Abstract
Objective
The association between serum lipid levels and clinical outcomes following intracerebral hemorrhage remains unclear.
Methods
Data from consecutive patients hospitalized with intracerebral hemorrhage were prospectively collected between May 2005 and May 2018 and retrospectively analyzed. We documented the participants’ baseline characteristics, clinical features, risk factors, routine laboratory test results, and outcomes at 12 months post-intracerebral hemorrhage.
Results
A total of 1450 patients with intracerebral hemorrhage were included in the final analysis. In the unadjusted model, triglyceride levels were negatively associated with the risk of poor outcomes when analyzed as a continuous variable (odds ratio, 0.83; 95% confidence interval: 0.73–0.94; P = 0.005) and a categorical variable (comparing levels above the median vs. below the median; odds ratio, 0.658; 95% confidence interval: 0.532–0.820; P < 0.001). Consistent results were observed in the fully adjusted model: triglyceride levels remained significantly negatively associated with unfavorable outcomes (continuous variable: odds ratio, 0.87; 95% confidence interval: 0.760–0.999; P = 0.049 and quartile-based categorical variable: odds ratio, 0.699; 95% confidence interval: 0.546–0.893; P = 0.005). Restricted cubic spline analysis revealed linear and nonlinear relationships between triglyceride levels and outcomes (P for linearity = 0.0005 and P for nonlinearity = 0.0075), with the most pronounced trend observed in patients aged 70–100 years.
Conclusions
Low admission triglyceride levels are an independent predictor of poor 12-month outcomes in patients with intracerebral hemorrhage.
Introduction
Intracerebral hemorrhage (ICH) represents the most severe subtype of stroke, accounting for 10%–15% of all stroke cases. 1 Its incidence ranges from 10 to 30 per 100,000 individuals annually. Despite advances in treatment strategies, ICH remains responsible for 50% of stroke-related mortality and long-term disability.2,3 Although triglyceride (TG) levels have been linked to an elevated risk of ischemic stroke,4,5 the association between TG levels and ICH remains poorly understood.
Several large cohort studies have reported that low TG levels are associated with an increased risk of hemorrhagic stroke but a decreased risk of ischemic vascular events.6,7 Some cohort studies have reported that TG levels exert minimal influence on the prognosis of patients with ischemic stroke, 8 whereas others have demonstrated an inverse relationship between TG levels and unfavorable outcomes in patients with spontaneous ICH.9–11 For instance, a study analyzed the effect of TG concentrations within the first 48 h post-ICH on in-hospital mortality, adjusting for potential confounding variables. It found that low serum TG levels (P = 0.011; hazard ratio (HR) = 0.986; 95% confidence interval (CI): 0.976–0.997 mg/dL) were independently associated with higher in-hospital mortality following ICH. 12 Additionally, some reports have suggested that low TG levels correlate with an increased number of deep cerebral microbleeds, 13 an independent risk factor for ICH. Although conflicting findings exist, some studies14,15 have reported that patients with ICH have higher serum TG levels than healthy controls, whereas others have found no significant association between TG levels and spontaneous ICH.16,17 Therefore, this study aimed to investigate the association between serum TG levels and outcomes in patients with spontaneous ICH.
Patients and methods
Study participants
This was a single-center retrospective study. Data were extracted from the Stroke Registry System of Tianjin Huanhu Hospital, the largest specialized neurological hospital in Tianjin, China. All consecutive patients diagnosed with ICH and enrolled in the Stroke Registry System between May 2005 and May 2018 were initially considered for inclusion in the study. ICH diagnosis was made in accordance with the World Health Organization (WHO) criteria, and all diagnoses were confirmed via brain computed tomography (CT) and reviewed by two independent, blinded neurologists (WJZ and QL).
Inclusion criteria were as follows: (a) age ≥18 years and (b) presentation to the hospital emergency department with an imaging-confirmed ICH diagnosis within 72 h of symptom onset or the time the patient was last known to be well. Exclusion criteria were as follows: (a) presence of subarachnoid hemorrhage, traumatic brain injury, brain tumor, vascular malformation, hemorrhagic transformation of cerebral infarction, or any other cause of secondary ICH and (b) missing data on serum lipid levels.
All patient information was deidentified. The study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024) and reported following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 18 The study protocol was approved by the Medical Research Ethics Committee of Tianjin Huanhu Hospital, and all participants provided written informed consent prior to enrollment.
Data collection and group assignments
No lipid-lowering medications were administered to patients with ICH after admission, irrespective of their use prior to admission. For included patients, data on baseline characteristics, clinical features, risk factors, routine laboratory test results, and outcomes at 12 months post-ICH onset were collected.
Clinical features analyzed included sex, age, educational level, neurological deficits, hematoma location, risk factors, and laboratory data. Neurological deficits were assessed using the National Institutes of Health Stroke Scale (NIHSS), Barthel Index (BI), and modified Rankin Scale (mRS) scores on admission. Serum lipid levels at admission were also collected. Venous blood samples were obtained from all patients at approximately 6:00 a.m. on the day after admission. Fasting TG levels were measured using standardized enzymatic methods (Hitachi Analyzer, Roche Diagnostics, Medical Biochemical Analyzer-LABOSPECT 008), and the precision and accuracy of lipid measurements met WHO standards. Risk factors were defined as follows: (a) History of stroke. Self-reported prior stroke diagnosis; (b) Hypertension. Self-reported hypertension history or current use of antihypertensive medications; (c) Diabetes mellitus (DM). Self-reported DM history or current use of hypoglycemic medications; and (d) Atrial fibrillation (AF). Self-reported AF history confirmed by at least one electrocardiogram (ECG) or AF detected during hospitalization.
Lifestyle factors were also recorded, which are as follows: (a) Current smoking. Smoking ≥1 cigarette per day for ≥1 year and (b) Alcohol consumption. Consuming ≥1 drink per week for ≥1 year. Patients were divided into two groups based on the median TG level: <1.26 mmol/L and ≥1.26 mmol/L. For subgroup analysis, patients were further stratified into three age groups: 20–50 years, 50–70 years, and 70–100 years.
Outcomes measurement and follow-up
Functional outcomes at 12 months post-ICH onset were evaluated using mRS. 19 Patients with an mRS score >2 were classified as having unfavorable outcomes, whereas those with an mRS score ≤2 were classified as having favorable outcomes.
Outcome data were collected through either in-person examinations or telephonic interviews, following a standardized protocol. Two trained neurologists conducted outpatient re-evaluations for all patients 12 months after ICH. All patients completed follow-up; most were assessed via face-to-face interviews, and those unable to attend in-person follow-up completed it through telephonic interviews.
Statistical analysis
Continuous variables with a normal distribution were expressed as mean ± SD, whereas those with a skewed distribution were expressed as median (interquartile range (IQR)). Categorical variables were presented as counts (percentages). TG levels were analyzed as a continuous variable and a quartile-based categorical variable.
Continuous variables were compared using the Mann–Whitney U test or unpaired t-test, as appropriate. Dichotomous variables were compared using the chi-squared test. Two logistic regression models were constructed to explore the association between TG levels and ICH outcomes. Multivariate logistic regression was used to assess the association between TG levels and 12-month unfavorable outcomes. Inverse probability weighting (IPW) was applied to account for potential imbalances in baseline characteristics across different TG level groups.
A restricted cubic spline (RCS) model with 3 knots was used to examine potential nonlinear associations between TG levels and ICH outcomes. The number of knots (n = 3) was chosen to minimize the Akaike Information Criterion (AIC). RCS analysis was adjusted for all covariates. All associations in the logistic regression models were reported as odds ratios (ORs) with 95% CIs. A two-tailed P-value <0.05 was considered statistically significant for the univariate analysis. All statistical analyses were performed using R software (version 4.1.1; R Foundation for Statistical Computing; Vienna, Austria).
Results
A total of 1856 patients with ICH were initially identified from the Stroke Registry System during the study period, of which 1450 met the inclusion criteria. The participant selection flowchart is presented in Figure 1, and the baseline characteristics and risk factors of the included patients are summarized in Table 1.

Flowchart of included patients with ICH. ICH: intracerebral hemorrhage; sICH: symptomatic intracerebral hemorrhage; SAH: subarachnoid hemorrhage.
Characteristics of participants stratified by different outcome of sICH.

sICH: symptomatic intracerebral hemorrhage; NIHSS: National Institute of Health stroke scale; mRS, modified Rankin Scale; SBP, systolic blood pressure; DBP, diastolic blood pressure; hCRP, high-sensitivity C-reactive protein; Q1: quartile 1; Q3: quartile 3.
Of the 1450 included patients, 541 (37.3%) had unfavorable outcomes and 909 (62.7%) had favorable outcomes. Patients with unfavorable outcomes were more likely to be female and have lower educational levels. Additionally, patients with deep hematomas or hematomas in ≥2 locations were more prone to poor outcomes. An association between high-density lipoprotein (HDL) levels and poor outcomes was also observed.
Table 2 presents the results of the logistic regression models examining the association between TG levels and unfavorable outcomes. In the unadjusted model (Model 1), TG levels were negatively associated with the risk of poor outcomes when analyzed as a continuous variable (OR: 0.831, 95% CI: 0.732–0.943; P = 0.005) and as a categorical variable (comparing above vs. below the median; OR: 0.658, 95% CI: 0.532–0.820; P < 0.001).
Association of TG levels with unfavorable outcome in patients with sICH.
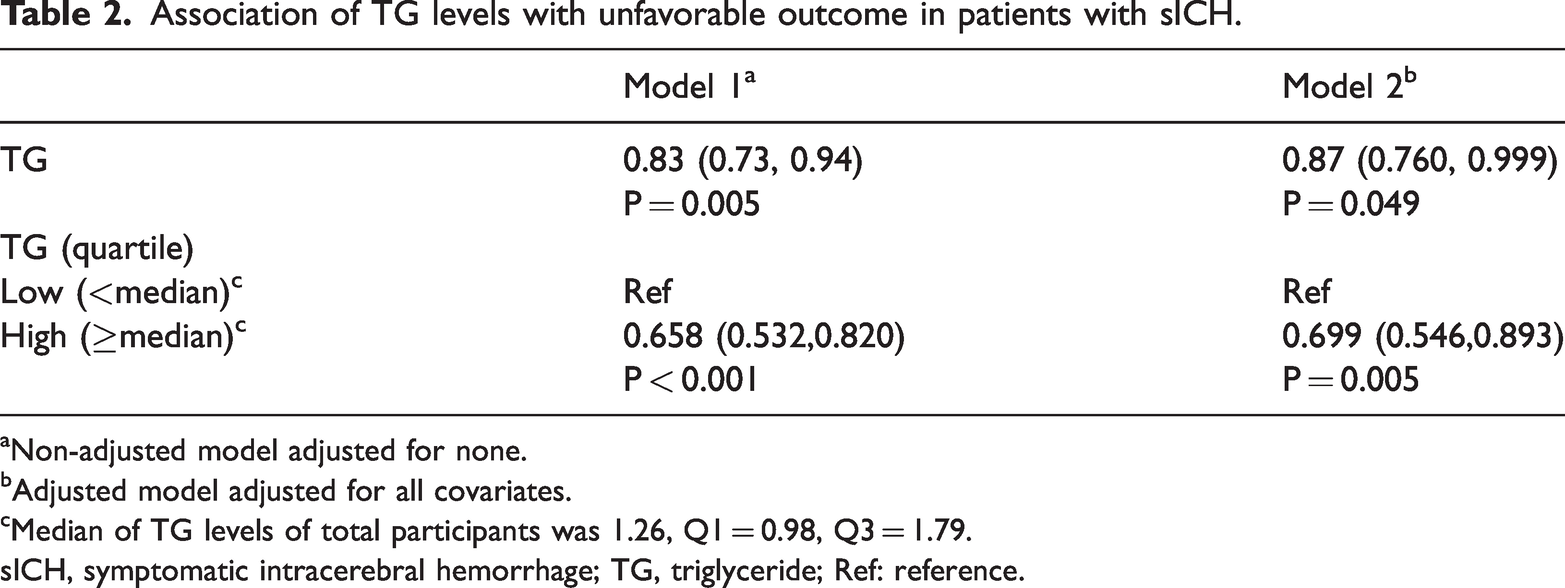
Non-adjusted model adjusted for none.
Adjusted model adjusted for all covariates.
Median of TG levels of total participants was 1.26, Q1 = 0.98, Q3 = 1.79.
sICH, symptomatic intracerebral hemorrhage; TG, triglyceride; Ref: reference.
Consistent results were observed in the fully adjusted model (Model 2). TG levels remained significantly negatively associated with unfavorable outcomes (continuous variable: OR, 0.87; 95% CI: 0.760–0.999; P = 0.049 and quartile-based categorical variable: OR, 0.699; 95% CI: 0.546–0.893; P = 0.005).
When analyzed as a categorical variable, TG levels were also negatively associated with the risk of unfavorable outcomes (Figure 2). Figure 2 depicts the associations between baseline characteristics, risk factors, and unfavorable outcomes, as determined by multiple logistic regression. After adjusting for potential confounders, TG levels, coronary heart disease, and diastolic blood pressure (DBP) were identified as independent predictors of poor 12-month functional outcomes following ICH. Particularly, lower TG levels were significantly and independently associated with a higher risk of 12-month unfavorable outcomes (OR: 0.87, 95% CI: 0.76–1.00; P = 0.049).

Associations between patient characteristics and unfavorable ICH outcomes. ICH: intracerebral hemorrhage; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; DBP: diastolic blood pressure.
IPW was used to further control for confounding effects. Figure 3 shows a comparison of the baseline characteristics before and after weighting, demonstrating that IPW effectively reduced variability in variables across the TG level quartiles.

Confounding effect control among covariates using the inverse probability weighting method. NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; DBP: diastolic blood pressure.
RCS analysis was performed to explore the linear and nonlinear associations between TG levels and functional outcomes. The test for linearity yielded a P-value of 0.0005, confirming a significant linear relationship between TG levels and poor outcomes. The test for nonlinearity also yielded a significant P-value (0.0075), indicating a nonlinear association. As shown in Figure 4, the RCS curve exhibited an overall downward trend, confirming a negative association between TG levels and poor ICH outcomes.

Restricted cubic spline analysis of the association between TG levels and unfavorable ICH outcomes. TG: triglyceride; ICH: intracerebral hemorrhage; OR: odds ratio; 95% CI: 95% confidence interval.
Subgroup analysis using RCS was conducted to explore age-specific associations between TG levels and poor outcomes. Patients were divided into three age groups, and the RCS curve exhibited a downward trend in all age groups (Figure 5). The most pronounced trend was observed in the 70–100 years age group, indicating that older patients with lower TG levels were more likely to experience poor outcomes after ICH. However, the underlying mechanism warrants further investigation.

Restricted cubic spline analysis of the association between TG levels and unfavorable ICH outcomes by age group. TG: triglyceride; ICH: intracerebral hemorrhage; OR: odds ratio; 95% CI: 95% confidence interval.
Discussion
Using 12-month follow-up data from this large hospital-based cohort study, our results demonstrated an inverse association between serum TG levels and poor ICH outcomes in a fully adjusted multivariate analysis. Patients with low TG levels (<1.26 mmol/L) were more likely to experience unfavorable outcomes after ICH. This conclusion was further supported by additional analyses, including IPW and nonlinearity testing. Thus, low TG levels may represent an independent risk factor for unfavorable outcomes in patients with ICH. Furthermore, when stratified by age, the risk of poor outcomes exhibited a more significant downward trend with increasing TG levels in the 70–100 years age group, indicating that older patients with lower TG levels are at higher risk of poor outcomes following ICH.
However, few studies have focused on the association between TG levels and long-term ICH outcomes. Our study demonstrated that low TG levels are independently associated with an increased risk of 12-month unfavorable outcomes following ICH, a finding consistent with previous research. Feng et al. 10 measured serum TG and other lipid levels in 260 female patients and reported that low TG levels were independently associated with poor prognosis in female patients with spontaneous ICH. Roquer et al. 12 analyzed the impact of TG concentrations on in-hospital mortality following ICH and found that low TG levels within the first few hours post-ICH were a strong predictor of in-hospital mortality in patients with spontaneous supratentorial ICH.
However, conflicting findings exist. Some studies have failed to identify an association between TG levels and worse outcomes in spontaneous ICH.20,21 For instance, analyses from the Stroke Registry in Chang-Gung Healthcare System Study included only a small number of events and used nonfasting TG levels—factors that may have biased the results. 20 Noda et al. 22 reported no association between serum TG levels and ICH-related mortality. These discrepancies may reflect differences in study populations, highlighting the need for further research to validate our findings.
Additionally, we observed that older patients with lower TG levels are more likely to experience unfavorable ICH outcomes. TG levels are regulated predominantly by lifestyle factors rather than genetic factors 23 and may reflect the nutritional status of patients with ICH. Low TG levels may serve as a surrogate marker for nutritional deficiencies, a predictor of low serum albumin levels, 24 or an indicator of underlying debilitating diseases—all of which may increase the risk of unfavorable ICH outcomes. Thus, older individuals with low TG levels are more likely to be frail and have a poor prognosis. Future studies using time-varying analyses are needed to determine whether intraindividual fluctuations in TG levels and other confounders (e.g. age) affect study results.
The mechanisms by which low TG levels contribute to worse hemorrhagic stroke outcomes remain unclear. In vitro studies have shown that TG-rich lipoproteins can promote factor VII activation via activation of factor XII (XIIa) and the intrinsic coagulation pathway. 25 Other studies 26 have reported positive associations between factors VIIag and VIIc and serum TG levels, suggesting that serum TG levels are associated with factor VII protein production. Low TG levels after ICH may therefore lead to reduced factor VII protein levels, which could contribute to prolonged bleeding and poor outcomes.
Additionally, low TG levels may be associated with the presence of cerebral microbleeds, 9 an independent risk factor for future ICH recurrence. 27 Chen et al. 28 reported that cerebral microbleeds in patients with ICH (particularly in the basal ganglia region) are indicative of microangiopathy and are associated with vascular leakage and perihemorrhagic edema formation. Recent studies have also demonstrated that dietary triglycerides can influence serum fatty acid concentrations and their relative proportions, which may be associated with the age of ICH onset. 29 However, whether these mechanisms can explain the observed association between low TG levels and ICH outcomes remains unclear and warrants further investigation.
Study limitations
This study has several limitations. First, we did not continuously monitor the changes in TG levels during follow-up. A single baseline TG measurement may have underestimated the association between TG levels and ICH outcomes due to regression dilution bias. Second, although statins and anticoagulants may directly impact the study outcomes, 30 we did not collect data on their use, which may have affected the accuracy of our results. Third, this was a single-center retrospective study; therefore, we could not establish a causal relationship between TG levels and ICH outcomes. Fourth, given the potential impact of TG on the coagulation system, the exclusion of patients with coagulopathy may have introduced selection bias. Finally, the lack of data on lipid-lowering therapy may have influenced the evaluation of outcomes.
Conclusions
This cohort study demonstrated that low TG levels are associated with long-term unfavorable outcomes in patients with ICH. Older patients are more likely to exhibit lower TG levels and higher rates of unfavorable outcomes. Clinicians should pay particular attention to older patients with low TG levels following ICH to prevent poor outcomes. Low TG levels may serve as a predictor of unfavorable outcomes in patients with ICH, and TG levels may help identify high-risk patients, particularly older patients with ICH. Large prospective studies are needed to elucidate the mechanism underlying the association between TG levels and ICH outcomes.
Footnotes
Acknowledgments
The authors thank the staff of the Stroke Registry System at Tianjin Huanhu Hospital for their support in data provision. No artificial intelligence (AI) tools were used in the conduct of this research.
Author contributions
Conceptualization: Qian Liu, Jiannan Yu; Data curation: Xunxiao Li; Formal analysis: Luming Chen; Methodology: Na Li; Writing-original draft: Qian Liu; Writing-review & editing: Qian Liu, Wenjuan Zhao.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no relevant financial or non-financial interests.
Disclaimers
The authors have no relevant financial or non-financial interests to disclose.
Funding
This work was supported by the National Clinical Key Specialty Construction Project (Comprehensive treatment of Cerebrovascular Diseases), Tianjin Key Medical Discipline (Specialty) Construction Project (No. TJYXZDXK-052B), Tianjin Education Commission Scientific Research Project (No. 2023KJ061), and Xinjiang Uygur Autonomous Region Health Commission Special science and technology project of young medical talents (No. WJWY202417).
Statement
This study has been reviewed by the relevant ethics review board, and an exemption has been granted.
