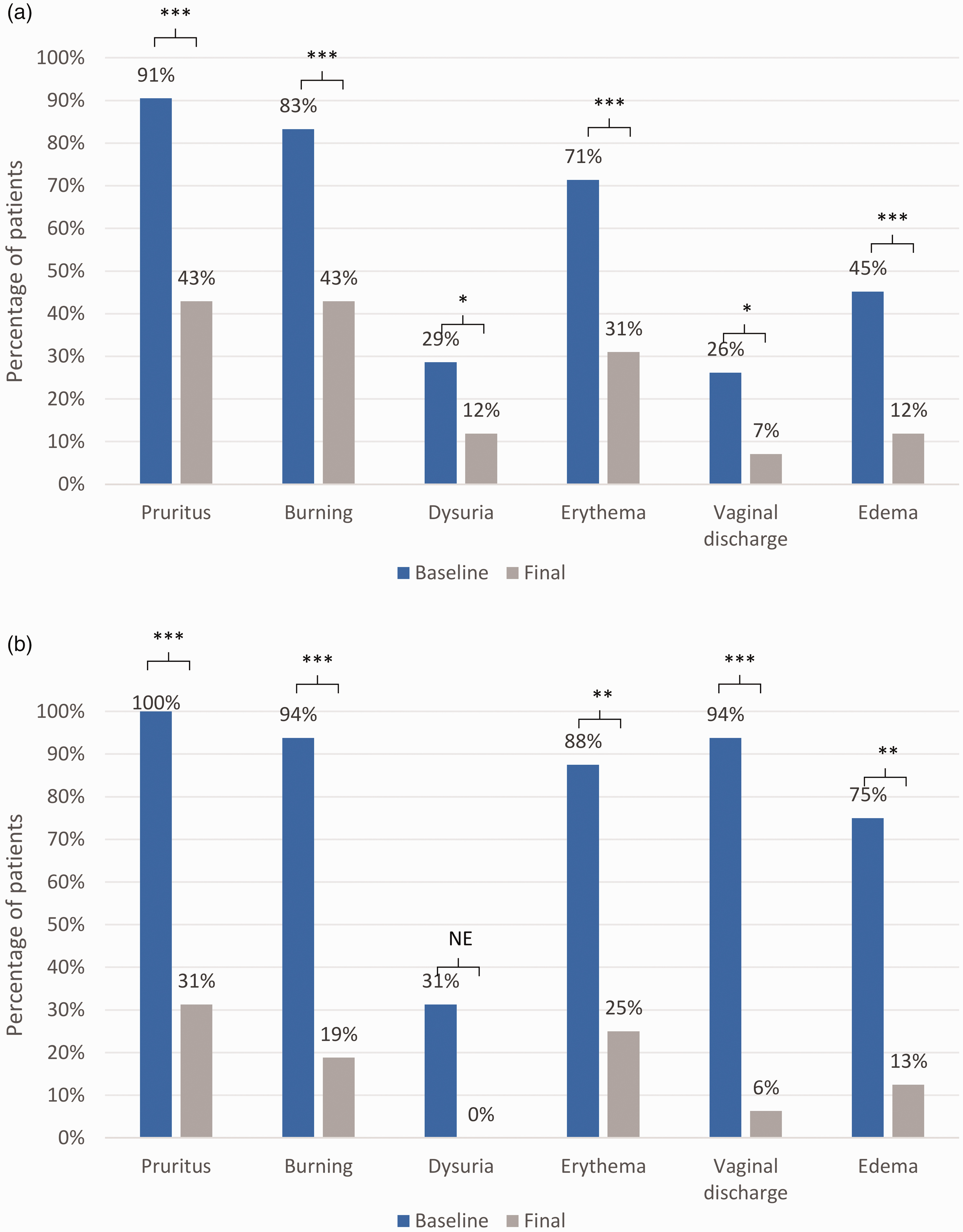
Prevalence of individual signs and symptoms at baseline and after treatment with Zelesse cream in participants with non-infectious VV (a) and in participants with infectious VV (b).
According to the structured questionnaire, most patients considered that Zelesse cream had “some contribution” (n=22, 38%) or “great contribution” (n=26, 45%) to improvement of symptoms. Similarly, most gynecologists considered that Zelesse cream contributed in some way (n=19, 33%) or greatly (n=28, 48%) to improvement of the VV episode. The most frequently highlighted feature of Zelesse cream among participants was its property for relief of itching, followed by its soothing and moisturizing actions, particularly in women in the non-infectious VV group who only received Zelesse cream (Figure 2).

Features of Zelesse cream reported by the study participants.
Acceptability and tolerability
According to the acceptability score, which was rated on a 5-point Likert scale, 49 (84%) participants rated Zelesse cream as good or very good acceptability. The corresponding percentage in the diagnostic groups was higher than 80% (Figure 3).

Acceptability of Zelesse cream reported by the study participants.
Overall, four (6.9%) participants reported a total of six adverse events, and none of them were serious. One patient interrupted treatment for 2 days because of a non-related adverse event. Two patients experienced treatment-related adverse events (i.e., worsening of pruritus and burning after product application) and these events resolved completely after discontinuing treatment.
Discussion
In this observational, prospective study, we found that treatment with a vaginal cream based on chamomile, burdock, and aloe vera (Zelesse cream) had good tolerability. Zelesse cream also significantly reduced the prevalence and severity of signs and symptoms of VV, including pruritus, burning, dysuria, erythema, vaginal discharge, and edema. The ability of Zelesse cream to reduce signs/symptoms of VV was observed in two therapeutic settings of women who used only Zelesse cream for treating non-infectious (unspecific) VV and women who used Zelesse cream as adjuvant treatment to antibiotic/antifungal therapy for infectious VV. Most participants had good or very good acceptability and particularly highlighted the anti-itching, soothing, and moisturizing actions.
As expected, a remarkable number of women in our study reported predisposing factors for VV, particularly those with non-infectious VV. Menopause and the use of hormonal contraceptives were the most prevalent predisposing factors among women with non-infectious VV and infectious VV, respectively. This finding is consistent with evidence showing that a decrease in estrogen levels not only causes atrophy of the vaginal mucosa, but also induces changes in vaginal pH.1,15 Irrespective of the cause of VV, all women in our study attended the gynecology unit seeking alleviation of typical symptoms of VV, such as pruritus (the most prevalent), burning, and dysuria. Similarly, gynecological assessment showed signs, such as erythema, vaginal discharge, and edema, in a remarkable number of participants, particularly those with infectious VV. All of these signs and symptoms are unlikely to resolve spontaneously and, therefore, require therapeutic intervention.1,3 Furthermore, women with confirmed infectious VV require treatment with antibiotic/antifungal agents, depending on the microorganism. To assess the capability of a vaginal cream to improve symptoms, we decided to split our cohort of interest (i.e., women who attended the gynecology unit because of VV symptoms) into two distinct therapeutic scenarios as follows: women who used the product as the only treatment for VV and those who used it as adjuvant treatment to antimicrobial therapy for infectious VV.
In line with previous studies conducted with Zelesse,12,13 in our study, the score and prevalence of signs and symptoms were significantly decreased after treatment compared with baseline in women with non-infectious VV who had Zelesse only and in women with infectious VV who had adjuvant treatment to antimicrobial therapy. The scarcity of clinical trials, case mix of extract combinations, and heterogeneity of assessment of effectiveness of natural products for treating VV preclude indirect comparison of effectiveness based on the literature. 9 Furthermore, to the best of our knowledge, no parallel trials have directly compared such products. Because of the limited clinical evidence associated with natural products for VV, the rationale of their effectiveness must rely on pre-clinical data of their components (i.e., chamomile, burdock, and aloe vera in Zelesse cream). Chamomile extract is associated with anti-inflammatory activity, presumably by reducing production of tumor necrosis factor alpha, as observed in mice with lipopolysaccharide-induced inflammation. 16 Similarly, a remarkable body of literature supports the anti-inflammatory effects of burdock extract.17–19 Finally, aloe vera extract shows re-epithelizing activity, presumably through a local increase of collagen production. 20 Taken together, these activities are expected to stop local inflammation associated with VV and aid in restoring damaged tissue.
Of all symptoms associated with VV, pruritus is an important contributor to the morbidity burden of VV episodes owing to associated anxiety and impairment of sleep. 21 Pruritus was the most prevalent symptom in our cohort (91% and 100% of participants in the non-infectious and infectious VV groups, respectively). Despite the high baseline prevalence of this symptom, approximately 90% of women who were treated solely with Zelesse cream (i.e., non-infectious VV) reported absent or mild pruritus at the end of treatment. Consistent with the decline in severity and prevalence of pruritus in these women, all participants most frequently highlighted antipruritic activity as an outstanding feature of Zelesse cream.
Women with VV often present with chronic or recurrent episodes (nearly half of our study sample).2,6 These patients are likely to be repeatedly exposed to over-the-counter products for alleviating the symptoms of VV, 22 and their safety and tolerability profiles become of paramount importance. Nevertheless, most of the over-the-counter products marketed with this indication lack formal assessment of safety and tolerability. In our study, no remarkable treatment-related adverse events were reported, and most participants reported good or very good acceptability of Zelesse cream.
The main limitation of our study was the lack of placebo control, which prevented drawing strong conclusions regarding the efficacy of Zelesse cream. However, because Zelesse cream was already marketed, we wished to collect real-world information on its effect on the progression of signs and symptoms, as well as on patient-reported outcomes, such as acceptability and highlighted properties. Our interest in collecting real-world data also prompted us to recruit all consecutive women attending the gynecology unit. This resulted in a highly heterogeneous study sample, such as the range in age and days from the last menstruation. Although this sample is likely to reflect the actual scenario encountered in day-to-day practice, future studies should address the clinical outcomes of Zelesse cream in specific groups of adult women. Finally, no sample size calculation was performed to assess the study outcomes. Therefore, the limited number of participants might have affected the statistical significance of some of the analyses.
Conclusions
Our study shows that Zelesse cream is well tolerated and reduces the patient-reported severity of signs and symptoms of VV when used either as monotherapy for non-infectious VV or adjuvant treatment to antimicrobial therapy for infectious VV. Our findings should encourage the use of Zelesse cream in routine management of women with signs and symptoms of VV. Future randomized, placebo-controlled trials with a larger sample size should be conducted to characterize the efficacy of Zelesse cream and provide more information on the type of patient who can benefit most from this treatment.
Footnotes
Acknowledgements
The authors would like to thank Dr. Borja Caballero, Dr. Cristina Guillán, Dr. Isabel Ramírez, Dr. Carloggero Jose Pace, Dr. Armand Marti Laporte, Dr. Susana Rodríguez Falcón, Dr. María Mercedes Falcón Martín, Dr. Ma Ángeles Calleja García, and Dr. Sonsoles Andreu Álvarez for their contribution of data acquisition. BioClever 2005 S.L. and i2e3 Procomms provided statistical and medical writing support, respectively.
Declaration of conflicting interest
MP, SV-O, and CN are employees of ITF Research Pharma (Medical Department). ARM, LMSF, and EV declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by ITF Research Pharma S.L.U.
Author contributions
ARM contributed to acquisition and analysis of data, and writing of the first draft of the manuscript. LMSF contributed to the conception and design of the study, acquisition and analysis of data, and writing of the first draft of the manuscript. EV contributed to the acquisition and analysis of data, and writing of the first draft of the manuscript. SV-O contributed to conception and design of the study, analysis and interpretation of data, writing of the first draft of the manuscript, and critical review of the manuscript. MP contributed to conception and design of the study, analysis and interpretation of data, writing of the first draft of the manuscript, and critical review of the manuscript. CN contributed to conception and design of the study, analysis and interpretation of data, and critical review of the manuscript.
